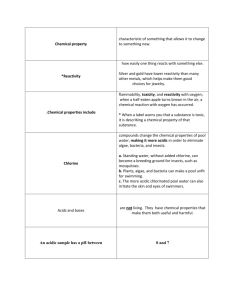
Solutions and Acids
advertisement

Chem 1 Solutions and Acids and Bases Practice Quiz 1. Acetic acid is found in significant quantities in a) lemons b) vinegar c) sour milk d) apples 2. Acids generally release H2 gas when they react with a) nonmetals b) semimetals c) metals d) glass 3. Acids react with a) bases to produce salts and water b) salts to produce bases and water c) water to produce bases and salts d) neither bases, salts, nor water 4. Bases feel a) rough b) moist c) slippery d) dry 5. Which acid is used in batteries? a) hydrochloric acid b) phosphoric acid c) nitric acid d) sulfuric acid 6. What acid is produced in the stomach? a) hydrochloric acid b) phosphoric acid c) nitric acid d) sulfuric acid 7. What acid is found in vinegar? a) acetic acid b) nitric acid c) phosphoric acid d) hydrochloric acid 8. According to the traditional definition, an acid contains a) hydrogen and does not ionize b) hydrogen and ionizes to form hydrogen ions c) oxygen and ionizes to form hydroxide ions d) oxygen and ionizes to form oxygen ions 9. A substance that ionizes nearly completely in aqueous solution and produces H 3O+ is a a) weak base b) strong base c) weak acid d) strong acid 10. Which of the following is NOT a strong acid? a) HNO3 b) HC2H3O2 c) H2SO4 d) HCl 11. Hydroxides of Group 1 metals a) are all strong bases b) are all weak bases c) are all acids d) may be either strong or weak bases 12. Which of the following is a weak base? a) KOH b) Ca(OH)2 c) NH3 d) HCl 13. Which compound is produced by a neutralization? a) H2O(l) b) HNO3(aq) c) Ca(OH)2(s) d) H3PO4(aq) 14. Which of the following gases does NOT dissolve in atmospheric water to produce acidic solutions? a) SO2 b) NO2 c) O2 d) CO2 15. The reaction of an acid with a carbonate does NOT produce a) a salt. b) water. c) carbon dioxide. d) oxygen. 16. What is the concentration of H3O+ in pure water? a) 10-7 M b) 0.7 M c) 55.4 M d) 107 M 17. Which expression represents the pH of a solution? a) log[H3O+] b) -log[H30+] c) log[OH-] d) -log[OH-] 18. If [H3O+] of a solution is greater than the [OH-], the solution a) is always acidic. b) is always basic. c) is always neutral. d) might be acidic, basic, or neutral. 19. The pH scale in general uses ranges from a) 0 to 1. b) -1 to 1. c) 0 to 7. d) 0 to 14. 20. The pH of an acidic solution is a) less than 0. b) less than 7. c) greater than 7. d) greater than 14. 21. A water solution whose pH is 4 a) is always neutral. b) is always basic. c) is always acidic. d) might be neutral, basic, or acidic. 22. If [H3O+] = 8.26 x 10-5 M, what is the pH of the solution? a) 2.161 b) 3.912 c) 4.083 d) 8.024 23. What is the hydronium ion concentration of a solution whose pH is 4.12? a) 4.4 x 10-8 b) 5.1 x 10-6 c) 6.4 x 10-5 d) 7.6 x 10-5 24. In an acid-base titration, equivalent quantities of hydronium ions and hydroxide ions are present a) at the beginning point b) at the midpoint c) at the endpoint d) throughout the titration 25. What is the molarity of an HCl solution if 50.0 mL is neutralized in a titration by 40.0 mL of 0.400M NaOH? a) 0.200M b) 0.280M c) 0.320M d) 0.500M 26. What is the molarity of an NaOH solution if 4.37 ml is titrated to neutralization by 11.1 ml of 0.0904M HNO3? a) 0.230M b) 0.355M c) 0.460M d) 0.620M 27. Calculate the volume of a 0.0347M Ba(OH)2 solution that is completely neutralized by 261 ml of 0.505M HNO3? a) 3810 ml b) 2600 ml c) 2050 ml d) 1900 ml 28. If 72.1 ml of 0.543 M H2SO4 completely neutralizes 39.0 ml of KOH solution, what is the molarity of the KOH solution? a) 0.317M b) 0.502M c) 1.00M d) 2.01M 29. What is the molarity of an H3PO4 solution if 358 ml is completely neutralized by 876 ml of 0.0102 M Ba(OH)2 solution? (Hint - I am asking for the H3PO4 solution concentration, not the H+ concentration!) a) 0.0111M b) 0.0166M c) 0.0500M d) 0.0333M ----------Key---------1. (b) 2. (c) 3. (a) 4. (c) 5. (d) 6. (a) 7. (a) 8. (b) 9. (d) 10. (b) 11. (a) 12. (c) 13. (a) 14. (c) 15. (d) 16. (a) 17. (b) 18. (a) 19. (d) 20. (b) 21. (c) 22. (c) 23. (d) 24. (c) 25. (c) 26. (a) 27. (d) 28. (d) 29. (b)