Supplementary Information
advertisement

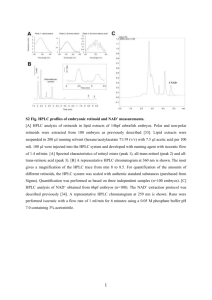
SUPPLEMENTARY MATERIAL FOR Arsenolipids in oil from blue whiting Micromesistius poutassou – evidence for arsenic-containing esters Mojtaba S. Taleshi1,2, Georg Raber1, John S. Edmonds1, Kenneth B. Jensen1, and Kevin A. Francesconi1* 1 Institute of Chemistry-Analytical Chemistry, University of Graz, Universitaetsplatz 1, 8010 Graz, Austria 2 Department of Marine Chemistry, Faculty of Marine Science, University of Mazandaran, Babolsar, Iran *Corresponding author kevin.francesconi@uni-graz.at The following Supplementary material accompanies this paper: Figure S1. Classification of arsenolipids. Figure S2. Flow diagram for fractionation of arsenolipids in blue whiting oil. Figure S3. HPLC/ESI-MS chromatogram of arsenolipids in the isopropanol extract of bluewhiting oil, post-silica column. Figure S4. High resolution accurate mass spectrum for As-HC440 in blue-whiting oil. Figure S5. High resolution accurate mass spectrum for As-HC442 from blue-whiting oil. Figure S6. High resolution accurate mass spectrum for As-HC444 in blue-whiting oil. Figure S7. High resolution accurate mass spectrum for As-HC542 in blue-whiting oil. Figure S8. HPLC/ESI chromatograms of As-HC444). Figure S9. High resolution mass spectrum for synthesized As-HC 444. Figure S10. HPLC/ICPMS chromatogram of the less polar arsenolipids (iso-propanol fraction) of the blue-whiting oil. Figure S11. HPLC/ESI-MS chromatograms of thio analogues of arsenolipids 1 Arsenolipids Polar Less polar Non-polar (As fatty acids + smaller As hydrocarbons) (As hydrocarbons + unknowns) (Unknown structures) 40000 Arsenic-containing hydrocarbons Arsenic-containing fatty acids intensity 30000 Low-polarity arsenolipids (unknown) 20000 10000 0 0 10 20 30 40 50 60 70 80 90 Time (min) Figure S1. Classification of arsenolipids. On the basis of their extraction by solvent partitioning, and their retention on reversed-phase HPLC, arsenolipids in fish oils can be categorized into three broad polarity groups: polar (mainly arsenic-containing fatty acids), less-polar (mainly arsenic-containing hydrocarbons), and non-polar (containing unknown arsenolipids). In this example of crude blue whiting oil, HPLC conditions were: Atlantis dC18 (1501.0 mm, 5 µm) at 30 oC and a mobile phase comprising a mixture of 10 mM NH4OAc pH 6.0 and ethanol at a flow rate of 100 µL min-1. The chromatography was performed with linear gradient elution: 060 min for 35%95% ethanol). 2 Blue-Whiting Oil 2.16 µg As/g 200.7 g, 434 2 µg As Dissolved in hexane (500 mL) Extracted with MeOHaq (300 mL) Hexane (1) fraction MeOH (1) fraction 199 g, 333 7 µg As 1.0 g, 103 6 µg As MeOHaq (300 mL) DEAE Sephadex MeOH (2) fraction Hexane (2) fraction 1.0 g, 45 2 µg As 195 g, 297 7 µg As Basic/neutral fraction Acidic fraction 145 mg, 67 3 µg As 170 mg, 14 0.3 µg As EtOHaq (300 mL) EtOH fraction Hexane (3) fraction 1.2 g, 37 1 µg As 190 g, 260 4 µg As Hexane (3) fraction 184 g, 252 3 µg As Isopropanolaq (IPA) (2300 mL) IPA fraction Hexane (4) fraction 9.5 g, 130 2 µg As 173 g, 143 9 µg As Figure S2. Flow diagram for fractionation of arsenolipids in blue whiting oil. 3 a) Measured in SIM mode at m/z 91 60000 E Intensity 40000 20000 A B C D F 0 0 5 10 15 20 25 30 35 40 45 50 Time (min) + b) Measured at [M+H] 150000 Arsenolipid 542 Arsenolipid 442 Intensity 100000 Arsenolipid 444 Arsenolipid440 50000 0 0 5 10 15 20 25 30 35 40 45 50 Time (min) Figure S3. HPLC/ESI-MS chromatogram of arsenolipids in the isopropanol extract of blue-whiting oil, post-silica column. a) Measured at m/z 91 with fragmentor voltage 400 V and b) Performed in scan mode (m/z 100-1000) with fragmentor voltage 150 V and the masses were extracted from scan mode. HPLC conditions: Atlantis dC18 (150 × 4.6 mm, 5 µm) at 30 oC and a mobile phase comprising a mixture of 92% ethanol-buffer (100 mM NH4OAc, pH 5.0) (85+15, v/v) and 8% chloroform at a flow rate of 0.5 mL min-1 (isocratic elution). 4 O H3C As CH3 8 As-HC440 kbj09070808_108_Recal_triton381 #1 RT: 0,01 T: FTMS + c NSI Full ms [120,00-1000,00] 441,3075 100 AV: 1 NL: 1,05E6 95 Arsenolipid 440 90 85 80 75 70 65 60 55 50 45 40 35 30 443,3141 25 20 467,1022 15 10 455,3346 413,2664 416,3737 432,2383 457,3024 473,2973 5 487,3609 423,3236 495,3296 0 410 420 430 440 450 m/z 460 470 480 490 500 Figure S4. High resolution accurate mass spectrum for As-HC440 in blue-whiting oil. Molecular formula C25H49AsO: calculated for [M+H]+ 441.3077; found 441.3075; Δm/m = 0.5 ppm. 5 O H3C As 9 CH3 As-HC442 kbj09070806_108_Recal #72-146 RT: 1,36-2,44 T: FTMS + c NSI Full ms [150,00-2000,00] 215,1260 100 AV: 75 NL: 1,98E7 443,3235 95 239,1623 90 Arsenolipid 442 381,2981 85 80 75 70 65 60 353,2667 55 50 393,2981 45 40 35 30 309,2042 25 20 301,1416 15 255,1572 10 273,1678 467,1026 5 325,2355 416,3740 487,3612 0 250 300 350 m/z 400 450 Figure S5. High resolution accurate mass spectrum for As-HC442 from blue-whiting oil. Molecular formula C25H51AsO: calculated for [M+H]+ 443.3234; found 443.3235; Δm/m < 0.3 ppm. 6 O H3C As 10 CH3 AS-HC444 kbj09070807_108_Recal_triton381 #1 RT: 0,01 AV: 1 T: FTMS + c NSI Full ms [120,00-1000,00] 445,3385 100 NL: 5,95E6 95 90 85 80 Arsenolipid 444 75 70 65 60 55 50 45 40 35 30 447,9147 25 20 462,1466 471,3542 15 10 5 418,3528 404,3734 482,3243 437,2663 487,2795 0 400 420 440 460 m/z 480 500 515,3921 520 Figure S6. High resolution accurate mass spectrum for As-HC444 in blue-whiting oil. Molecular formula C25H53AsO: calculated for [M+H]+ 445.3390; found 445.3385; Δm/m = 1.1 ppm. 7 O H3C As 11 CH3 As-HC542 kbj09070805_108_Recal_triton381 #1 RT: 0,01 AV: 1 T: FTMS + c NSI Full ms [150,00-2000,00] 543,3545 100 NL: 5,91E6 95 Arsenolipid 542 90 85 80 75 70 65 60 55 50 45 40 544,3578 35 30 25 20 536,1658 15 556,4939 10 531,3544 5 537,1647 522,3558 555,4236 545,3611 559,3496 565,3364 550,4675 575,3446 0 520 530 540 550 m/z 560 570 Figure S7. High resolution accurate mass spectrum for As-HC542 in blue-whiting oil. Molecular formula C33H55AsO: calculated for [M+H]+ 543.3547; found 543.3545; Δm/m = 0.4 ppm. 8 O H3C As 10 CH3 AS-HC444 (synthesised sample) kbj_AsLipid_445_+1pmPPG_090929174233_Recal #204-219 T: FTMS + p NSI Full ms [100,00-1000,00] 445,3389 100 RT: 2,97-3,11 AV: 16 NL: 7,87E6 95 90 85 80 75 70 65 447,2929 60 55 50 45 40 35 30 25 20 448,2963 15 10 5 0 435,3457 435 441,3380 440 449,2986 450,3010 443,3234 445 450 455,3162 457,3018 455 m/z Figure S8. High resolution mass spectrum for synthesized As-HC 444. Molecular formula C25H53AsO: calculated for [M+H]+ 445.3390; found 445.3389; Δm/m < 0.3 ppm. 9 Figure S9. HPLC/ESI-MS chromatograms of As-HC444). Purified As-HC444 from the isopropanol-fraction of blue-whiting oil (gray line) and the same sample spiked with synthesized As-HC444 (solid line). HPLC conditions: Atlantis dC18 (150 × 1.0 mm, 5 µm) at 30 oC and a mixture of 85% of ethanol-buffer (100 mM NH4OAc, pH 5.0) (9+1) and 15% chloroform (isocratic elution) at a flow rate of 100 µL min-1. Selected ion monitoring was performed for [M+H]+ at m/z 445 with fragmentor voltage 150 V. 10 Figure S10. HPLC/ICPMS chromatogram of the less polar arsenolipids (iso-propanol fraction) of the blue-whiting oil. a) Pre-silica column and b) post-silica column. The newly appeared arsenolipids showed peaks between those of arsenic-containing fatty acids (e.g. As-FA362) and arsenic-containing hydrocarbons (e.g. As-HC332). HPLC conditions, for both a) and b) were: Atlantis dC18 (1501.0 mm, 5 µm) at 30 oC and a mobile phase comprising a mixture of 10 mM NH4OAc pH 6.0 and ethanol at a flow rate of 100 µL min-1. The chromatography was performed with linear gradient elution: 060 min for 35%95% ethanol. 11 Figure S11. HPLC/ESI-MS chromatograms of thio analogues of arsenolipids. (a) polar fraction of blue whiting oil post-silica column after elution with H2S/acetone; and (b) thioarsenolipid standards prepared by bubbling of H2S gas into standard solutions of oxo arsenolipids As-FA362, As-FA436, As-FA390, and As-HC404. HPLC conditions: Atlantis dC18 (150 × 1.0 mm, 5 µm) at 30 oC and a mobile phase comprising a mixture of 10 mM NH4OAc (pH 6.0) and ethanol at a flow rate of 100 µL min-1. The chromatography was performed with linear gradient elution: 060 min with 35%95% ethanol. Selected ion monitoring was performed for [M+H]+ at m/z 379, 453, 407, 421 and 377 with fragmentor voltage 150 V. The slight differences in retention times are attributed to small changes in column performance between the HPLC runs. 12