Detailed Methods
advertisement

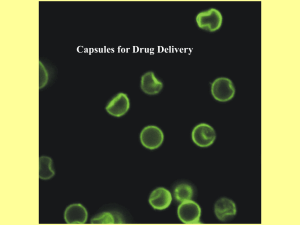
Electronic Supplementary Material: Detailed Methods Production of capsules. 14 wt% of PMSQ solution was prepared by mixing appropriate amounts of polymer with ethanol and stirring for 900 s at ambient temperature (21 °C), pressure and humidity. The source and relevant properties of the PMSQ are previously described [12,13, see main paper bibliography for these references]. 90 mg of gentamicin sulfate USP (Sigma-Aldrich) was dissolved in 3 ml of MilliQ-purified, deionized water (100 mg/ml). For the preparation of encapsulated structures, whereby a secondary material is incorporated into a polymeric particle, we used a CEHDA processing system (Fig. 1A). In the CEHDF set-up, the needles were connected via a silicone tube to plastic syringes (BD PlasticTM, VWR, Lutterworth, UK) containing the solutions. The flow rates of each solution through the needles were controlled by high-precision Harvard syringe pumps (Harvard Apparatus Ltd., Edenbridge, UK). The polymer and drug solutions were pumped into the device at inner and outer needle flow rates of 650 and 150 μl/min, respectively. This optimized combination of flow rates was determined experimentally to provide a suitable compromise between jet stability and the required encapsulation efficiency. Electric field was applied via a high voltage DC power supply unit (FC30 P4 12w, Glassman Europe Limited, Bramley, UK). Once the electric field was applied, the body of the droplet was charged resulting in a fine jet emerging from the needle apex. The output voltage range of the unit was 5-8 kV and the output current range was up to 2 mA. This subsequently led to the formation of encapsulated structures, which were collected in a glass vial containing sterilized water. As a control, we also prepared drug-free control capsules in which only the outer needle was used for the flow of just the polymer solution, with all other processing conditions the same. For live cell tracing experiments, we prepared capsules loaded with fluorescein isothiocyanate (FITC), a fluorescent tracker dye, in place of gentamicin. 20 mg/ml of FITC was added to 14 wt% of PMSQ solution and stirred for 2 h to ensure homogenous mixing of the solution. The solution was then sprayed, as described earlier, at a voltage of 5.5 kV and flow rate of 650 µl/min and then collected at a distance of 100 mm in a glass vial containing distilled water. The resultant suspension of polymer-encapsulated dye was then freeze-dried overnight to obtain powdered particles which were stored in the dark at 4°C until use. Electron microscopy. For scanning EM (SEM), samples were collected on a glass cover slip and dried for 1 h prior to imaging. Following drying, samples were gold-coated for 2 min via gold sputtering (Edward sputter coater S 1 50 B) to improve the conductivity of the sample surface. SEM studies were performed on the JEOL JSM-6301F field emission SEM. For transmission EM (TEM) imaging, we prepared samples by collecting drops of the suspension of water containing the capsules on a formvar/carbon 400 mesh copper TEM grids and air-drying prior to analysis. TEM studies were performed on the JEOL- 2000FX TEM, with an operating voltage of 80 kV. Antimicrobial assay. We prepared a broth culture of a strain of the common uropathogen Enterococcus faecalis. The bacteria were grown on fresh chromogenic CPS3 agar plates (bioMérieux) under aerobic conditions for 24 hours at 37°C, after which a colony was resuspended in fresh mammalian cell culture medium (CO2 independent media [Gibco]) at a concentration of 4 x 107 colony-forming units (CFU)/mL. We compared a dilution series of unencapsulated gentamicin (Genticin ® Injectible, Andipharm, 40 mg/mL) to a dilution series of the gentamicin-containing capsules, and to drug-free control capsules. We mixed the intervention solutions 1:1 with the working bacterial stock and incubated them aerobically in loose-capped tubes in an orbital shaking incubator at 37°C. At various times post-inoculation, an aliquot of each culture was removed and streaked onto Columbia Blood Agar plates (bioMérieux) to enumerate living bacteria after overnight incubation as above. Overgrown cultures where colonies could still be distinguished but not easily counted were set at 600 for Fig 1a and 800 for Figure 1b (which had a larger inspection zone); a lawn of bacteria whose individuals could not be detected at all were considered saturated and scored arbitrarily at 1000 CFU in both experiments. Cell biology experiments and cell staining. A transformed cell line, T24, derived from a human bladder carcinoma (a kind gift from Aled Clayton, Cardiff University) was maintained at 37°C in a humidified incubator under 5% CO2 in 9 cm dishes in RPMI 1640 medium (Gibco) supplemented with 10% fetal bovine serum (FBS) (PAA) and antibiotics (50 µg/ml penicillin and 50 mg/ml streptomycin; Gibco). For experiments, lyophilized microcapsules were resuspended in sterile complete RMPI medium and agitated for 1 hr at RT to facilitate resuspension. T24 cells were seeded the day before onto fetal bovine serum-coated glass coverslips so that cells were 80% confluent the next day. 80 µL of microcapsule suspension (approximately 5 x 104 particles/mL) was added to wells, or 80 µL of medium as a control. At various times post-treatment, coverslips were removed and fixed in freshly prepared 4% formaldehyde (Thermo Scientific, Fisher Scientific) in PBS at room temperature for 15 minutes. The formaldehyde was aspirated and the preparation washed three times with PBS. Next, cells were stained for 1 hr at RT with DAPI (4’6-diamidino-2-phenylindole, Sigma-Aldrich) at 1μg/ml in PBS to stain DNA, and red-fluorescent wheat germ agglutinin ((AlexafluorWGA-555, Invitrogen) in Hanks Balance Salt Solution (Invitrogen) to stain the outer cell membrane. Coverslips were washed 4x in PBS and mounted with FluorSave reagent (Calbiochem). Cells were imaged with a Leica SP5 confocal microscope and ImageJ 1.44P software. Ethical committee approval for human urine sampling was obtained from East Central London REC1 (Research Ethical Committee). Sampling was conducted at Professor James Malone-Lee’s outpatient clinic, University College London, Division of Medicine. The patients gave informed consent, and 30 mL of freshly harvested urine was centrifuged at 2000rpm, 600 rcf for 5 mins. Supernatant was discarded and the sediment was resuspended in 400 µL of CO2-independent media supplemented with 10% FBS. 80 µL of microcapsule suspension at 1.7 x 104 microcapsules/mL was added to the sediment solution and incubated in a orbital shaking incubator at 37°C for 2 hr. At the end of the incubation, 80μl of the suspension was cytocentrifuged onto a glass slide at 75 rcf for 5 minutes in a Shandon Cytospin 2. The resulting visible disc of urinary particulate on the slide was circumscribed with a hydrophobic barrier pen (ImmEdge pen, Vector Laboratories) and then fixed, stained and mounted as for T24 cells above, except we omitted the membrane stain. Samples were imaged using the Olympus CX-41, and the resulting images were processed using ImageJ 1.44P.