DIFFRACTION FROM CRYSTAL PLANES
advertisement

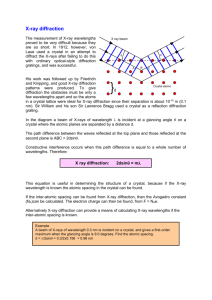
Crystal structures, unit cell, symmetry elements Powder X-Ray Diffraction INTRODUCTION X-rays are electromagnetic radiation of wavelength about 1 Å (10-10 m), which is about the same size as an atom. They occur in that portion of the electromagnetic spectrum between gamma-rays and the ultraviolet. The discovery of X-rays in 1895 enabled scientists to probe crystalline structure at the atomic level. When certain geometric requirements are met, X-rays scattered from a crystalline solid can constructively interfere, producing a diffracted beam. In 1912, W. L. Bragg recognized a predictable relationship among several factors. Fig. 1 Reflection of x-rays from two planes of atoms in a solid. The path difference between two waves: 2x = 2dsin(theta) For constructive interference nλ = 2dsinθ Bragg equation X-ray diffraction has been in use in two main areas, for:1. Fingerprint characterization of crystalline materials and 2. The determination of their structure. Each crystalline solid has its unique characteristic X-ray powder pattern which may be used as a "fingerprint" for its identification. Once the material has been identified, X-ray crystallography may be used to determine its structure, i.e. how the atoms pack together in the crystalline state and what the interatomic distance and angle are etc. X-ray diffraction is one of the most important characterization tools used in solid state chemistry and materials science. We can determine the size and the shape of the unit cell for any compound most easily using the diffraction of x-rays. The figure below shows the x-ray diffraction pattern from a single crystal of a layered clay. Strong intensities can be seen for a number of values of n; from each of these lines we can calculate the value of d, the interplanar spacing between the atoms in the crystal. Fig. 2 X-ray diffraction pattern from a layered structure vermiculite clay. Two reflections from different planes Lets imagine one piece of solid. That satisfies Bragg reflection as shown. We will observe strong diffraction But if the planes are misaligned We will observe no diffraction In any real solid we have a chance orientation. It would be almost impossible to study diffraction. However, 99% of all materials are polycrystalline or can be prepared (by grinding) so as to present many grains of material. In these some will always be at the correct alignment. Those planes at the right orientation will give strong diffraction A single crystal, also called monocrystal, is a crystalline solid in which the crystal lattice of the entire sample is continuous and unbroken to the edges of the sample, with no grain boundaries. Because grain boundaries can have significant effects on the physical and electrical properties of a material, single crystals are of interest to industry, and have important industrial applications. The most notable of these is the use of single crystal silicon in the fabrication of semiconductors. Monocrystals are often made by Czochralski process, controlled crystallization from the melted material. The opposite of a single crystal sample is a polycrystalline sample, which is made up of a number of smaller crystals known as crystallites. Usually those crystallites are connected through a amorphous material to form extended solid. A galvanized surface (a steel "strong-tie" joiner) showing visible spangle (of approx. 5mm) X-ray diffraction provides a 2D image of the atomic arrangement. Since nλ = 2d sin θ or sin θ = nλ/2d The image is a recipricol one. In a polycrystalline or powder sample though we do not see one specific arrangement or rotation of the small crystallites. If we scan across the image diagonally gives peaks EXPERIMENTAL SET-UP Modern machines rotate the x-ray source and the detector. detector source sample X-ray sources are cumbersome. To generate sufficient signal need to generate lots of power i.e x-rays. Frequently run at 50 kV and 50 mA. anode e’s 5V -ve 50 kV +ve filament Moving these are difficult because of the electric supplies and water used to cool anode to prevent melting. Older machines moved detector through 2Θ and sample through Θ. This maintains angle incidence = angle of reflection. For historical reason we plot intensity versus angle 2Θ X-RAY LINE BROADENING When x-rays enter a solid they undergo refraction. For x-rays this is very small. But the refraction angle differs from the incidence angle by only parts per thousand. But because of this the path length difference slowly varies from planes deeper into the material. The constructive interference slowly becomes destructive. Provided the sample is thick enough (if a sample is 10 μm there are 1 x 10-5/10-10 atom planes = 105) then all these slightly out of phase reflections will cancel. Leave just the perfect constructive interference feature. Shown by rocking curves. Scan detector across the diffracted beam. 2500 intensity 2000 1500 1000 500 0 44.8 45 45.2 45.4 45.6 45.8 46 46.2 angle 2-theta Called rocking curve – it is a measure of how crystalline a material is. A Si wafer rocking curve could have a rocking curve of 0.1 mrad. If the samples are thin incomplete cancelling is observed. The diffracted peaks are not narrow they become broad. The broadness can be used to estimate the sample thickness. Formulism was drawn up by Scherrer:Sample thickness, t = 0.9 λ/(B cos θ) B = Bactual = √(B2obs – B2o) where Bobs = FWHM of reflection Bo = instrumental FWHM minimum Small-angle X-Ray scattering. We have considered that Bragg's Law, d = /(2 sin), supports a minimum size of measurement of /2 in a diffraction experiment …but does not predict a maximum size. We have discussed the use of diffraction to measure crystalline and amorphous structures on the atomic scale, but clearly, many morphologies are of importance that have characteristic sizes much larger than the atomic scale – nano-sized objects. Guinier was one of the fathers of an outgrowth of diffraction aimed at large-scale structures in the 1950's. Bragg's Law predicts that information pertaining to such nano- to colloidal-scale structures would be seen below 6° 2 in the diffractometer trace. It is possible to design specialized instruments to measure down to less than 1/1000 of a degree for measurement of up to 1-micron scale structures using x-rays! In XRD the atomic scattering factor, f2 = ne2(1/q), where q is 4π sin(θ)/λ. Additionally, the intensity of scattering is known to be proportional to the number of scattering elements in the irradiated volume, Np(1/q). Then, in small-angle scattering we can consider a generalized rule that describes the behavior of scattered intensity as a function of Bragg size "d" or "r" that is observed at a given scattering angle 2q, where r = 1/q. I(q) = Np(1/q) ne2(1/q) All scattering patterns in the small-angle regime reflect a decay of intensity in q and this can be easily described by considering that at decreasing size scales the number of electrons in a particle is proportional to the decreasing volume, while the number of such particles increases with 1/volume. Then the scattered intensity by equation (1) is proportional to the decay of the particle volume with size. This analysis implies that the definition of a particle, i.e. r, does not necessarily reflect a real domain, but reflects the size, r, of a scattering element that could be a component of a physical domain. The characteristics of materials at these larger size scales are fundamentally different than at atomic scales. Atomic scale structures are characterized by high degrees of order, i.e. crystals, and relatively simple and uniform building blocks, i.e. atoms. On the nano-scale, the building blocks of matter are rarely well organized and are composed of rather complex and non-uniform building blocks. The resulting features in x-ray scattering or diffraction are sharp diffraction peaks in the XRD range and comparatively nondescript diffuse patterns in the SAXS range. For example polyethylene sub-micron sized fibrils High Density Polyethylene showing XRD at high-q, SAXS at intermediate q and LS (light scattering) at lowq. One distinct exclusion: Ordered Mesoporous Materials (OMMs). Somehow show relatively sharp and intensive X-ray lines, similarly to crystalline solids. - No atomic arrangement, essentially amorphous but - Long-range order of mesopores with defined and controllable pore sizes in the range of 2 – 10 nm. - Various types of mesophase structures hexagonal, cubic, etc. - Hybrid organic/inorganic structures, prepared by selfassembly of surfactants and inorganic molecular precursors. 100 nm 100 nm 40 nm Powder XRD of mesoporous solids 6000 (100) 5000 Intensity (cps) 4000 3000 2000 1000 (110) (200) 0 1 2 3 4 5 2 Theta (degrees) d(010) d(110) c d(100) a b Show how to calculate the unit cell size from the d(100) 1500 Intensity (cps) 1000 500 0 2 3 4 5 6 7 2 Theta (degree) Reflections at 2.42, 2.82, 3.68, 3.94, 4.4 and 4.6 degrees 2, give the indexation, if you know that is body centred cubic and calculate the unit cell size. Reflection and grazing-incidence geometry for thin film X-ray analysis Refection geometry is similar to that of PXRD geometry; it is usually called (θ – θ) as shown on the scheme. The X-ray source and the detector are moved in symmetrical mode thus scanning varying angels of θ. In this geometry only planes parallel to the substrate surface are detectable. X-ray tube scanning direction detector scanning direction incident beam diffracted beam d transmitted beam Substrate Example: hexagonally ordred silica mesoporous thin films (MTFs) on silicon wafer. Case a) synthetically not achievable. Cases b) and c) show mono-oriented cases where the extinction of certain reflections suggests preferential arrangement of mesoporous channels. Compare to powders where different possible orientations are possible and the PXRD patterns in (θ – θ) show all possible reflections. Problem with this geometry is that the reduced number of reflections most often hinders the unique identification of the structure, since larger numbers of reflections are required to index the structure. Grazing-incidence diffraction (GID) is a scattering geometry combining the Bragg condition with the conditions for X-ray total external reflection from surfaces when the incident angle of the X-rays is small enough (typically 0.05 – 3°, depending on the substrate electron density and the X-ray energy), close to the so called critical angle c. In the case of Cu Kα radiation, the critical angle is o 0.22 for silicon, 0.420 for nickel, and 0.570 for gold. At this point the surface is not entirely invisible to the X-rays, but only an evanescent wave penetrates and scatters from it. These conditions provide superior characteristics of GID as compared to other diffraction schemes in the studies of thin surface layers. In order to perform grazing-incidence diffraction, a highly intense, parallel, and stable X-ray beam is desirable that can be obtained only by using Synchrotron radiation. When coupled with 2D detector (CCD) camera full structural information can be obtained. Usually the beam is directed at small angels (grazing) angels close to the c of the material of interest where both reflected out-ofplane and in-plane reflections can be generated and recorded by the 2D detector as shown on the scheme. qz out of plane diffraction qx specularly reflected beam CCD 2o αi qy αf 2o incident beam mesoporous film beam stop substrate primary beam sample horizon in plane diffraction Example: Determination of the structure of the MTFs by grazing incidence SAXS (GI-SAXS). 100 3.73 nm 100 010 4.68 nm The real symmetry is not hexagonal but centered rectangular because of the contraction normal to the substrate. Contraction dh(010) dr(110) dr(110) ch cr cr ar dr(200) dh(100) ar ah bh br br Unique identification of the cubic MTFs is possible only with this set up. Much of the information is not detectable in reflection geometry. 3.6 nm 4.3 nm 211 121 21-1 210 200 120 12-1 021 2-1-1 02-1 020 -120 -121 dr(200)