Lab: Determination of the - Greenwich Public Schools
advertisement

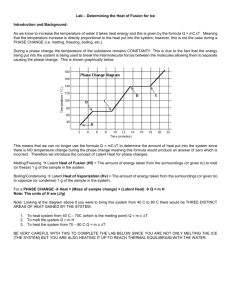
Lab: Determination of the Heat of Fusion of Ice Name________________________________ Date________ Block___ In this lab, you will determine experimentally the heat of fusion of ice, Hf. This is the amount of heat energy required to melt 1.00 g ice at its melting point of 0.00 oC. You have carried out the general procedure in a previous lab in which you cooled water at room temperature until the temperature no longer decreased. The cooling was done by adding ice cubes and keeping ice present in the water at all times. This lab will require more concentration on: . following the directions exactly as they written – no short cuts or leaving out a step . measuring and recording volume, mass, and temperature as accurately as possible using sig. figs. and units . calculations using the appropriate formula and showing ALL work, cancelled units, etc. The following relationships will be used in this lab: a) heat lost (or gained) by water = original mass of water X change in temp. of water specific heat of water X The equation to be used is: Q = m x T x c where Q = amount of heat in joules lost or gained by the substance m = mass of the substance in grams T = change in temperature in (oC); T = (T2 T1) c = specific heat of the substance; specific heat of liquid water is 4.18 J (g)(oC) b) law of conservation of energy: amount of heat lost by one mass = amount of heat gained by another mass c) using the formula Q = Hf x m and rearranging to solve for Hf : Hf = __Q_ m heat of fusion of ice = Materials/Equipment 2 styrofoam cups ice cubes tap water access to a balance amount of heat absorbed to melt the ice mass of the melted ice thermometer 250-mL beaker hot water source aprons/goggles crucible tongs paper towels 100-mL graduated cylinder beaker tongs 1 Procedure 1. Determine the mass of the remaining clean, dry styrofoam cup. Record the mass of the Styrofoam cup in the Data Table provided. (Be sure to include all sig. figs.!) 2. Add enough tap water to the styrofoam cup to about 1/2 full. 3. Determine the mass of the styrofoam cup and the water. Subtract the mass of the cup from the mass of the cup and water. Record the mass of the water alone in the Data Table as M1. Remove the styrofoam cup with water from the balance. 4. Immediately measure the temperature of the water in the styrofoam cup as accurately as possible (sig. figs.!!!) and record this initial temperature as T1 in the DataTable. 5. IMMEDIATELY add 2-3 ice cubes to the water in the Styrofoam cup and begin stirring gently with the thermometer. Stir continuously! As the last of the ice appears about to melt, add another ice cube. The cup should contain ice at all times. 6. Continue stirring and adding ice as needed until the temperature of the water/ice mixture no longer decreases. Record this final temperature as T in the Data Table. 2 7. Using the spoon, carefully remove the unmelted ice – allow as much water as possible to drip from the unmelted ice into the styrofoam cup. Place the unmelted ice into the sink. 8. Mass the water that is now in the styrofoam cup. Subtract the mass of the empty dry cup from the mass of the cup and water. Record this final mass of the water alone as M in the Data Table. 2 9. Repeat Steps 1 – 8 for two more trials. You should now have a total of 3 trials. 10. When doing the calculations, use the average values from the Data Table. Pay strict attention to units and to significant figures!!!!!!!!!! 11. Write up the lab in your lab notebook following the lab write-up model given to you. NOTE: there is no hypothesis, no dependent or independent variables. I have reduced the print size of the Materials and Procedure to make it easier to fit in your notebook. 2 DATA TABLE Original Mass of Mass of Final clean, dry Water Mass styrofoam (g) of cup (g) (M1) Water Trial # (g) (M2) Mass of Melted Ice (g) Initial Water Temp (oC) (T1) (M2 M1) Final Change Water in Temp Temp (oC) (oC) (T2) (T) 1 2 3 Avg. Values Calculations. SHOW ALL WORK!!!!!! Use the AVERAGE VALUES in the calculations. Pay attention to units and sig. figs.!!!!! 1. Calculate the heat lost by the original mass of water. 2. Calculate the heat of fusion of ice. 3. Calculate the amount of heat gained by the ice during melting. Hf of ice = 334.5 J/g. 4. Calculate your percent error. The accepted value for the heat of fusion of ice is 334.5 J/g. Conclusions and Analysis. Write the questions and the answers in your lab notebook. Use complete sentences! Use paragraphs when necessary! You may word process this section. NOTE: “b/c” is NOT an abbreviation for “because.” 1. Why were the MASS of the water and the MASS of the melted ice used instead of the volumes of each? 3 2. List at least 3 possible sources of error in this lab. Remember, sources of error are things over which you have NO control. 3. Explain how two (2) of the sources of error listed above may have affected your results. 4. Using evidence from this lab, explain how calorimetry is a demonstration of the law of conservation of energy. 5. a) Is the process of melting endothermic or exothermic? b) State evidence from this lab to support your answer. 4