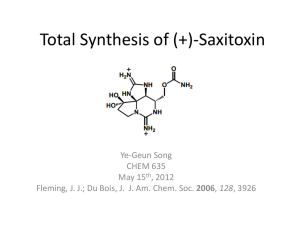
Ionic Liquid Mediated One Pot Synthesis of Substituted 2,4,6
advertisement

International Journal of ChemTech Research CODEN (USA): IJCRGG ISSN: 0974-4290 Vol.2, No.1, pp 289-294, Jan-Mar 2010 Ionic Liquid Mediated One Pot Synthesis of Substituted 2,4,6-Triarylpyridines Pravin G. Ingole, Sumit V. Jadhav and Hari C. Bajaj* Discipline of Inorganic Material and Catalysis, Central Salt and Marine Chemicals Research Institute, Council of Scientific and Industrial Research (CSIR) G.B.Marg, Bhavnagar-364002, Gujarat-India *Corres. Author: ingolepravin@gmail.com Tel: +91-278-2567760,Fax: +91-278-2567562 Abstract: A novel system for the synthesis of series of 2,4,6-triarylpyridines and their hydroxyl substituted analogues using cheap and easy to synthesize ionic liquid, Ethyl ammonium nitrate (EAN) as a benign reaction medium is demonstrated. The reaction of acetophenone or p-hydroxyacetophenone with aryl aldehydes in EAN, in the presence of base, followed by treatment with ammonium acetate affords title compounds in significant yields using a 'one pot' procedure. A sequential Aldol condensation and Michael addition reactions in ionic liquid, yields 1,5-diones, which can be readily converted to expected triarylpyridines, via ring closure with ammonium acetate. The significance of our findings relates to reducing the use of volatile organic solvents, simplicity of the process, as well as its good yields, mild conditions and low costs. Keywords: Triarylpyridine; Ionic liquid; Ethyl ammonium nitrate; Green chemistry. Introduction The N-heterocyclic compounds comprising Krohnke type pyridines and other substituted pyridines such as 2,4,6-triarylpyridines are prominent synthons in supramolecular chemistry, as a consequence of their πstacking ability along with directional H-bonding capacity. Moreover this class of compound is identified as very useful intermediate in the development of molecules for pharmaceutical or biological interest. Rich coordination properties of these compounds have been greatly exploited for the development of nanoscale metallosupramolecular architectures [1]. Pyridines exhibit range of biological activities as; antimalarial, anticonvulsant, anesthetic, antioxidant, antibacterial and antiparasitic properties [2,3]. In recent years these compounds are deeply investigated as potential candidates for calcium channel blockers and as antiviral agents [4]. Moreover, pyridine derivatives have remarkable versatility in synthetic organic chemistry as intermediates in the preparation of natural products and as ligands to be used in asymmetric synthesis [5]. Some analogues of pyridines are also highlighted as useful intermediates in the synthesis of pesticides, desiccants, surfactants and so forth [6,7]. For the diverse range of potential applications, easy access to differently functionalized Krohnke type triarylpyridines on a large scale and high yields is a prerequisite. Numbers of methods have been reported for the synthesis of these compounds either involving the condensation of 1, 5-diketones with formamideformic acid [8] or by other synthetic procedures such as Chichibabin method [9,10]. Successful synthesis of Krohnke type pyridines is also reported through the reaction of N-phenacylpyridinium salts with α, βunsaturated ketones in the presence of ammonium acetate [11] however, the pyridinium salts and the unsaturated ketones have to be synthesized first, turning this method relatively expensive. More recently many improved processes for synthesis of Hari C. Bajaj et al /Int.J. ChemTech Res.2010,2(1) Krohnke type pyridines have been established, involving variety of precursors such as, α-ketoketene dithioacetals [12], N-phosphinylethanimines [13] and lithiated β-enaminophosphonates [14]. However, most of the established methodologies suffer from several disadvantages, as multi-step procedures, long reaction time, use of toxic reagents and volatile organic compounds (VOC’s). As a consequence, for the stringent and growing environmental regulations, organic chemists are requested to develop environmentally benign synthetic methods with minimization of waste, a foremost requirement in principles of green chemistry [15]. Among the environment friendly approaches to synthesize triarylpyridines; microwave assisted method using polyethylene glycol (PEG) is been demonstrated by Huang et al [16]. In a typical attempt to avoid solvent media Heravia et al [17] have synthesized target compounds in reasonable yields by using heteropolyacids as a catalyst, whereas Wu et al [18] have synthesized substituted triarylpyridines via catalyst free Diels–alders reaction. To the date PEG mediated synthesis of triarylpyridines via 1, 5-diketone intermediate reported by Raston et al [19] is one of the most prominent approach towards synthesis of triarylpyridines, both in the context of green chemistry, 290 generality and simplicity of the procedure. In keeping up with theme of green chemistry, we examined the potential for using novel green reaction media for the synthesis of triarylpyridines. One of the promising approaches is to employ Ionic liquid (IL) media. However, to the best of our knowledge, synthesis of triarylpyridines in IL media has rarely been reported. Herein, we have reported facile and clean synthesis of series of substituted 2,4,6 triarylpyridines starting from appropriate aromatic aldehydes and acetophenones employing IL’s as reaction media. Ethyl ammonium nitrate (EAN), a cheap and easy to synthesize IL was chosen as solvent system and successfully employed to synthesize target compounds under conventional heating conditions. Experimental Materials Acetophenone, p-hydroxyacetophenone and corresponding aryl aldehydes were procured from Sigma-Aldrich. Ethyl amine, nitric acid, ammonium acetate, sodium hydroxide and common organic solvents were obtained from S.D.fine chemicals Ltd. India. All the chemicals were used as received, without further purification. Scheme: Synthesis of triarylpyridines using IL solvent media Hari C. Bajaj et al /Int.J. ChemTech Res.2010,2(1) General 1 H and 13C NMR spectra were recorded on a Bruker Avance-II spectrometer operating at 500 MHz and 125 MHz respectively, using DMSO-d6 as solvent and TMS as an internal reference. GC-MS analyses were performed on Shimadzu QP-2010 spectrometer. Elemental analyses were determined by using a Perkin-Elmer 2400 CHNS/O analyzer instrument. Melting points were recorded with Metler-Toledo melting point apparatus and are uncorrected. Synthesis of EAN EAN is synthesized via simple process reported elsewhere [20]. Nitric acid (30%) was added dropwise to an aqueous solution of ethyl amine, (70%, 50 ml) under vigorous stirring maintaining the temperature below 10°C, till the pH of the mixture reaches nearby 7.3, then addition was stopped and mixture further stirred for 1 h. Most of the water from the mixture was removed using rotary evaporator in a boiling water bath at a pressure of 200 mm of Hg and final traces of water were removed at 100°C in presence of high vacuum of 1 mm Hg. The IL is obtained as viscous colourless liquid and used as such for further applications. General Procedure for the Synthesis of 2,4,6Triarylpyridines In a typical reaction, Acetophenone (2 mmol, 240 mg) was added to a suspension of crushed NaOH (2 mmol, 80 mg) in EAN (3 cm3) at 80°C and stirred for few minutes. Benzaldehyde (1mmol, 106 mg) was then added along with ammonium acetate. (1.5 g, excess) The reaction mixture was heated for 4 h at 110°C. (Scheme 1) The progress of the reaction was monitored by TLC. Upon completion of the reaction, cold water was added to facilitate the precipitation of product. If necessary some ethanol was added to reaction mixture and further stirred for 30 minutes, then solvent removed in vacuo and crude product was subjected to column chromatography on silica gel. Final product was collected in purified form with recrystallization in hot acetone. (Yield 405 mg, 66%). Characterized by 1H and 13C NMR spectroscopy. Similar stoichiometric procedure and characterization techniques were employed for other entries. Results and Discussions Synthesis of 2,4,6-triarylpyridines bearing various functionalities, in simple one pot method is demonstrated. Treatment of acetophenone or phydroxy acetophenone (2 mmol) with appropriately substituted aromatic aldehydes (1 mmol) and ammonium acetate (excess) in IL as reaction medium afforded the triarylpyridines in single step reaction. Initiation is done with generation of carbanion at the acetophenone centre by treatment with strong base 291 such as sodium hydroxide, crushed within IL medium by heating the reaction mixture at 80ºC. The reaction is believed to proceed through previously reported 1, 5 dione intermediate. (See also ref. 19) Final ring closure with insertion of nitrogen atom in the central pyridine core is done with excess of ammonium acetate added. This method to synthesize triarylpyridines is simple and easy to operate, also applying the principles of ‘green chemistry’, notably the use of IL’s the results of which are reported herein. IL’s are becoming prominent, benign, alternative reaction medium in synthetic chemistry as they have many fascinating properties, with few of them are; no measurable vapor pressure and hence can not emit VOC’s, nonflammability, high polarity, solubility for wide range of materials including inorganic, organic and even polymeric materials, with potential to be reused and recycled. To optimize the reaction temperature, a series of reactions were carried out at temperatures ranging from 100 to 150°C in an increment of 10°C each time. The cleaner reaction conditions and better yields are obtained at 110°C. Under these optimized reaction conditions, we have synthesized a series of compounds a-j in acceptable purity and yields. (Table 1). The crude product was subjected to column chromatography on silica gel. Final product was collected in purified form with recrystallization in hot acetone. IL is water-soluble, thus goes to the aqueous layer, which was distilled under vacuum using rotavapor to remove water, living behind the IL. The recovered EAN was recycled and reused several times to carry out the same experiment (Table 2). Conclusions A new, convenient route for the synthesis of substituted 2, 4, 6-triarylpyridines using IL has been demonstrated. The ‘one pot’ base catalyzed reaction is tolerant to various functionalities attached to the aldehyde core, leading to the formation of libraries of differently substituted triarylpyridine synthons for wide range of potential applications. EAN has been successfully employed as a benign reaction medium proving its synthetic versatility over volatile organic solvents. Good yields of products, accessible starting compounds and relatively easy experimental procedure feasible in single step is sufficient to attract green chemists to open the new avenue in synthesis of triarylpyridines in greener conditions. a. δH (500 MHz, DMSO) 8.37 (d, 4H, Ar), 8.33 (s, 2H, Pyr), 8.08 (m, 4H, Ar), 8.04 (d, 2H, Ar), 7.57 (m, 2H, Ar), 7.53 (m, 2H, Ar), 7.29 (m, 1H, Ar). δC (125 MHz, DMSO) 157.51, 150.59, 139.80, 138.73, 129.28, 128.35, 127.95, 126.84, 117.59. GCMS (EI): m/z (%) 307 (100) [M]+. Elemental analysis calcd.(%) for C23H17N: C, 89.87; H, 5.57; N, 4.56; found C, 89.62; H, 5.60; N, 4.43. Hari C. Bajaj et al /Int.J. ChemTech Res.2010,2(1) 292 b. δH (500 MHz, DMSO) 8.25 (d, 4H, Ar), 8.24 (s, 2H, Pyr), 8.08 (d, 2H, Ar), 8.01 (s, 1H, Ar), 7.99 (m, 4H, Ar), 7.56 (m, 2H, Ar), 5.77 (s, 1H, OH), δC (125 MHz, DMSO ) 156.56, 153.78, 152.61, 143.40, 142.74, 133.56, 133.14, 130.45, 128.95, 125.13, 125.11, 123.55, 118.61. GCMS (EI): m/z (%) 358 (100) [M]+. Elemental analysis calcd.(%) for C23H16ClNO: C, 77.20; H, 4.51; N, 3.91; found C, 77.10; H, 4.75; N, 3.93. c. δH (500 MHz, DMSO) 8.27 (d, 4H, Ar), 8.05 (s, 2H, Pyr), 7.98 (s, 2H, Ar), 7.78 (s, 1H, Ar), 7.66 (m, 4H, Ar), 7.56 (m, 2H, Ar). δC (125 MHz, DMSO) 158.35, 155.86, 147.05, 138.66, 133.75, 133.36, 129.40, 129.35, 129.22, 127.89, 126.79, 119.21. GCMS (EI): m/z (%) 392 (100) [M]+. Elemental analysis calcd.(%) for C23H15Cl2NO: C, 70.42; H, 3.85; N, 3.57; found C, 70.25; H, 3.76; N, 3.69. d. δH (500 MHz, DMSO) 8.66 (d, 4H, Ar), 8.00 (s, 2H, Pyr), 7.96 (m, 4H, Ar), 7.85 (d, 1H, Ar), 7.80 (m, 2H, Ar), 7.79 (d, 1H, Ar), 7.67 (m, 1H, Ar), 6.31 (s, 1H, OH). δC (125 MHz, DMSO) 153.78, 153.70, 133.65, 133.16, 130.43, 129.06, 128.99, 128.69, 128.65, 127.89, 124.00, 117.57, 114.91. GCMS (EI): m/z (%) 368 (100) [M]+. Elemental analysis calcd.(%) for C23H16N2O3: C, 74.99; H, 4.38; N, 7.60; found C, 74.95; H, 4.30; N, 7.56. e. δH (500 MHz, DMSO) 8.24 (s, 2H, Pyr), 8.14 (m, 4H, Ar), 7.97 (m, 2H, Ar), 7.56 (m, 2H, Ar), 7.37 (m, 1H, Ar), 7.11 (m, 4H, Ar), 5.65 (s, 2H, OH ). δC (125 MHz, DMSO) 156.50, 149.57, 138.70, 133.09, 129.27, 128.71, 127.34, 125.82, 122.01, 116.58. GCMS (EI): m/z (%) 339 (100) [M]+. Elemental analysis calcd.(%) for C23H17NO2: C, 81.40; H, 5.05; N, 4.13; found C, 81.52; H, 5.11; N, 4.06. f. δH (500 MHz, DMSO) 8.30 (s, 2H, Pyr), 8.13 (m, 4H, Ar), 7.85 (m, 1H, Ar), 7.51 (m, 1H, Ar), 7.25 (m, 1H, Ar), 6.93 (m, 1H, Ar), 6.76 (d, 4H, Ar), 5.70 (s, 3H, OH). δC (125 MHz, DMSO ) 162.34, 154. 41, 136.27, 132.61, 131.39, 128.69, 119.20, 119.14, 118.63, 118.26, 117.82, 117.54, 116.02. GCMS (EI): m/z (%) 355 (100) [M]+. Elemental analysis calcd.(%) for C23H17NO3: C, 77.73; H, 4.82; N, 3.94; found C, 77.67; H, 4.78; N, 3.88. Table 1: Structure yields and melting point of the synthesized triarylpyridines. MP (°C) Found Reported Time(h) Yield(%)a a 4 66 135 (137-138)10 b 5 52 112 -- c 4 54 165 -- 6 69 122 -- e 5 59 151 -- f 5 76 109 -- g 6.5 80 116 (118-119)10 h 6 75 200 (202-203)13 i 5 82 199 (197)17 j 4.5 87 126 (129-130)13 Entry Ar1 Ar2 d Ar1: Aldehydes; Ar2: Acetophenone/p-hydroxyl acetophenone; aYields refer to isolated products. Hari C. Bajaj et al /Int.J. ChemTech Res.2010,2(1) 293 Table 2: Recovery of ethylammonium nitrate [EtNH3] NO3 in the synthesis of 2, 4, 6triarylpyridines. Yield (%) of [EtNH3]NO3 Entry Recycle3 Recycle1 Recycle2 g 80 72 67 h 75 68 62 i 82 78 71 j 87 83 80 Acknowledgements We gratefully acknowledge Council of Scientific and Industrial Research (CSIR), New Delhi-India. We also thank Analytical sciences discipline of the institute for analysis facilities. References 1. Thompson C., The synthesis of 2,2′:6′,2″terpyridine ligands -- versatile building blocks for supramolecular chemistry, Coord. Chem. Rev., 1997, 160, 1-52. 2. Kim B.Y., Ahn J.B., Lee H.W., Kang S.K., Lee J.H., Shin J.S., Ahn S.K., Hong C.I. and Yoon S.S., Synthesis and biological activity of novel substituted pyridines and purines containing 2,4-thiazolidinedione, Eur. J. Med. Chem., 2004, 3, 433-447. 3. Adib M., Tahermansouri H., Koloogani S.A., Mohammadi B. and Bijanzadeh H.R., Krohnke pyridines: an efficient solvent-free synthesis of 2,4,6-triarylpyridines, Tetrahedron Lett., 2006, 47, 5957-5960. 4. Kappe C.O., Biologically active dihydro pyrimidones of the Biginelli-type - a literature survey, Eur. J. Med. Chem., 2000, 35, 10431052. 5. Chen C., Tagami K. and Kishi Y., Ni(II)/Cr(II)-mediated coupling reaction: an asymmetric process, J. Org. Chem., 1995, 60, 5386-5387. 6. Heathcock C.H., Norman M.H. and Uchling D.E., Acyclic stereoselection. 29. Stereoselection in the Michael addition reaction. 1. The Mukaiyama-Michael reaction, J. Am. Chem. Soc., 1985, 107, 2797-2799. 7. Mukaiyama T., Kobayashi S. and Murakami M., An efficient method for the preparation of threo cross-aldols from silyl enol ethers and aldehydes using trityl perchlorate as a catalyst, Chem. Lett., 1985, 14, 447-450. 8. Chubb F., Hay A.S. and Sandin R.B., The Leuckart Reaction of Some 1,5-Diketones, J. Am. Chem. Soc., 1953, 75, 6042-6044. 9. Frank R.L. and Seven R.P., Pyridines. IV. A Study of the Chichibabin Synthesis, J. Am. Chem. Soc., 1949, 71, 2629-2635. 10. Lombard R. and Stephen J.P., Bull. Soc. Chim. Fr., 1958, 1458. 11. Krohnke F. and Zecher W., Syntheses Using the Michael Addition of Phridinium Salts, Angew. Chem. Int. Ed., 1962, 1, 626-632. 12. Potts K.T., Cipullo M.J., Ralli P. and Theodoridis G., Ketene dithio acetals as synthetic intermediates. Synthesis of unsaturated 1,5- diketones, J. Am. Chem. Soc., 1981, 103, 3584-3585. 13. Kobayashi T., Kakiuchi H. and Kato H., On the Reaction of N-(Diphenylphosphinyl)-1phenylethanimine with Aromatic Aldehydes Giving 4-Aryl-2,6-diphenylpyridine Derivatives, Bull. Chem. Soc. Jpn., 1991, 64, 392-395. 14. Palacios F., Retana A.M.O. and Oyarzabal J., A “one pot” synthesis of polysubstituted pyridines from metallated alkylphosphonates, nitriles and α,β-unsaturated ketones, Tetrahedron Lett., 1996, 37, 4577-4580. 15. Anastas P.T. and Warner J.C., Green Chemistry, Theory and Practice. Oxford University Press, 1998. 16. Huang X.Q., Li H.X., Wang J.X. and Jia X.F., A Rapid and Efficient Synthesis of 2, 4, 6Triarylpyridines under Microwave Irradiation, Chinese Chemical Lett., 2005, 16, 607-608. 17. Heravia M.M., Bakhtiaria K., Darooghehaa Z. and Bamoharram F.F., An efficient synthesis of 2,4,6-triarylpyridines catalyzed by heteropolyacid under solvent-free conditions, Catalysis Comm., 2007, 8, 1991-1994. Hari C. Bajaj et al /Int.J. ChemTech Res.2010,2(1) 294 18. Wu H., Lu L., Shen Y., Wan Y. and Yu K., Sequential Aldol/Michael Addition Reaction in Ionic Liquid Catalyzed by Morpholine: A Convenient Synthesis of 1,3,5-Triaryl-1,5pentanedione, Synthetic Comm., 2006, 36, 1193-1200. 19. Smith N.M., Raston C.L., Smith C.B. and Sobolev A.N., PEG mediated synthesis of ***** amino-functionalised 2,4,6-triarylpyridines, Green Chem., 2007, 9, 1185-1190. 20. Rajagopal R. and Srinivasan K.V., Ultrasound promoted para-selective nitration of phenols in ionic liquid, Ultrasonics sonochemistry, 2003, 10, 41-43.