View
advertisement

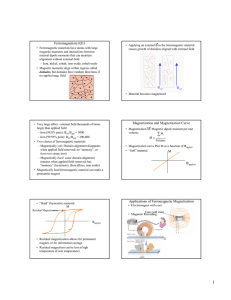
Synthesis and Magnetic properties of SmFeMnO5 N. Aparnadevi, K. Saravana Kumar, A. Muthukumaran, and C. Venkateswaran* Department of Nuclear Physics, University of Madras, Guindy Campus, Chennai 600025. India. *Corresponding author’s e-mail: cvunom@hotmail.com Abstract: SmFeMnO5 is obtained by high-energy ball milling of stoichiometric oxide precursors, followed by sintering at 950 ̊ C for 12 h. The crystal structure is orthorhombic with Pbam space group as revealed by Rietveld analysis of X-ray diffraction data. Room temperature magnetization study show a ferromagnetic hysteresis loop with a saturation magnetization of 6.27 emu/g. Keywords: Mechanical Milling; Multiferroic; Frustrated Magnets; Rietveld; Ferromagnetic INTRODUCTION SmMn2O5 is a magnetoelectric material having frustrated geometry in their magnetic lattice. The mixed oxidation state of Mn ions (Mn3+, Mn4+) determine their magnetic behaviour through superexchange interaction, inducing unique ferroelectric phases [1]. The magnetic spin ordering in SmMn2O5 can be altered by the substitution of Fe3+ ions in Mn3+ sites.. The effect of Fe in SmMn2O5 is not yet reported. EXPERIMENTAL Polycrystalline powder of SmFeMnO5 is obtained from high energy ball milling of the precursor oxides, followed by sintering at high temperatures. polyhedra, in addition to the decrease in the effect of Jahn Teller Mn3+ ion resulting in alteration of bond angles and distances and changing magnetic properties. A narrow hysteresis loop, characteristic of ferromagnetic material is obtained from RT VSM measurement as shown in Fig. 2. The saturation magnetization (MS) is 6.57 emu/g at a maximum applied field of 15 kOe. The remanent magnetization (MR) and coercivity (HC) are 2.81 emu/g and 222 Oe, respectively with the ratio MR/MS of 0.43. The contribution from the secondary phase SmMnO3 is absent due to its paramagnetic nature whereas the ferrimagnetic Fe3O4 has a saturation magnetization of 92 emu/g [3]. Fig. 2. Magnetization curve of SmFeMnO5. Fig. 1.Rietveld refinement of SmFeMnO5 X-ray diffraction pattern. RESULTS AND DISCUSSION The unit cell parameters of SmFeMnO5 are found to be a=7.4358(1) Å, b=8.5856(2) Å, and c=5.6944 (1) Å with a volume 363 Å3, from Rietveld refinement of the X-ray diffraction data, as shown in Fig. 1. Refinement is carried out using FULLPROF, with the assumption that Fe3+ ions replace the Mn3+ sites in parent SmMn2O5. Secondary peaks related to SmMnO3 and Fe3O4 were also identified. Formation of SmMnO3 phase, may be related to the size of the Sm3+ cations [2]. In SmMnFeO5, substitution of Fe3+ at Mn3+ sites affect the size of the ACKNOWLEDGEMENTS KSK acknowledges DRDO, India for support through fellowship. NAD acknowledges Sampathu for his kind help. REFERENCES [1] S.W. Cheong, Multiferroics: a magnetic twist for ferroelectricity, Nature materials, 6, 1, 13-20 (2007). [2] J. A. Alonso et al., Moderate-pressure synthesis and neutron diffraction study of new metastable oxides, Z. Naturforsch, 61b, 1507-1514 (2006). [3] X. Yang et al., Superparamagnetic graphene oxideFe3O4 nanoparticles hybrid for controlled targeted drug carriers, J. Mat. Chem., 19, 2710-2714 (2009).