File - Doug Miller e
advertisement

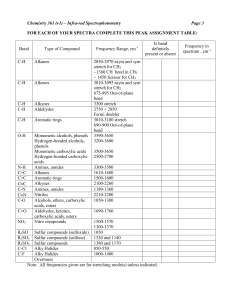
CHEM 201 Sec C3A Sp 2014 Name: Douglas Miller Date: 09-23-14 Date of Experiment: 09-17-14 Date of Submission: 09-24-14 Experiment #3: Infrared Spectroscopy identification of Unknowns Abstract: An unknown sample was identified via its boiling point, index of refraction and infrared spectra. Boiling point was determined utilizing standard laboratory techniques. While the index or refraction and IR spectra were determined via the use of an ABBE-3L Refractometer and a Perkin Elmer 1420 respectively. The identity of the unknown sample was determined to be cyclohexanone (C6H8O). Introduction: Infrared Spectroscopy makes use of the fact that the chemical bonds holding atoms together are in constant motion. This motion is caused by both the bending and stretching of the individual bonds within the molecule. By passing a beam of light, within the infrared (IR) spectrum, through a sample some of the energy will be absorbed and the loss of energy along with the corresponding frequency can be recorded. The frequency of the absorption corresponds to particular bonding patterns within the molecule thus allowing us to determine which types of bonds are present. This in conjunction with other information such as the molecular formula, boiling point and index of refractions can often allow us to make determinations on the chemical structure of an unknown samples. A major limitation of IR is its inability to measure bonds that do not adsorb energy within the IR region, one such example is the C=C bond within ethylene. In this experiment, an unknown sample, in this case #621, is to be identified via the use of it boiling point, index of refraction and IR spectra. Boiling point was determined by following the procedures as outlined in Ref (1) Experiment 1: Part B with a recorded result of 142-143OC. The index of refraction was determined via the use of an ABBE-3L Refractometer at 20oC with a record result of 1.4525. An IR spectra was produced on a Perkin Elmer 1420 Infrared Spectrophotometer with the neat sample held between Sodium Chloride (NaCl) plates (Figure 2). Experimental Procedure: This experiment followed the procedures outlined in Ford & Yau (Ref. 1). Results: Sample #621 was translucent with a slight yellow color and a tested boiling point of 142143OC. Index of refraction was tested on two separate refractometers with one reading 1.4525 and the other reading 1.425. These reading were confirmed by a second testing with a different individual arriving at the same results. After discussion with Dr. Evon G. Ford, it was determined that one of the instruments must be providing a false reading and or be out of calibration. The IR spectra obtained (see Figure 2 attached) contained the following significant peaks: Table 1. IR Spectra Absorption. Significant Absorption (cm-1) Likely Source 2960 – 2920 (strong, sharp) C-C Single Bonds C-H Stretch (Alkane) 2850 (strong, sharp) C-H Stretch Aldehyde Secondary Peak 2760 (weak, sharp) C-H Stretch Aldehyde Secondary Peak 1740-1700 (strong, sharp) C=O Stretch, Aldehyde Discussion: Based off the experimental data Cyclohexanone (C6H10O) and Cyclopentanone (C5H8O) where selected from the table on page 47 (Ref. 1) for comparison, both being aldehydes with characteristics similar to the unknown sample, Table 2. Table 2. Comparison of Unknown Sample with Similar Compounds. Reagents or Products Sample #621 Physical Properties Experimental Values Literature Values (Ref 2.) Boiling Point = 142-143oC Index of Refraction = 1.4525 / 1.425 Cyclohexanone C6H10O O Boiling Point = 154-156oC Index of Refraction = 1.4507 Cyclopentanone C5H8O O Boiling Point = 130-131oC Index of Refraction = 1.437 While the boiling point of the unknown sample tested as in between cyclohexanone (C6H10O) and cyclopentanone (C5H8O), it is more closely approximates that of cyclohexanone. Also while two different index of refraction were obtained for the unknown the one that is most similar to one of the above compounds is 1.4525 that corresponding to the 1.4507 for cyclohexanone. For these reason the recorded index of refraction of 1.425 on the unknown is being considered erroneous, as the refractometer it was recorded on is likely in need of calibration. IR Analysis: The IR spectrum (Figure 2) is consistent with that of Cyclohexanone. The strong, sharp peaks at 2960 – 2920cm-1 is due to the C-C single bond stretch and the C-H alkene stretch. The strong, sharp 1740-1700cm-1 peak is consistent with the C=O stretch of an aldehyde and is confirmed by the secondary aldehyde peaks at 2850cm-1 and 2760cm-1. The lack of any intense, broad peaks in the 3600-3200 cm-1 region indicate that the sample does not contain the O-H stretch of an alcohol. Additionally, the N-H stretch associated with this same region is not present nor is the C=N stretch at 2400-2200cm-1 indicating the absence of N. Also as the first primary peak is below 3000cm-1 both the C-H stretch of C=C-H and C=C are absent. An IR Spectra for Cyclohexanone (Figure 1) was obtained from (Ref. 3). It also shows a strong, sharp absorption in the 2960 – 2920cm-1 region along with the C=O stretch of aldehyde at 1717cm-1 and the secondary peaks at 2867cm-1 and 2750cm-1. Figure 1. IR Spectra of Cyclohexanone. Conclusion: Based on the experiment boiling point, index of refraction and a comparison of the IR spectra unknown sample #621 is cyclohexanone. References: (1) Ford, E. G.; Yau, C. L. CHEM 201 Experiments in Organic Chemistry I Laboratory 8th Ed; Academx: Bel Air, MD, 2014; pp 27-34. (2) http://www.chemspider.com (accessed 9-14-14) (3) http://sdbs.db.aist.go.jp/sdbs/cgi-bin/direct_frame_top.cgi (accessed 9-19-14) Answers to Post Lab Questions: 1. Based on the formula C3H8O, the IHD for the compound would be zero, indicating that there are only single bonds present. Based off that, there are three possible configurations for the compound, see below: OH H3C H3C H3C OH CH3 O CH3 In the case of the first two compounds, 1-propanol and 2-propanol (Isopropanol) there is the presence of an OH alcohol group. In the third case ethyl-methyl-ether, the OH alcohol group is not present. This is relevant in that according to the IR spectra there is a strong, broad band at 3340cm-1 and a strong band at 1080cm-1 both of which point to the presence of an alcohol. From here the boiling points need to be examined. In the first case 1-propanol has a listed BP of 97OC, while 2-propanol is listed at 82-83OC. Based off this the compound in question would H have to be 2-propanol, with a full chemical structure of: H C H H C O H H H C H