Supplementary Figure S1
advertisement

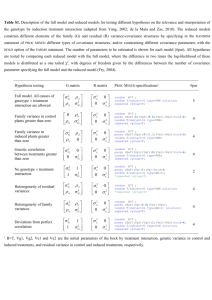
Supplementary material Supplementary Figure S1. Average (A) scotopic and (B) photopic ERG and VEP waveforms an hour after IOP was returned to baseline (10 mmHg). These were elicited by the same flash intensity as in Figures 1 and 3, respectively, of the manuscript. Both control (gray traces) and IOP-treated waveforms (black traces) taken at the initial baseline (thin traces) and at one hour after cessation of the IOP challenge (bold traces), were comparable. The exception being the photopic ERG (Fig. (B) left panel), the potential reasons for which are discussed in the main text. Page 1 of 4 Supplementary Figure S2. (A) Schematic ‘occlusion’ configurations to determine retinal contributions to the pigmented rat full-field flash VEP. Occlusion was achieved via non-invasive (i.e. dim flash sub-VEP threshold, -5.93 log cd.s.m-2) or invasive means (i.e. pharmacological blockade). Unfilled eyes; unoccluded, filled eyes; occluded, cross; VEP measurement location; thick solid and dashed afferents; retinostriate contributions from stimulated contralateral and ipsilateral eyes, respectively, thin afferents; those that are blocked, horizontal arrows; potential inter-hemispheric influences. Line thickness represent relative amount of contribution given the occlusion configuration. (B) Representative ERG and STR (inset) waveforms illustrating the effect of each drug used for uni- and bilateral retinal blockade. ERG effects are in agreement with literature.1-4 Briefly, pre- and post-cannulated test flashes (0.24 log cd.s.m-2) confirmed that the ERG remained normal within test/retest variability. BaCl2 (inset, thick trace) inhibits the inner retinal STR (-4.99 log cd.s.m-2, vs. untreated, inset thin trace). PDA and APB block OFF- and ON-bipolar cell output (1.52 log cd.s.m -2), respectively. Only the photoreceptoral negativity Page 2 of 4 remained when ‘All’ agents are injected (thick trace). Thus there should be no retinal output. (C) Average monocular VEP waveforms recorded following unilateral retinal block with BaCl2 (n = 7). The contralateral VEP (cortical response from the control eye, thin black trace) was markedly larger (~ 2 to 2.5 times) than the ipsilateral VEP (from BaCl2-treated eye, thick black trace), as have been previously reported.5-7 Also included is the putative binocular VEP (gray trace) calculated post hoc. (D) Comparison of non-visual cortical (thick traces) and ipsilateral retinal contributions (thin traces) to the VEP. Average signals generated after bilateral injection of a drug combination that blocks retinal output from both eyes (bilateral APB/PDA/BaCl2, n = 6), as well as extrapolated from cortical recording to a sub-VEP threshold flash (-5.93 log cd.s.m-2, n = 7). Cortical responses from both methods were significantly smaller than the ipsilateral retinal VEP. (E) Summary of VEP amplitudes (mean ± SEM, P1N1 and N1P2, see legend) from the various isolation methods described. Average values are provided in Table S1. (F) Comparison of relative contribution of remnant ipsilateral eye vs. non-visual cortical waveforms (see legend). P1N1 and N1P2 amplitudes are shown as a percentage of their respective controls for each group (mean ± SEM). Ipsilateral waveform amplitudes (from unilatateral BaCl2) were significantly larger than those of non-visual cortical noise (binocular dimmest flash and bilateral drugs). Arrows; putative visual P1N1 and N1P2 proportions from the ipsilateral eye. Supplementary Table S1 - Summary of the VEP parameter amplitudes and implicit times (mean ± SEM) recorded from the approaches described in Figure S2 to examine relative monocular (contralateral, ipsilateral) and non-ocular contributions to the binocular VEP. Scotopic 1.52 log cd.s.m-2, unless stated otherwise. Amplitude (µV) P1N1 N1P2 P1 N1 P2 280 13 439 15 23 2 34 1 73 4 Contra-eye VEP (contra-BaCl2 control) 273 11 414 12 21 2 35 1 70 3 Ipsi-eye VEP (contra-BaCl2) 112 23 171 28 18 2 45 5 77 7 29 6 49 12 23 3 44 6 76 7 Non-visual signal (bilateral drugs) 40 8 56 11 25 5 49 6 76 7 Contra-eye VEP, photopic (contra-BaCl2 C) 143 15 135 16 21 2 47 2 65 3 Ipsi-eye VEP, photopic (contra-BaCl2) 44 10 61 9 29 4 46 2 63 3 Binocular VEP (putative‡) Non-visual signal ‡ Implicit time (ms) (dim†) From post hoc addition of monocular contralateral and ipsilateral waveforms. † VEP contralateral to the untreated control eye under scotopic conditions elicited with a -5.93 log cd.s.m-2 flash. Supplementary Table S2 – Summary of VEP amplitude comparisons between visual and nonvisual signals described in Figure S2 and Table S1. Signals measured under scotopic conditions. Binocular VEP‡ ContraVEP Contra-eye VEP (contra-BaCl2 control) ns - Ipsi-eye VEP (contra- BaCl2) * * - Non-visual (bilateral drugs) * * * - Non-visual (dim†) * * * ns Page 3 of 4 IpsiVEP Non-visual (bilateral drugs) Non-visual (dim†) - ‡ From † VEP post hoc addition of monocular contralateral and ipsilateral waveforms. contralateral to the untreated control eye under scotopic conditions elicited with a -5.93 log cd.s.m-2 flash. * indicates p < 0.05; paired, two-tailed T-test. References 1. 2. 3. 4. 5. 6. 7. Stockton RA, Slaughter MM. B-wave of the electroretinogram. A reflection of ON bipolar cell activity. J Gen Physiol 1989;93:101-122. Green DG, Kapousta-Bruneau NV. A dissection of the electroretinogram from the isolated rat retina with microelectrodes and drugs. Vis Neurosci 1999;16:727-741. Wong VHY, Vingrys AJ, Bui BV. Glial and neuronal dysfunction in Streptozotocin-induced diabetic rats. J Ocul Biol Dis Inform 2012;Accepted 2nd December 2011 Raz-Prag D, Grimes WN, Fariss RN, et al. Probing potassium channel function in vivo by intracellular delivery of antibodies in a rat model of retinal neurodegeneration. Proc Natl Acad Sci U S A 2010;107:12710-12715. Creel DJ, Dustman RE, Beck EC. Differences in visually evoked responses in albino versus hooded rats. Exp Neurol 1970;29:298-309. Creel DJ, Dustman RE, Beck EC. Visually evoked responses in the rat, guinea pig, cat, monkey, and man. Exp Neurol 1973;40:351-366. Jehle T, Ehlken D, Wingert K, Feuerstein TJ, Bach M, Lagreze WA. Influence of narcotics on luminance and frequency modulated visual evoked potentials in rats. Doc Ophthalmol 2009;118:217-224. Page 4 of 4