tpj12151-sup-0012-Suppinfo
advertisement

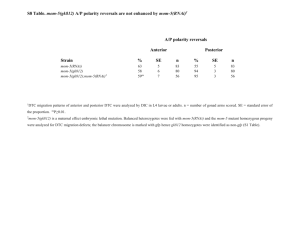
1 Supplemental data 2 3 4 Description 5 6 Generation of Bntt16 RNAi plants 7 In order to study the regulatory function of BnTT16 proteins in endothelial development, RNAi 8 Brassica napus plants were generated previously using a hairpin (HP) RNAi construct (Deng et 9 al. 2012; Figure S4, S5). Since all four BnTT16 homologs exhibit a high level of similarity and 10 the corresponding proteins displayed a high degree of functional overlap in their ability to 11 recover the phenotype of an Attt16 mutant (Figure 5, S3), silencing of one particular homolog in 12 B. napus may not have yield a detectable phenotype. For this reason, the RNAi construct was 13 designed to silence all four BnTT16 homologs simultaneously (Figure S4a)(Deng et al. 2012). 14 Briefly, a 146-bp BnTT16 cDNA fragment encoding a portion of the variable K domain was 15 amplified from a B. napus cDNA library and cloned into the pENTR/D-TOPO vector 16 (Invitrogen, Burlington, ON). The pENTR/D-TOPO vector provides flanking attB1 and attB2 17 sequences enabling directional cloning into the pHellsgate 12 vector. The Gateway LR 18 recombination reaction was performed according to manufacturer’s instructions (Invitrogen) and 19 the resulting vectors were verified by restriction analysis. Positive clones contained two identical 20 BnTT16 fragments in opposite orientations, separated by a combination of Pdk and Cat introns 21 (Figure S4a). The pHellsgate-BnTT16-146 cassette was then electroporated into Agrobacterium 22 tumefaciens GV3101 (pMP90) cells and used in the genetic transformation of B. napus line 23 DH12075 as described previously (Bondaruk et al. 2007). 1 24 The RNAi method (HP) used in this study has been proven to be a reliable technique to 25 decrease the level of target protein in agricultural plants (Liu et al. 2002, Stoutjesdijk et al. 2002, 26 Mietkiewska et al. 2008, Chen et al. 2011). In Arabidopsis, only Attt16 knock-out mutants with 27 an absence of TT16 protein, but not AtTT16 overexpression lines, showed abnormal endothelial 28 development and decreased PA content (Nesi et al. 2002). We also over-expressed BnTT16 in 29 wild-type Arabidopsis, and the seed color was reverted to that of wild-type. Therefore, the 30 decreased PA content and abnormal endothelial development in the B. napus RNAi plants, as 31 well as the normal seed coat morphology in the transgenic Arabidopsis lines carrying the 32 35S::BnTT16 construct in the Attt16 background indicated that BnTT16 protein levels were 33 decreased in the RNAi plants as expected. 34 As shown in 4 representative independent transgenic lines, BnTT16 expression was 35 diminished by approximately 47% to 72% compared to that in the wild-type plants (Figure S4b). 36 Expression levels of each individual BnTT16 gene were also assayed in these transgenic lines, 37 and it was found that all four genes were down-regulated in every case (Figure S5). To confirm 38 that the RNAi silencing was specific to BnTT16 homologs, we analyzed our RNAi construct with 39 a publicly available Web-based computational tool (siRNA Scan) and did not find any potential 40 off-target-silenced genes (Xu et al. 2006). In addition, the expression level of B. napus 41 AGAMOUS-LIKE 6 (BnAGL6), a gene sharing the highest cDNA sequence similarity to the 42 BnTT16 homologs based on the B. napus genome database (http://compbio.dfci.harvard.edu/cgi- 43 bin/tgi/gimain.pl?gudb=oilseed_rape) was tested by qRT-PCR. The transcriptional levels of the 44 BnAGL6 gene exhibited no difference between wild-type and Bntt16 RNAi lines in either 2-DAP 45 or 15-DAP samples (Figure S4 c). 46 2 47 Microarray analysis and data processing 48 In order to broadly identify genes regulated by the BnTT16 proteins, microarray data obtained 49 previously (Deng et al. 2012) was analyzed to compare gene expression levels in 2-DAP siliques 50 of Bntt16 RNAi and wild-type plants. In brief, microarray analysis was performed using an 51 Agilent 4x44k Brassica Gene Expression Microarray chip. Total RNA was isolated from 2-DAP 52 samples using the RNeasy Plant Mini Kit and was Cy3-labeled with the Quick Amp Labeling Kit 53 (Agilent Technologies). Hybridized arrays were scanned with a GenePix 4000B scanner 54 (Molecular Device, Sunnyvale, CA) at 5 µm resolution. Image processing was performed using 55 the Feature Extraction Software 10.5.1.1 (Agilent Technologies). 56 Microarray data were analyzed using the open-source R statistical programming language 57 (http://www.r-project.org/) and Bioconductor packages (Gentleman et al. 2004). The raw data 58 were imported into the R statistical environment, and their overall high quality was determined 59 using the arrayQualityMetrics package (Kauffmann et al. 2009). Data were normalized using 60 Variance Stabilization Normalization. Differential gene expression analysis was subsequently 61 performed using an empirical Bayes method implemented in Linear Models for Microarray 62 Analysis (Smyth 2004). A gene was considered differentially expressed in the Bntt16 RNAi line 63 compared with wild-type if its B statistic (log odds of differential expression) was ≥ 2.0, 64 corresponding to a ≥80% probability of differential expression. For homologous genes, each 65 specific copy was defined by its sequence in the genome database on the microarray chip (for 66 example, TC number from assembly (TC, Tentative Consensus sequence from assembly of 67 ESTs)), and a unique probe was designed to measure the expression level of that particular copy 68 (homolog). The functional annotations of differentially expressed genes (DEGs) in Brassica were 69 updated by running BLAST analyses (E value ≤ 10-5) against TAIR 10, the latest Arabidopsis 3 70 genome release at The Arabidopsis Information Resource (http://www.arabidopsis.org). PA- and 71 seed coat-associated genes reported in other plants were also used to assist gene identification. 72 The gene ontology (GO) annotation of Arabidopsis genes was used for GO analysis of the DEGs. 73 As expected, global similarity analysis indicated that the four wild-type samples clustered 74 together whereas the four Bntt16 RNAi samples formed a separate group, demonstrating the 75 reliability of our microarray experiment (Figure S8). Subsequently, the expression of eighteen 76 genes related to seed coat development and flavonoid biosynthesis was measured by qRT-PCR. 77 In all cases, the transcript enrichment patterns of qRT-PCR analysis were consistent with 78 microarray data (Figure S9). The consistency between qRT-PCR and microarray data was also 79 obtained in another set of genes in the same experiment (Deng, et al. 2012). The microarray data 80 have been submitted to the NCBI’s Gene Expression Omnibus and are accessible through GEO 81 Series accession number GSE37449. 82 The microarray results indicated that 1032 genes were up-regulated and 691 genes were 83 down-regulated in the Bntt16 RNAi samples compared to WT samples (Table S2, p<0.01), 84 which were subsequently summarized by gene ontology (GO) analysis. Genes with roles in 85 biological processes, protein metabolism, transport, secondary metabolism, stress and defense 86 processes, development, nucleotide metabolism, cell organization and biogenesis, and 87 transcription were significantly affected by Bntt16 down-regulation (Figure S10a). In terms of 88 molecular function, a number of these genes encoded proteins with binding properties (including 89 DNA binding and protein binding), as well as enzymes including hydrolase, transferase, and 90 kinase (Figure S10b). In terms of cellular localization, the majority of genes affected by Bntt16 91 down-regulation encoded proteins that were localized within the plastid, followed by those 92 localized to the nucleus and plasma membrane (Figure S10c). To a lesser degree, Bntt16 down- 4 93 regulation also affected genes encoding proteins localized within the cell wall, ER, and 94 mitochondria (Figure S10c). In summary, the BnTT16 proteins regulate the expression of genes 95 involved in a variety of molecular functions and biological processes. As is the case for many 96 genome-scale surveys, a large portion of the identified genes have functions that are unknown as 97 of yet (Table S2) and therefore this data may result in the identification of novel genes regulated 98 by BnTT16s. 99 100 101 Legends 102 103 Figure S1. Comparison of Brassica TT16 genomic sequences. Exons and introns were labelled 104 with black and white backgrounds respectively. Bn, B. napus; AA, B. rapa; CC, B. oleracea. The 105 TT16 genomic DNA sequences cloned in this study were submitted to GenBank as follows: 106 BnTT16.1, JF970611; BnTT16.2, JF970612; BnTT16.3, JF970613; BnTT16.4, JF970614; 107 BoTT16.1, JF970615; BoTT16.2, JF970616; BoTT16.3, JF970617; BoTT16.4, JF970618; 108 BrTT16.1, JF970619; BrTT16.3, JF970620; BrTT16.4, JF970621. 109 110 Figure S2. Schematic diagram of BnTT16 overexpression constructs used to complement the 111 Arabidopsis tt16-6 mutant phenotype. 112 113 Figure S3. Localization of proanthocyanidins using p-dimethylaminocinnamaldehyde staining in 114 developing seeds of complemented Arabidopsis lines. (a) wild-type Arabidopsis Ws-3 as a 115 positive control; (b) Arabidopsis transparent testa 16-6 (Attt16-6) mutant as a negative control; 5 116 (c) representative complemented line expressing Brassica napus BnTT16.1 (Attt16- 117 6/35S::BnTT16.1); (d) representative complemented line expressing BnTT16.2 (Attt16- 118 6/35S::BnTT16.2); (e) representative complemented line expressing BnTT16.3 (Attt16- 119 6/35S::BnTT16.3); (f) representative complemented line expressing BnTT16.4 (Attt16- 120 6/35S::BnTT16.4). Scale bar = 100 µm. The arrows indicate the endothelium containing PA. 121 122 Figure S4. RNAi-mediated silencing of BnTT16 expression. (a) Schematic diagram of the 123 construct utilized for RNAi silencing of BnTT16 homologs in B. napus. (b) The overall level of 124 expression of BnTT16 genes in open flowers (0-DAP) was down-regulated in Bntt16 RNAi lines. 125 (c) The expression level of B. napus AGAMOUS-LIKE 6 (BnAGL6) exhibited no significant 126 difference between wild-type (WT) and Bntt16 RNAi lines (RNAi) (p < 0.05). The data in (b) 127 and (c) were presented as the relative expression levels (2-ΔCT) of target genes compared with 128 reference genes (Table S1). 4-1, 10-3, 12-4, and 23-4 are 4 representative Bntt16 RNAi lines. 129 130 Figure S5. Transcriptional expression (2-ΔCT) of each individual BnTT16 gene in open flowers (0 131 day after pollination) was down-regulated in RNAi plants. (a) BnTT16.1; (b) BnTT16.2; (c) 132 BnTT16.3; (d) BnTT16.4. 4-1, 10-3, 12-4, and 23-4 are 4 representative Bntt16 RNAi lines. The 133 data were presented as the relative expression levels of each BnTT16 gene compared with 134 reference genes listed in Table S1. 135 136 Figure S6. Vanillin staining of whole developing seeds (14 day after pollination) indicated that 137 proanthocyanidin content was less in Bntt16 RNAi plants than wild-type plants. (a) Wild-type 6 138 whole seeds; (b) halved wild-type seeds; (c) whole seeds from RNAi plants; (d) halved seeds 139 from RNAi plants. Bars = 1 mm. 140 141 Figure S7. Localization of proanthocyanidins with vanillin in developing seeds of B. napus 142 wild-type (WT) and RNAi lines. Arrows indicate PAs that have accumulated in the endothelium. 143 (a) WT 15-DAP; (b) RNAi 15-DAP; (c) WT 20-DAP; (d) RNAi 20-DAP; (e) WT 24-DAP; (f) 144 RNAi 24-DAP; (g) WT 31-DAP; (h) RNAi 31-DAP. RNAi, Bntt16 RNAi plants; DAP, days 145 after pollination. Scale bar = 100 µm. 146 147 Figure S8. Global similarity of the eight microarray samples for wild-type B. napus and RNAi 148 plants. 149 Sample clustering was used to assess the overall similarity of the eight samples. The four 150 replicates of wild-type plants (sample 1-4) clustered with one another, while the four replicates 151 of RNAi plants formed a separate group (sample 5-8). 152 153 Figure S9. Validation of microarray data (fold-difference) using qRT-PCR. (a) Candidate genes 154 involved in flavonoid biosynthesis and regulation. (b) Candidate genes involved in seed coat 155 development. Data include the mean relative transcript levels ± standard error of four biological 156 replicates and four technical replicates. 157 158 Figure S10. Effects of Bntt16 silencing on the gene expression profile of B. napus (n=4). (a) 159 Biological process. (b) Molecular function. (c) Cellular component. GO, gene ontology. 160 Unknown and ambiguous genetic programming categories were not included. 7 161 162 Figure S11. Vanillin staining of representative dissected developing seeds. Samples were 163 harvested at 15 day after pollination (15-DAP, cotyledon stage) from wild-type B. napus plants. 164 (a) embryo; (b) endosperm; (c) inner integument; (d) epidermis. Bar = 1 mm. Briefly, we 165 dissected 15-DAP developing seed coats into three contiguous components: inner fraction 166 composed primarily of developing endosperm, middle fraction containing inner integument and 167 palisade cells, and outer fraction composed of epidermal and sub-epidermal cells (Jiang and 168 Deyholos 2010). To detect which fraction contained the endothelium, we stained these three 169 fractions and embryos with vanillin. The middle fraction highly stained to red, and the inner 170 fraction was only slightly stained. Since almost all PA accumulated in the endothelium at 15- 171 DAP (Figure 7), we confirmed that the middle fraction contained virtually all the endothelium. 172 For convenience, we named the four sub-tissue fractions as embryo, endosperm, inner 173 integument and epidermis in this study. 174 175 Table S1. Primers utilized in this study. 176 177 Table S2. Microarray data. 178 179 180 REFERENCES 181 Bondaruk, M., Johnson, S., Degafu, A., Boora, P., Bilodeau, P., Morris, J., Wiehler, W., 182 Foroud, N., Weselake, R. and Shah, S. (2007) Expression of a cDNA encoding palmitoyl- 183 acyl carrier protein desaturase from cat's claw (Doxantha unguis-cati L.) in Arabidopsis 8 184 thaliana and Brassiea napus leads to accumulation of unusual unsaturated fatty acids and 185 increased stearic acid content in the seed oil. Plant Breed., 126, 186-194. 186 Chen, X., Truksa, M., Snyder, C.L., EI-Mezawy, A., Shah, S. and Weselake, R.J. (2011) 187 Three homologous genes encoding sn-glycerol-3-phosphate acyltransferase 4 exhibit different 188 expression patterns and functional divergence in Brassica napus. Plant Physiol., 155, 851-865. 189 Deng, W., Chen, G., Peng, F., Truksa, M., Snyder, C.L. and Weselake, R.J. (2012) 190 Transparent testa16 plays multiple roles in plant development and is involved in lipid 191 synthesis and embryo development in canola. Plant Physiol., 160, 978-989. 192 Gentleman, R.C., Carey, V.J., Bates, D.M., Bolstad, B., Dettling, M., Dudoit, S., Ellis, B., 193 Gautier, L., Ge, Y.C., Gentry, J., Hornik, K., Hothorn, T., Huber, W., Iacus, S., Irizarry, 194 R., Leisch, F., Li, C., Maechler, M., Rossini, A.J., Sawitzki, G., Smith, C., Smyth, G., 195 Tierney, L., Yang, J.Y.H. and Zhang, J.H. (2004) Bioconductor: open software development 196 for computational biology and bioinformatics. Genome Biol., 5, R80. 197 198 199 200 201 Jiang, Y. and Deyholos, M. (2010) Transcriptome analysis of secondary-wall-enriched seed coat tissues of canola (Brassica napus L.). Plant Cell Rep., 29, 327-342. Kauffmann, A., Gentleman, R. and Huber, W. (2009) arrayQualityMetrics-a bioconductor package for quality assessment of microarray data. Bioinformatics, 25, 415-416. Liu, Q., Singh, S.P. and Green, A.G. (2002) High-stearic and high-oleic cottonseed oils 202 produced by hairpin RNA-mediated post-transcriptional gene silencing. Plant Physiol., 129, 203 1732-1743. 204 205 Mietkiewska, E., Hoffman, T.L., Brost, J.M., Giblin, E.M., Barton, D.L., Francis, T., Zhang, Y. and Taylor, D.C. (2008) Hairpin-RNA mediated silencing of endogenous FAD2 gene 9 206 combined with heterologous expression of Crambe abyssinica FAE gene causes an increase in 207 the level of erucic acid in transgenic Brassica carinata seeds. Mol Breeding, 22, 619-627. 208 Nesi, N., Debeaujon, I., Jond, C., Stewart, A.J., Jenkins, G.I., Caboche, M. and Lepiniec, L. 209 (2002) The TRANSPARENT TESTA16 locus encodes the ARABIDOPSIS BSISTER MADS 210 domain protein and is required for proper development and pigmentation of the seed coat. 211 Plant Cell, 14, 2463-2479. 212 213 Smyth, G.K. (2004) Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet. Mol. Biol., 3, Article3. 214 Stoutjesdijk, P.A., Singh, S.P., Liu, Q., Hurlstone, C.J., Waterhouse, P.A. and Green, A.G. 215 (2002) hpRNA-mediated targeting of the Arabidopsis FAD2 gene gives highly efficient and 216 stable silencing. Plant Physiol., 129, 1723-1731. 217 Xu, P., Zhang, Y., Kang, L., Roossinck, M.J. and Mysore, K.S. (2006) Computational 218 estimation and experimental verification of off-target silencing during posttranscriptional gene 219 silencing in plants. Plant Physiol., 142, 429-440. 220 10