Core Chemistry (C1) check list
advertisement

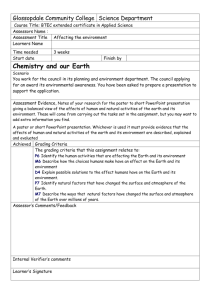
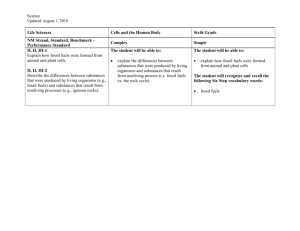
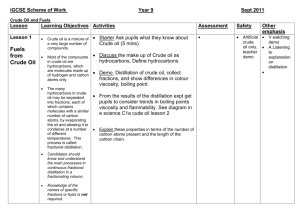
Chemistry 1: How can I improve? Tick the ‘I can statements’ on the right hand side once completed in lesson. Your teacher will write objective numbers in your book to help show you what you need to spend time on in order to improve your grade.(E.g Ob 9). C1.1.1 Atomic Structure Test grade □ C1.1.2 Reactivity and the periodic table Test grade □ C1.1.3 Bonding and equations Test grade □ C1.2.1 Calcium carbonate Test grade □ 1 .Define the terms element and compound 2. Describe the properties of the subatomic particles 3. Describe the relationship between the sub-atomic particles and the atomic number and mass number 4. Describe how the sub-atomic particles are arranged in an atom. 5. Explain how the atomic structure of an element is related to its position in the periodic table 6. Construct diagrams of the electronic structures of the first 20 elements 7. State the connection between the group number and the number of outer electrons of an element. 8.Relate the chemical behaviour of a group to the number of electrons 9.Explain why group 0 elements are inert. 10. State that elements react together to form compounds 11. Describe the 2 ways that elements can obtain a full outer shell. 12. Explain why elements react. 13.Define a chemical reaction 14. Explain why mass is conserved in a chemical reaction 15. Write word equations. 16. Write symbol equations (HT) C1.2.2 Extracting metals Test grade □ 17. State the chemical name and formula of limestone. 18. Describe how limestone is quarried and used as a building material. 19. Evaluate the effect of quarrying on the environment. 20. Describe the thermal decomposition of the carbonates of calcium, zinc, magnesium, copper and sodium. 21. Describe the reaction of calcium oxide with water and how the product is used in neutralisation reactions. 22. Describe the test for carbon dioxide 23. Describe the reaction of metal carbonates with acid. 24. Describe how limestone is used to make cement, mortar and concrete. 25. List examples of metals that are found in the native state or as ores 26. Describe how copper and iron are extracted from their oxides. 27. Explain why carbon is used in the extraction of iron and copper. 28. Explain why metals above carbon in the reactivity series are extracted by electrolysis. 29. Explain why current methods of extracting titanium and aluminium are expensive. 30. Compare the different methods of extracting copper in terms of economics and environmental impacts. 31. Explain the benefits of recycling metals. C1.3.2 Alloys Test grade □ 33. Define and draw a diagram of an alloy. 34. State why iron from the blast furnace is not very useful 35.Explain why alloys are harder than the pure metal C1.4.3 hydrocarbon fuels Test grade □ 36 Compare the properties of different alloys 37. Evaluate the use of different alloys when given information about composition and properties. C 1.3.3 Properties and uses of metals Test grade □ C1.4.1 Crude oil Test grade □ 51. Describe the environmental effects of burning fossil fuels. 52. Identify solutions to the environmental effects of burning fossil fuels. 53. Describe the economic, ethical and environmental issues around the use of biofuels. 38.State the properties of transition metals 39.Describe the uses of iron, steel, aluminium, titanium and copper. 40. Explain why copper is used for electrical wiring and plumbing. 41. State that crude oil is a mixture of a large number of compounds and define the term mixture. 42. Describe how a mixture may be separated using the physical properties of the compounds that make it up (distillation). 43. Describe the compounds in crude oil using the terms hydrocarbons, saturated and alkanes and using the general formula for an alkane. C1.5.1 Obtaining useful substances from crude oil Test grade □ C1.5.2 Polymers Test grade C1.4.2 Hydrocarbons Test grade □ 44. Recognise alkanes from their formulae and know the names and structures of methane, ethane, propane and butane. 45. Describe the purification of crude oil by fractional distillation. 46. Describe the relationship between molecular size and the boiling point, viscosity and the flammability of the fractions. 47. Relate the products of burning fuels to the elements present in the fuels and to the extent of combustion (partial or complete). 48. Describe the combustion of fuels as an oxidation reaction that releases energy 49. Write word equations to describe the combustion for hydrocarbon fuels 50. Write symbol equations for the combustion of hydrocarbon fuels (HT) □ 54. Describe the process of cracking 55. Explain why cracking is useful. 55..Describe the products of cracking. 56.State the general formula for an alkene 57. Recognise alkenes from their formulae and know the names and structures of ethene and propene. 58. State the test for an alkene. 59. Use the terms polymerisation, polymer and monomer. 60. Represent the formation of a polymer from a given monomer. 61. Describe the environmental issues of using polymers and solutions to the issues. C1.5.2 Test grade 63. Compare the 2 methods of producing ethanol. □ C1.6.1 Vegetable oils Test grade □ C1.6.2 Emulsions Test grade □ C1.7.1 The Earth’s crust Test grade □ 64. Describe the 2 methods of producing ethanol using word equations. 65. Describe the extraction of plant oils by pressing and distillation. 66. Outline the importance of vegetable oils as foods and fuels. 67. Compare cooking foods using plant oils and boiling in water. 68. State how emulsions are produced and identify the role of emulsifiers. 69. Relate the uses of emulsions to their properties. 70. Describe the structure of the molecules in emulsions (HT) 71. Label a diagram showing the structure of the Earth and say what the sections of crust are called. 72. Describe how the tectonic plates move 73. Compare Wegeners theory of continental drift with the theory of the shrinking Earth. 74. Explain why Wegeners theory was not accepted for many years 75. Describe what happens at the boundaries between tectonic plates. 76. Explain why scientists cannot accurately predict earthquakes and volcanic eruptions. C1.7.2 The Earth’s atmosphere Test grade □ 77. List the proportions of gases in the atmosphere today. 78. Describe the role of volcanic activity in the formation of the early atmosphere 79. Compare the early atmosphere of Earth to that on other planets when provided with information. 80. State that there are many theories as to how life was formed 81. Relate the Miller-Urey experiment to the Primordial soup theory (HT) 82. Describe the role of plants and algae in forming the current atmosphere. 83. Describe how carbon is locked up in sedimentary rocks. 84. Explain the role of the oceans as a reservoir for carbon dioxide and the impact of increased amounts of carbon dioxide on the marine environment. 85. Describe the effect of burning fossil fuels on the atmosphere and the environment 86. Describe how fractional distillation can be used to separate the gases in the air for use in industrial processes