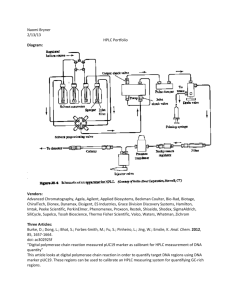
sladetx01
advertisement

1 Auxiliary Material for 2 Multiphase OH Oxidation Kinetics of Organic Aerosol: 3 The Role of Particle Phase State and Relative Humidity 4 J. H. Slade1 and D. A. Knopf1* 5 6 7 8 9 1 Institute for Terrestrial and Planetary Atmospheres/School of Marine and Atmospheric Sciences, Stony Brook University, Stony Brook, NY 11794, USA * Correspondence to: Daniel.Knopf@StonyBrook.edu Geophysical Research Letters, 2014 10 1. Observed Loss Rate Dependence (k1) on Relative Humidity 11 As shown in Eq. (3) in the manuscript, dL depends on both the diffusion coefficient of 12 OH, DOH, in levoglucosan, and the observed pseudo-first order loss rate of condensed-phase OH, 13 𝑘1𝐵 = k2[LEV]B [Abbatt et al., 2012], which both vary as a function of RH. k2 is the second- 14 order rate constant derived from γ at a specific RH (see Table 1 in article), which is also a 15 function of the pseudo-first order loss rate of gas-phase OH, k1. [LEV]B is the bulk concentration 16 of levoglucosan, approximated as 6.27x1021 molecule cm-3, which assumes a densely packed 17 system formed exclusively by LEV molecules (molecular volume of 1.59x10-22 cm3) [Arangio et 18 al., 2015] Therefore, to plot a smooth dL as a function of RH as shown in Fig. 1(a) requires a 19 parameterization for 𝑘1𝐵 as a function of RH. Figure S4 shows derived 𝑘1𝐵 as a function of RH. 20 Based on the scatter in the data, we assume that 𝑘1𝐵 depends linearly on RH with 𝑘1𝐵 = 4x107RH 21 + 109. Note, however, that 𝑘1𝐵 and also by extension k1 varies with particle surface area density, 22 Sa, as shown in Fig. S3, and thus the linear function does not account for changes in 𝑘1𝐵 or k1 due 23 to changes in Sa. For example, the large scatter in k1_cond at RH=0% as shown in Fig. S4 is due to 24 the wide range in applied Sa. A sensitivity analysis on the dependence of k1 as a function of RH 25 at fixed Sa may resolve this bias, but is beyond the scope of this study. 26 2. Langmuir-Hinshelwood Description Accounting for Co-Adsorption of H2O 27 The modeled γ as shown in Fig. 1(b) in the manuscript assumes OH undergoes a 28 Langmuir-Hinshelwood reactive uptake mechanism with 4-methyl-5-nitrocatechol (MNC) and 29 that H2O partially inhibits reactive uptake of OH by MNC by competing for surface reactive sites 30 and is based on the following equation [Pöschl et al., 2001] 31 𝛾=𝜎 4𝑘s [Org]𝐾OH , OH 𝜔OH (1+𝐾OH [OH]+𝐾H2 O [H2 O]) 32 where ks is the surface reaction rate constant between OH and the organic, [Org] is the surface 33 concentration of the organic, i.e. MNC, estimated to be 2×1014 molecule cm-2 [Slade and Knopf, 34 2013], KOH is the Langmuir adsorption constant of OH taken as 3.96×10-10 cm3 molecule-1 as 35 derived from OH adsorption on nitroguaiacol, an isomer of MNC [Slade and Knopf, 2013], σOH 36 is the surface area occupied by one adsorbed OH molecule approximated as 1.08×10-15 cm2 37 molecule-1 [Slade and Knopf, 2013], and [OH] and [H2O] are the gas phase concentrations of OH 38 and H2O, respectively. [H2O] was determined from the RH measurements in the flow reactor and 39 the vapor pressure of water at T=293 K. Both ks and KH2O are free fitting parameters, which were 40 determined by fitting the data in Fig. 1(b) to the above equation applying a non-linear least 41 squares fit using a Levenberg-Marquardt algorithm. The best fit parameters to ks and KH2O are 42 4.29(±0.70)×10-17 cm2 molecule-1 s-1 and 2.01(±1.07)×10-17 cm3 molecule-1, respectively. The 43 derived ks is in agreement with ks of 8.38(±8.44)×10-17 cm2 molecule-1 s-1 derived from the OH 44 surface reaction with nitroguaiacol [Slade and Knopf, 2013]. Excluded in this function are the 45 possible influences of other co-adsorbates that may suppress OH reactive uptake. For example, 46 recombination of OH to H2O2 in the adsorbed water layer [Hanson et al., 1992] may limit OH 47 reactive uptake by the organic. Increasing RH would result in more monolayers of water on the 48 surface of the particle, thus extending the time for OH to diffuse through the water layers to react 49 with the organic, enabling greater production of H2O2. Since H2O2 is less reactive than OH with 50 organics [Lai et al., 2014], less of the organic is degraded and the additional presence of H2O2 51 may compete as well with OH for surface reactive sites. Clearly, more systematic studies are 52 needed to properly assess the roles of other possible co-adsorbates that may also limit the 53 reactive uptake of OH. 54 55 References 56 Abbatt, J. P. D., A. K. Y. Lee, and J. A. Thornton (2012), Quantifying trace gas uptake to tropospheric 57 aerosol: recent advances and remaining challenges, Chem. Soc. Rev., 41(19), 6555-6581, 58 doi:10.1039/C2cs35052a. 59 Arangio, A. M., J. H. Slade, T. Berkemeier, U. Poschl, D. A. Knopf, and M. Shiraiwa (2015), Multiphase 60 chemical kinetics of OH radical uptake by molecular organic markers of biomass burning aerosols: 61 humidity and temperature dependence, surface reaction, and bulk diffusion, J. Phys. Chem. A, 62 doi:10.1021/jp510489z. 63 Hanson, D. R., J. B. Burkholder, C. J. Howard, and A. R. Ravishankara (1992), Measurement of OH and 64 HO2 radical uptake coefficients on water and sulfuric-acid surfaces, J. Phys. Chem., 96(12), 4979-4985, 65 doi:10.1021/J100191a046. 66 Lai, C., Y. Liu, J. Ma, Q. Ma, and H. He (2014), Degradation kinetics of levoglucosan initiated by 67 hydroxyl 68 doi:10.1016/j.atmosenv.2014.03.054. 69 Pöschl, U., T. Letzel, C. Schauer, and R. Niessner (2001), Interaction of ozone and water vapor with 70 spark discharge soot aerosol particles coated with benzo[a]pyrene: O3 and H2O adsorption, 71 benzo[a]pyrene degradation, and atmospheric implications, J. Phys. Chem. A, 105(16), 4029-4041, 72 doi:10.1021/jp004137n. 73 Slade, J. H., and D. A. Knopf (2013), Heterogeneous OH oxidation of biomass burning organic aerosol 74 surrogate compounds: assessment of volatilisation products and the role of OH concentration on the 75 reactive uptake kinetics, Phys. Chem. Chem. Phys., 15(16), 5898-5915, doi:10.1039/C3cp44695f. 76 radical under different environmental conditions, Atmos. Environ., 91, 32-39,