Fractional Distillation of Two Volatile Liquids
advertisement

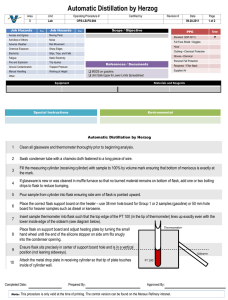
Simple and Fractional Distillation of Two Volatile Liquids Containing Non-volatile Impurities 1. Simple Distillation of Two Volatile Liquids Containing Non-volatile Impurities Assemble the simple distillation apparatus as shown in the pictures below. Use a 100 mL round-bottomed flask for the still pot. Remove the Thermometer/adapter assembly from your apparatus, and insert your long-stem plastic funnel. Pour 60 mL of mixture through still head into the 100 mL round-bottomed flask. Note the color and clarity of your mixture on your laboratory data sheet. Remove the funnel from your apparatus and add your magnetic stirring bar. Replace the thermometer/ adapter assembly onto your apparatus. Place a 100 mL graduated cylinder so that the tip of the condenser is centered over the neck of the cylinder. Turn the water on so that a gentle flow comes from the discharge end of the apparatus. Have your instructor check your apparatus before you start heating. Start stirring and begin heating the distillation flask on a setting of “4”. As soon as the liquid begins to boil and the condensing vapors have reached the thermometer bulb, regulate the heat supply so that distillation continues steadily at 1 drop per second. As soon as drops begin falling into the receiver, record the temperature from the thermometer in the distillation head. Record the temperature every four mL thereafter. Once you have observed a second temperature plateau – two consecutive equivalent temperature readings – discontinue heating and allow the flask to cool until the remaining solution in the flask no longer bubbles. The distillation is halted at this point to streamline the exercise to conserve time. Draw a Temperature vs. Volume graph of your lab data on your laboratory report form. Note the boiling ranges of the mixture on your lab report form, and note the color and clarity of your distillate. 2. Fractional Distillation of Two Volatile Liquids Containing Non-volatile Impurities Pour the mixture collected from the simple distillation back into the cooled 100 mL round-bottomed flask. Make note of the color and clarity of the mixture on your lab data sheet. Place a 100 mL graduated cylinder so that the tip of the condenser is centered over the neck of the cylinder, as shown on the demonstration set-up. Place a 100 mL beaker directly under the suspended 100 mL graduated cylinder. Wrap aluminum foil around the Still head and still pot. Have your instructor examine your apparatus before commencing heating. Start the mixture stirring, heating initially at ”7”. Adjust heat so that the distillate comes over at About 1-2 drops/second. Collect the first fraction in your 100 mL graduated cylinder when the thermometer reaches a plateau. Record the temperature every 2 mL thereafter. When the drip rate slows to an occasional drop every so often, remove the 100 mL graduated cylinder, And substitute your 50 mL graduate. Continue collecting and noting temperature readings at 2 mL intervals until a second plateau is reached. When the thermometer reaches the second plateau, collect an additional 2 mL, then discontinue distillation. Note the volume of the first component recovered. Turn in this fraction in an appropriately labeled narrow mouth bottle. Record the volume of the second and third fraction collected, then, return them to the recovery bottle/flask in the front of the lab Plot a Temperature vs. Volume graph of your lab data on your lab report form. Che 301 Lab Data Sheet Title: Simple and Fractional Distillation of Two Volatile Liquids Containing Non-volatile Impurities Name:_____________________________________________________ Locker #:_____________ Date:______________________ Lab Data 1. Simple Distillation of Two Volatile Liquids Containing Non-volatile Impurities Liquid Used: _____________________________________________________________________ Appearance of the liquid ( circle the appropriate description(s) ) Colorless color cloudy clear other comments:_______________________________________________________________________ Volume of Liquid Used: mL. Temp. at which first drops enter the receiving vessel: __ oC Temperature/Volume Data: Fraction 1 Vol (mL) 4 8 12 16 20 Temp (oC) 2. Fractional Distillation of Two Volatile Liquids Containing Non-volatile Impurities Volume (mL) Temperature (oC) Temperature/Volume Data: Fractions 2 & 3 Volume (mL) Temperature (oC) Conclusions: (boiling range of first fraction) o C o (boiling range of third fraction) C Where would you change the receiver to collect fractions if you were trying to separate the components ? Calculate the mole fraction in a 1:1 (v/v) mixture using the data for the two components given below. Compound Boiling Range o C Molecular Weight Density gm/mL Volume mL Methanol 65 32.0 0.792 50 1-butanol 118 74.1 0.810 50