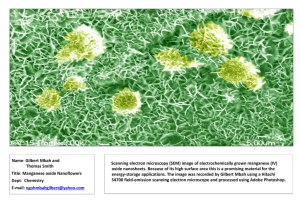
Manganese Chloride Lab
advertisement

Name __________________________________ Manganese Chloride Lab Mass your piece of manganese and record below. Balance each equation. Then, using your mass of manganese, calculate the theoretical mass of manganese chloride that can be produced for each hypothetical reaction. Theoretical mass of product Mn(s) + HCl(aq) MnCl(aq) + H2(g) _____________ g MnCl Mn(s) + HCl(aq) MnCl2(aq) + H2(g) _____________ g MnCl2 Mn(s) + HCl(aq) MnCl3(aq) + H2(g) _____________ g MnCl3 Mn(s) + HCl(aq) MnCl4(aq) + H2(g) _____________ g MnCl4 Mn(s) + HCl(aq) MnCl7(aq) + H2(g) ____________ g MnCl7 Mass the empty flask and then place the piece of manganese in the flask and react with an excess of 7M HCl. When the reaction is complete, boil off the excess acid until completely dry. Data Table: Mass of manganese metal mass of empty, dry flask Mass of dry product + flask Mass of just the dry product What is your product? Explain any difference between the actual and theoretical mass of your product.