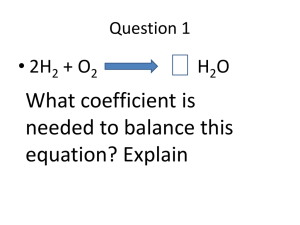
General Chemistry Midterm Name: Part I Vocab: Matter Compound
advertisement

General Chemistry Midterm Name:_______________________________ Part I Vocab: Matter Compound Element Density Heterogeneous mixture Homogeneous mixture Filtration Chromatography Distillation Chemical property / change Physical property / change Atomic number Mass number Isotope nonmetal Metal Metalloid Ductile Malleable Ion Synthesis reaction Decomposition reaction Combustion reaction Single replacement reaction Double replacement reaction Part II: Describe the following 1. Atomic theory 2. Atomic structure (use protons, neutrons, electrons) 3. Law of conservation of mass 4. The difference between an observation, hypothesis and conclusion 5. Democritus’s contribution to the atomic theory 6. Difference between a qualitative observation and a quantitative observation. 7. Difference between the independent and dependent variables. Part III: Know the following information: 1. How to name ionic and molecular compounds. 2. How to balance and classify chemical reactions. 3.. How to calculate density. D = mass / volume (g/ml) Part IV: Solve the following problems: 1. A 5mL sample of water has a mass of 5g. What is the density of water? 2. A cube of aluminum with volume = 5 cm3 has a mass of 20g. What is the density of aluminum? 3. What is the volume of a sample that has a mass of 20g and density of 4 g/mL? 4. What is the atomic number for the following elements? a. at atom with 37 electrons b. an atom with 72 protons c. an atom with 1 electron d. an atom with 6 neutrons and a mass number of 12