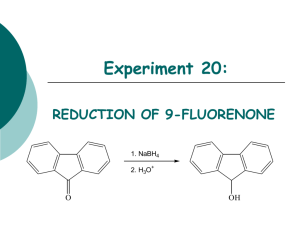
Electrophilic Aromatic Substitution Lab Assignment
advertisement

CHML212 Exp. 15 Substituent Effects on Electrophilic Aromatic Substitution IN-LAB ASSIGNMENT: (EACH STUDENT will record the following data in his/her laboratory notebook in a well organized manner during lab period. Yellow carbon copies will be submitted for grading along with Post-Lab assignment.) …..max score = 20 pts. 6. Data/Calculations a. b. c. d. Record reaction times for each aromatic compound Include a sketch of the TLC diagram including cm measurements for the solvent front and any spots which appear on the TLC plate Record the TLC developing solvent system identity Show Rf value calculations, using correct significant figures 7. In-lab Questions (The following questions should be answered in laboratory notebook.) a. Rate the relative reactivity toward electrophilic aromatic substitution of benzene substituted with the following: (1 = most reactive, 6 = least reactive) CH3 C N F CH2CH2CH3 O OCH2CH3 H3C N CH3 OH _______ b. _______ _______ _______ _______ _______ Draw the structure of the major product, and a complete mechanism for its formation, for the bromination of acetanilide which took place in this experiment. CHML212 Exp. 15 Substituent Effects on Electrophilic Aromatic Substitution POST-LAB ASSIGNMENT: (EACH LAB GROUP will submit one copy of a typewritten, paragraph style report addressing all of the points listed below. Must be written using PAST TENSE, PASSIVE VOICE. ) …..max score = 50 pts. 8. Experimental (Write 1-2 paragraphs including all of the following. Do NOT present a bulleted outline.) What type of reaction did you perform? Describe your actual synthetic procedure. Include names of any reactants used and desired product, as well as name of solvent and catalyst used (if any). Be sure to give names and actual volume/mass of any compound used during the synthesis (not just what the lab manual tells you to use). Describe the purification technique used to isolate the desired product, including names and actual volume/mass and any compound used during purification process. Describe the analytical technique used to evaluate the product, including name and actual volume/mass of any compounds used during TLC analysis. 9. Results (Cut and paste the completed tables into your document. May complete substituents and TLC diagram by hand once placed in document.) Compound ethyl benzene Table 15.1 Reactivity rates of aromatic compounds anisole acetanilide phenol benzaldehyde nitrobenzene Structure OPA, OPD, or MD Reaction Order (1 = most reactive, 6 = least reactive) Table 15.2 TLC results TLC Diagram Name of Compound Rf Values Standard Sample Solvent Front= _________cm o-bromoacetanilide m-bromoacetanilide TLC developing solvent used: _________________________ p-bromoacetanilide 10. Discussion (Write 1-2 paragraphs including the following.) Discuss the reaction order of the compounds listed in Table 15.1. Are the observed reaction rates consistent with what one would expect based on knowledge of activating and deactivating substituents? Describe what was expected to happen and what actually happened. If the obtained results are different than expected, explain the difference. In this experiment, the aromatic compound is used in excess and the bromine is the limiting reagent. Would one be able to determine the reaction rates based on the disappearance of color if bromine was used in excess? Briefly explain. Include a short comment addressing what could be done differently to improve the experimental results, if repeated.