Role of nitrogen in the biosphere
advertisement

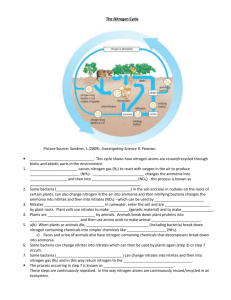
Role of nitrogen in the biosphere The growth of all organisms depends on the availability of mineral nutrients, and none is more important than nitrogen, which is required in large amounts as an essential component of proteins, nucleic acids and other cellular constituents. There is an abundant supply of nitrogen in the earth's atmosphere - nearly 79% in the form of N2 gas. However, N2 is unavailable for use by most organisms because there is a triple bond between the two nitrogen atoms, making the molecule almost inert. In order for nitrogen to be used for growth it must be "fixed" (combined) in the form of ammonium (NH4) or nitrate (NO3) ions. The weathering of rocks releases these ions so slowly that it has a negligible effect on the availability of fixed nitrogen. So, nitrogen is often the limiting factor for growth and biomass production in all environments where there is suitable climate and availability of water to support life. Microorganisms have a central role in almost all aspects of nitrogen availability and thus for life support on earth: a. some bacteria can convert N2 into ammonia by the process termed nitrogen fixation; these bacteria are either free-living or form symbiotic associations with plants or other organisms (e.g. termites, protozoa) b. other bacteria bring about transformations of ammonia to nitrate, and of nitrate to N2 or other nitrogen gases c. many bacteria and fungi degrade organic matter, releasing fixed nitrogen for reuse by other organisms. All these processes contribute to the nitrogen cycle. We shall deal first with the process of nitrogen fixation and the nitrogen-fixing organisms, then consider the microbial processes involved in the cycling of nitrogen in the biosphere. The nitrogen-fixing organisms All the nitrogen-fixing organisms are prokaryotes (bacteria). Some of them live independently of other organisms - the so-called free-living nitrogen-fixing bacteria. Others live in intimate symbiotic associations with plants or with other organisms (e.g. protozoa). Examples are shown below. Examples of Nitrogen fixing bacteria Symbiotic with plants Free Living Aerobic Azotobacter Klebsiella some cyanobacteria Anaerobic Clostridium Purple Sulphur bacteria Legumes Rhizobium other plants Azospirillum Frankia NITROGEN CYCLE Nitrogen fixation The nitrogen molecule (N2) is quite inert. To break it apart so that its atoms can combine with other atoms requires the input of substantial amounts of energy. Three processes are responsible for most of the nitrogen fixation in the biosphere: 1. biological fixation by certain microbes — alone or in a symbiotic relationship with some plants and animals 2. industrial fixation 3. atmospheric fixation by lightning Mechanism of biological nitrogen fixation Picture of root nodule Nitrogen-fixing Rhizobium lives in the root nodules of leguminous plants Biological nitrogen fixation can be represented by the following equation, in which two moles of ammonia are produced from one mole of nitrogen gas, at the expense of 16 moles of ATP and a supply of electrons and protons (hydrogen ions): N2 + 8H+ + 8e- + 16 ATP = 2NH3 + H2 + 16ADP + 16 Pi A point of special interest is that the nitrogenase enzyme complex is highly sensitive to oxygen. It is inactivated when exposed to oxygen, because this reacts with the iron component of the proteins. Biological nitrogen fixation requires a complex set of enzymes and a huge expenditure of ATP. Although the first stable product of the process is ammonia, this is quickly incorporated into protein and other organic nitrogen compounds. In the symbiotic nitrogen-fixing organisms such as Rhizobium, the root nodules of leguminous plants contain oxygen-scavenging molecules such as Leghaemoglobin,(L b) which shows as a pink colour when the active nitrogenfixing nodules of legume roots are cut open. Leghaemoglobin regulate the supply of oxygen to the nodule tissues in the same way as haemoglobin regulates the supply of oxygen to mammalian tissues. They also form symbiotic associations with other organisms such as the water fern Azolla, and cycads. The association with Azolla, where cyanobacteria (Anabaena azollae) are harboured in the leaves, has sometimes been shown to be important for nitrogen inputs in rice paddies, especially if the fern is allowed to grow and then ploughed into the soil to release nitrogen before the rice crop is sown. Industrial FixationHaber –Bosch process Haber-Bosch was the first industrial chemical process to use high pressure for a chemical reaction. It directly combines nitrogen from the air with hydrogen under extremely high pressures and moderately high temperatures. N2 (g) + 3 H2 (g) ---------------------2 NH3 (g) Atmospheric Fixation The enormous energy of lightning breaks nitrogen molecules and enables their atoms to combine with oxygen in the air forming nitrogen oxides. These dissolve in rain, forming nitrates, that are carried to the earth. Atmospheric nitrogen fixation probably contributes some 5– 8% of the total nitrogen fixed. Decay The proteins made by plants enter and pass through food webs just as carbohydrates do. At each trophic level, their metabolism produces organic nitrogen compounds that return to the environment, chiefly in excretions. The final beneficiaries of these materials are microorganisms of decay. They break down the molecules in excretions and dead organisms into ammonia. Nitrification Ammonia can be taken up directly by plants — usually through their roots. However, most of the ammonia produced by decay is converted into nitrates. This is accomplished in two steps: -Bacteria of the genus Nitrosomonas oxidize NH3 to nitrites (NO2−). -Bacteria of the genus Nitrobacter , Nitrococcus oxidize the nitrites to nitrates (NO3−). These two groups of autotrophic bacteria are called nitrifying bacteria. Through their activities (which supply them with all their energy needs), nitrogen is made available to the roots of plants. Nitrification occurs in well aerated soil and water. Many legumes, in addition to fixing atmospheric nitrogen, also perform nitrification — converting some of their organic nitrogen to nitrites and nitrates. These reach the soil when they shed their leaves. Excess of nitrate ions can be washed to water bodies causing Eutrophication. Denitrification The above processes remove nitrogen from the atmosphere and pass it through ecosystems. Denitrification reduces nitrates to nitrogen gas, thus replenishing the atmosphere. Denitrifying bacteria live in soil and water where oxygen supply is low and use nitrate ions as a source of oxygen for aerobic respiration examplePseudomaonas denitrificans. Once again, bacteria are the agents. They live deep in soil and in aquatic sediments where conditions are anaerobic. They use nitrates as an alternative to oxygen for the final electron acceptor in their respiration. Thus they close the nitrogen cycle.