Chapter 17: Particle Physics
advertisement

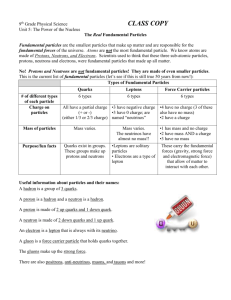
Chapter 17: Particle Physics Particles physics is governed by the standard model. This model is made up of 16 fundamental particles, which cannot be broken down any further and can be used to build the entire universe. There are 12 matter particles and 4 boson (force) particles. Each particle, besides the photon, also has a corresponding antiparticle. These are the 16 particles of the current standard model; Matter and Antimatter For ever particle there is a corresponding antiparticle which; Annihilates the particle and itself if they meet, converting their total mass into photons Has exactly the same mass as the particle at rest Has exactly the opposite charge to the particle, if it has a charge at all In annihilation, charge, momentum and energy must be conserved. When energy is converted directly into mass using 𝐸 = 𝑚𝑐 2 , equal amounts of matter and antimatter must be created. Pair Production If two protons are collided at high speed, a lot of energy is present at the point of collision. This energy can create more particles. If an extra proton is created then an anti proton must also be created. This process of creating particle-antiparticle pairs is called pair production. Any high energy collision can cause pair production (even a single high energy gamma photon) as long as the total energy at the start is more than the rest energy of the products of the collision. Electron-positron pairs are created more than any other pair of particles because they have the smallest combined mass, so the least energy is required to produce them. Pair production from the collision of two photons is incredibly rare and difficult to achieve. Electron-Positron Annihilation When an electron and positron collide all their mass is converted into energy. This energy takes the form of two gamma photons. There must be two in order to conserve the original momentum of the electron-positron pair. Particle Interactions When a single force acts on an object, it changes the momentum of the object. When two objects act on each other they exert equal and opposite forces on each other, momentum is transferred by these forces. If two electrons approach each other they repel each other and move away. This interaction is mediated by the electromagnetic force, using a virtual photon (virtual because it cannot be directly detected) to exchange momentum. The virtual photon exchange can be represented with a Feynman diagram; An electron-positron interaction also involves the exchange of a photon but in this case there is an attractive force, not a repulsive one. The electromagnetic field creates and destroys photons, photons are created at one point where they travel to another and are destroyed. They are created and destroyed by the interaction of the field with charges on particles. Classifying Particles The first, simplest division of particles is into a group of those who interact through the strong interaction, and a group who do not; hadrons and leptons respectively. Hadrons Are particles and antiparticles that interact through the strong interaction and the electromagnetic interaction (if they are charged). Hadrons are not fundamental particles, they are all made of quarks and antiquarks. The hadron group includes protons, neutrons, pi mesons, K mesons etc. Hadrons decay through the weak interaction, apart from the proton which is stable. Hadrons can be further divided into two subgroups; Baryons are protons and all other hadrons which decay into protons (such as the neutron) either directly or indirectly. Mesons are hadrons that do not include protons in their decay products (basically all the hadrons that aren’t baryons) Leptons Are particles and antiparticles which do not interact with the strong interaction. The lepton group includes electrons, muons, neutrinos etc. These particles still interact with the weak force and the electromagnetic force if charged. The Baryon Family Baryon particles include the proton and neutron. Protons and neutrons are two versions of the same particle; the nucleon. The neutron decays in 13 minutes, the proton is stable and does not decay, or at least not for 1032 years. All baryons (except the proton) decay into the proton. In a particle reaction the number of baryons is always conserved. The total baryon number on each side of an equation is always the same, baryons (three quarks) have a baryon number of +1, mesons (one quark, one antiquark) a baryon number of 0, and antibaryons (three antiquarks) have a baryon number of −1. The Meson Family Mesons comprise a wide range of particles and include pi mesons (pions), K mesons (kaons) and others. All mesons are unstable and have a baryon number of 0 (seeing as none of them can decay into protons, they are not baryons). Pions are the lightest mesons. The Lepton Family The strong force holds neutrons and protons in a nucleus but does not cause a proton to change into a neutron or vice versa. These changes cannot be due to the electromagnetic force as the neutron has no charge. It whatever causes it must also be weaker than the strong force or it would affect stable nuclei. This force is referred to as the weak nuclear force/weak interaction. Leptons are particles which are not affected by the strong force; this includes electrons, muons, neutrinos etc. The weak force changes leptons into other leptons. Leptons can be produced or annihilated in particle-antiparticle interactions. Leptons are/are made of fundamental particles. Leptons and antileptons can interact to produce hadrons Leptons are assigned a number with +1 for a lepton and -1 for antileptons, 0 for something that is not a lepton. Lepton number must be conserved in a particle reaction. Conservation of Energy The total energy of the particles and antiparticles before the collision is equal to their rest energy + their kinetic energy before, also equal to the total rest energy + kinetic energy afterwards. The resultant products of a collision must have a total rest energy less than or equal to the total energy before the collision. Particles and their Properties Particle, Symbol Charge Antiparticle, Symbol Charge Rest Energy (MeV) Interaction all are affected by the weak force Proton, 𝐩 +1 Antiproton, p̅ -1 938 Strong, electromagnetic Neutron, 𝐧 0 Antineutron, n̅ 0 939 Strong Electron, 𝐞− -1 Positron, e+ +1 0.511 Electromagnetic Neutrino, 𝐯 0 Antineutrino, v̅ 0 0 Weak only Muon, 𝛍− +1, 0, -1 Antimuon, μ+ -1, 0, +1 106 Electromagnetic Pi Meson 𝛑+ , 𝛑𝟎 +1, 0 Anti Pi Meson π− , π−0 -1, 0 140, 135, 140 Strong, electromagnetic K Meson 𝐊+, 𝐊𝟎 +1, 0 Anti K Meson K − , K −0 -1, 0 494, 498, 494 Strong, electromagnetic Nuclear Stability Not all combinations of neutrons and protons make for stable nuclei. Light nuclei contain a mostly equal number of protons and neutrons but as atomic mass increases, nuclei usually contain more neutrons than protons. Nuclei above the stability line undergo beta minus decay, nuclei below the line undergo beta plus decay. Heavy nuclei (one with more than 82 protons) undergo alpha decay. Alpha Decay Alpha radiation consists of alpha particles. These are helium nuclei (without electrons) consisting of 2 protons and 2 neutrons. Beta Decay In beta decay a beta particle (electron) is released from the nucleus. It is called a beta particle to distinguish this electron released from the nucleus, from the electrons orbiting the nucleus. The nucleus is then rearranged and as a result energy is lost through gamma radiation. There are two types of beta decay; Negative beta decay occurs in radioisotopes that have too many neutrons. Positive beta decay occurs in radioisotopes that have too many protons. In beta decay, a neutrino or antineutrino is also released in order to conserve lepton number, charge, and energy. The Weak Nuclear Force Both beta plus and beta minus decay are due to the exchange of virtual particles, analogous to photons, called W bosons. Unlike photons, these exchange particles; Have a non zero rest mass Have a very short range of only 10-18 m Are positively or negatively charged The W- boson is responsible for beta minus decay and the W+ boson for beta positive decay. Rutherford’s Atomic Model Before his experiment, Rutherford predicted that the alpha particles (which he knew to be positively charged) he fired at gold foil would mostly pass straight through with little or no deflection. However, some (about 1 in 10,000) of the alpha particles bounced right back from the gold. What could stop an alpha particle and turn it back in its tracks? The electric force of the protons in the gold nuclei was pushing the positively charged protons in the alpha particles away. This led Rutherford to conclude that atoms have a tiny positive nucleus in the centre with negative electrons orbiting at a much greater distance. Electron Scattering The electric repulsion between nucleus and alpha particle kept Rutherford’s alpha particles at arm’s length from the nucleus. This made the nucleus look just like a point positive charge; a more accurate method of looking into the nucleus is needed. In exactly the same way as alpha particles, electrons undergo Rutherford scattering, except this time the force on the electrons is attractive (positive nucleus, negative electron), and the electrons are not affected by the strong force in the nucleus. However, electrons have quantum properties and in these conditions must be thought of as acting like waves rather than particles. The wavelength of a matter particle is called its de Broglie wavelength which is given by; ℎ 𝐸 𝜆 = 𝑝 and 𝑝 ≃ 𝑐 so 𝜆 ≃ ℎ𝑐 𝐸 The diffraction pattern attained when electrons are fired towards a nucleus looks like this; Since 𝜆 is inversely proportional to E, considerable energy is needed to get a small enough wavelength for the electron diffraction to work. For electrons accelerated to about 300 MeV the wavelength comes to about 4 × 10−15 m, similar to the dimensions of a nucleus. The first minimum on the diffraction pattern occurs where; sin 𝜃 = 1.22𝜆 𝑑 Where 𝜃 = the angle of the minimum and d is the diameter of the nucleus. Inside the Nucleons Each nucleon is made up itself of a combination of three quarks. Two kinds of quark make up the proton and neutron; the up quark and down quark, denoted by u and d respectively. The up quark has a charge of +2/3 and the down quark has -1/3. Up-up-down or uud can be combined to make a proton with charge +1e. Down-down-up or udd can be combined to make a neutron with 0 charge. Each quark also has a corresponding antiquark; they are exactly the same except with an opposite charge. These two quarks and antiquarks can be combined in various combinations to account for a lot of the obscure particles physicists see in particle accelerator collisions. Deep Inelastic Scattering The electron scattering described so far has been elastic. It has consisted simply of electrons approaching atoms and being scattered because of Coulomb forces, the electron doesn’t cause any changes in the nucleus/nucleon. Huge energy particle accelerators were constructed to find out if the nucleons really consisted of the predicted quarks, each quark has its own electric charge and so will interact with any electrons fired towards it. When a very high energy electron collides with a quark, much more than deflection happens. The energy of the collision causes quark-antiquark pairs to materialise (from E=mc2). These particles emerge from the nucleon mainly as mesons, combinations of quarks and antiquarks. By examining the pattern of mesons emerging from the nucleon, we can deduce how charge (and thus how quarks) are arranged inside the nucleon. In baryons, there are three points of deflection, suggesting that baryons consist of three quarks. In mesons there are two points of deflection, suggesting they have two quarks. Scale of Energy To probe structures this small requires huge energies for many reasons. First of all, the particle must overcome Coulomb forces that repel it. The energy needed to get two opposite charges within a distance r of each other is given by; 𝐸𝑘 = 𝑞1 𝑞2 4𝜋𝜀0 𝑟 To get two protons as close to each other as the size of a nucleus requires about 1 MeV. Secondly, the de Broglie wavelength of the particle being fired must be smaller than the structure you want to probe. For small structures like quarks, this wavelength must be tiny which requires large amounts of energy. Thirdly, a lot of energy is needed to create particle-antiparticle pairs, double the rest energy of whatever particle needs to be created. The Colour of Quarks The Pauli Exclusion Principle says that no two particles can have the same quantum state, so how can two of the same quark occupy one nucleon? A new quantum property was needed to differentiate between two of the same quark, this property is called colour. Colour is to the strong interaction as charge is to the electromagnetic interaction. Within a nucleon, the three quarks are assigned three colours; red green or blue. The combination of these colours gives a colourless particle, only colourless particles are allowed. Antiquarks have anticolours; antired, antigreen, antiblue. Mesons can be colourless by having a red and an anti red, cancelling each other’s colour charge out. The idea of colour is supported by the fact that all commonly observed particles made of quarks contain either 3 quarks (one of each colour/anticolour), or two quarks (one colour, one anticolour) which means colour is always conserved. Gluons Quarks attract one another by exchanging force particles called gluons. These gluons are the mediators (or bosons) of the strong interaction, in the same way that the photon is the boson of electromagnetism and the W boson the boson of the weak force. Gluons are the glue that keep protons and neutrons together and lead to the further forces which keep whole nucleons together. Free quarks cannot exist because of the colour property. If two quarks are pulled apart, more and more energy is stored in the gluon field between them until the energy materialises as a quark-antiquark pair. The strong (gluon) interaction gets stronger with distance, contrary to the properties of electromagnetism and gravitation. Within a baryon, the quarks are relatively close so the gluon force is low; they have a certain degree of freedom. This is called quark confinement. As the distance between quarks increases the force becomes immensely strong, trying to separate quarks from each other will just create more quarks. The gluon between two quarks has a combination of their colour charges, such as red and antigreen. The Trapped Electron Electrons are trapped in a ‘potential well’ by the nucleus of an atom, in the same way that we are trapped in earth’s gravitational potential well. When the electron has enough energy to leave the electron its potential is ≥ 0. We can model this well of potential as a one dimensional, infinitely tall box in which the electron is trapped. Because the electron behaves like a wave, and it is trapped in a ‘box’ it forms standing waves across the width of the box. ℎ 𝜆 The standing waves allow only discrete values of wavelength. For each wavelength (from 𝑝 = ) there is a discrete value of momentum. For each wavelength there is a discrete value of kinetic energy; 1 𝑝 = 𝑚𝑣 and 𝐸𝑘 = 𝑚𝑣 2 2 𝑝= 𝑝2 = ℎ 𝑝2 and 𝐸𝑘 = 𝜆 2𝑚 ℎ2 ℎ2 and so, 𝐸 = 𝑘 𝜆2 2𝑚𝜆2 The smaller the box, the narrower the wavelength and so the higher the energy and vice versa, 1 because of 𝐸𝑘 ∝ 𝜆2 . The electrons then in a given atom can only have one of these discrete values of energy, no in between states exist, implied by their discrete wavelengths. These energy levels are the energy levels of electrons in atoms. The energy levels have increased separation as they increase. As n increases, energy is proportional to n2. The lowest energy level where n = 1 is called the ground state (𝐸0 ). Higher energy levels, where n = 2, 3, 4... are called excited states. Energy is always negative, this means that the potential of the electron is never enough to escape the atom, as the energy level increases, the energy approaches 0. Energy Changes and Transitions A trapped electron usually has the lowest allowed energy, it is normally, and wants to be, in the ground state. It can be moved up to an excited state (higher energy level) if it is given enough energy, that is, if it absorbs a photon with enough energy to move it up. The change in energy can be related to the photon needed by the Planck relation 𝐸 = ℎ𝑓. Size of Electron Orbits Equating the electron’s centripetal force with the electrostatic force and using the de Broglie relationship gives us; 𝜆= ℎ 4𝜋𝜀0 𝑟 √ = 33 × 10−11 m 𝑒 𝑚 Spectral Lines The spectral lines from a source can be used to determine the energy levels of the nucleus being used. The photon energy emitted can be worked out by; 𝐸 = 13.6 ( 1 1 2 − 2 ) eV 𝑛2 𝑛1 ...in a hydrogen atom. The Standard Model Matter is ultimately made of two different types of fermions; the electron (a lepton) and the up and down quarks. These two particles, combined with the neutrino (another lepton) explain almost all of the visible matter in the universe. These particles interact by exchanging bosons. Together these four particles make up the first generation of matter. This pattern of four fermions is repeated twice more, each at higher energies and higher masses, giving rise to 3 generations of matter. Most of what happens around us is due to the electromagnetic interaction which is mediated by the exchange of virtual photons. The strong force requires gluons to be exchanged between quarks, 8 different combinations of colour charge gluons are needed. The electromagnetic interaction has shown to be a weaker form of the strong and weak nuclear forces. Gravity is yet to be unified with the one (three) other force(s).