Electronic Supplementary Material, Padilla et al.
advertisement

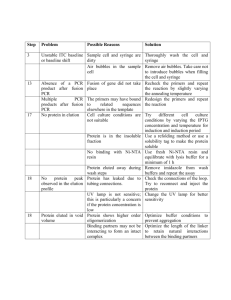
Electronic Supplementary Material ESM Tables S1-S3 Reference genome sequencing, paternity assignment methods and putative sire haplogroup confirmation figure S1 Padua, Zeh, Bonilla and Zeh “Sisters’ curse: sexually antagonistic effects constrain the spread of a mitochondrial haplogroup superior in sperm competition” Table S1. Primers and PCR conditions for next generation sequencing of the Cordylochernes scorpioides mitochondrial genome. Long PCR was used to amplify the genome in three adjacently overlapping fragments. Position in Ahaplogroup Amplicon genome length (bp) 1,405-1,432 9,011 10,443-10,416 Mitochondrial region amplified COX1CYTB Fragment 1 Primer names CscCOX1F1F CscCYTBF1R Primer sequence CTATAATAATTGGGGGTTTTGGDAACTG CCCCTCCTATTTTGTTAGGGACACAACG CYTBrRNA Fragment 2 CscCYTBF2F Cscl-rRNAF2R TRCTGACAATAAATTACTGCCCATCYCC AGAGAAAAATTACGCCAGGGATAACAGG 9,721-9,748 14,093-14,120 4,399 ND1COX1 Fragment 3 CscND1F3F CscCOX1F3R TTGCTGAGGTTAAAGAGTTTACAACTGG GATAAATTGTTCATCCAGCTCCACAYC 13,085-13,112 1,552-1,578 4,434 PCR was performed in a 35 µL reaction volume containing ~10 ng of genomic DNA, 3.5 µL of 10X Advantage Genomic LA Buffer (25 mM MgCl2), 350 mM dNTPs, 750 nM primers, and 1.5U of Advantage Genomic LA Polymerase Mix (Clontech, Mountain View, CA, USA). The PCR amplification conditions involved a 1 min hot start at 95°C, followed by 34 cycles of the following conditions: 93°C melting for 25 s, 54 to 620C melting for 40 s, and a 68°C extension for 10-12 min. Table S2. Mean number of nucleotide substitutions and mean uncorrected divergence within and between the A, B1 and B2 haplogroups of the Cordylochernes scorpioides mitochondrial genome. Comparisons were restricted to the 13 protein-coding genes of the oxidative phosphorylation pathway, the 22 tRNAs, and the large and small subunit ribosomal RNA genes. Total genome length, excluding the control region, varied from 13,704 to 13,736 bp across the 15 sequenced genomes. mean number of nucleotide mean uncorrected comparison substitutions divergence (%) Within A haplogroup 33 0.20 Within B1 haplogroup 47 0.29 Within B2 haplogroup 11 0.04 A versus B1 haplogroup 915 8.36 A versus B2 haplogroup 949 8.89 B1 versus B2 haplogroup 495 4.06 2 Table S3. Sequence lengths and nucleotide substitution properties of the 13 protein-coding genes of the Cordylochernes scorpioides mitochondrial oxidative phosphorylation system. Genes are listed in the order in which they are arranged in the genome. pi(-), pi(N) and pi(+) are the proportion of sites under negative selection, neutrality and positive selection, respectively. proportion of sites under number or percentage of negative selection, sequence length by codons with nonneutrality or positive haplogroup (bp) synonymous substitutions selection gene ND2 COX1 COX2 ATP8 ATP6 COX3 ND3 ND5 ND4 ND4L ND6 CYTB ND1 A B1 930 1536 705 183 672 807 345 1662 1356 270 465 1098 885 894 1536 705 183 675 807 345 1662 1257 270 465 1101 885 B2 894 1536 705 183 675 807 345 1662 1287 270 465 1101 885 number percentage 19 6.44 10 1.97 4 1.72 9 15.00 14 6.28 9 3.37 20 17.70 46 8.35 15 3.69 9 10.23 13 8.50 20 5.49 18 6.32 3 pi(-) 0.933 0.971 0.932 0.424 0.936 0.952 0.791 0.904 0.931 0.461 0.883 0.941 0.753 pi(N) 0.045 0.020 0.032 0.334 0.015 0.025 0.118 0.049 0.033 0.257 0.059 0.032 0.157 pi(+) 0.020 0.009 0.037 0.241 0.048 0.022 0.091 0.046 0.036 0.282 0.059 0.027 0.090 Reference genome sequencing, paternity assignment methods and putative sire haplogroup confirmation Sequencing of the reference C. scorpioides mitochondrial genome An entire C. scorpioides mitochondrial genome was PCR amplified in two segments, using the Expand Long Template PCR kit (Roche). High-Tm, C. scorpioides-specific primers were designed by first amplifying an ~ 660-bp segment of the COX1 gene, using the highly conserved chelicerate forward1 (5'-TACTCTACTAATCATAAAGACATTGG – 3’) and reverse2 (5’ – GGATGGCCAAAAAATCAAAATAAATG – 3’) primers [1], and an ~ 450 bp region of the CYTB gene Cytb424-449 (5’ – GGWTAYGTWYTWCCWTGRGGWCARAT – 3’ and Cytb876-847 (5’ – GCRTAWGCRAAWARRAARTAYCAYTCWGG – 3’) [2]. COXI and CYTB are located approximately opposite one another in the circular mitochondrial genome, and primers from these two genes can be used to amplify the entire genome in two fragments of similar length. In order to span the entire genome, 35 primer pairs were designed, with ~ 100-bp overlap between contiguous segments TreeSAAP Methods For each gene, the phylogeny of the 15 sequences was reconstructed using Bayesian methods and imported into TreeSAAP, which was then used to estimate the magnitude of change across the entire gene sequence for 31 physicochemical properties. TreeSAAP rates substitutions on a scale of 1 (most conservative) to 8 (most radical), with a significant positive z-score indicating more non-synonymous substitutions than expected under neutrality. Following author recommendations, only the most radical amino acid substitutions (categories 7-8), with z-scores 4 statistically significant at the 0.001 level, were considered biologically significant and further analyzed, using a sliding window of 15 codons to pinpoint specific gene regions under positive selection. Haplogroup effects on sperm competitive ability DNA was extracted from the dams, putative sires and protonymphs using Invitrogen Chargeswitch gDNA Micro Tissue Kit (Life Technologies, Grand Island, NY, USA). PCR was performed in a 30 µL reaction volume containing ~10 ng of genomic DNA, 3 µL of 10X Advantage Genomic LA Buffer (25 mM MgCl2), 350 mM dNTPs, 750 nM primers, and 1.5U of Advantage Genomic LA Polymerase Mix (Clontech, Mountain View, CA, USA). The PCR amplification conditions involved a 1 min hot start at 95°C, followed by 34 cycles of the following conditions: 93°C melting for 25 s, and 68°C annealing/extension for 8 min For each replication, PCR products from the mother, the two putative sires and an average of 22 offspring were run on a 1.5% agarose gel and stained with ethidium bromide to visualize alleles. Several measures were taken to ensure amplification of both alleles from heterozygous individuals. First, a strict consensus primer pair (cCscMS23 2+: CAGTGTTGCCGGGTGGATTATTAACC and cCscMS23 2-: ATGGCGTGKGAAGCAGAGAACTCTAGG) was previously designed from a large sample (N=22) of cCscMS23 alleles. Second, the high primer-pair Tm enabled a robust, two-step PCR reaction. Finally, PCR was performed using a mixture of DNA polymerases capable of amplifying high molecular weight alleles (to 22 kb) from femptogram quantities of DNA. 5 Putative sire haplogroup confirmation in sperm competition experiment DNA sequencing of the mitochondrial ND2 locus from our C. scorpioides laboratory matrilines has established that haplotypes in the A but not the B2 haplogroup possess a ClaI restriction site. ClaI digested ND2 amplicons were therefore used to confirm the mitochondrial haplotype of all putative sires from successful replicates of the sperm competition experiment. DNA from frozen adults was extracted as described above and PCR was conducted using the ND2 mitochondrial DNA locus forward (5’ – TGTAAGTCTTAAAAYAAAGAAAACC – 3’) and reverse primers (5’ – AAGTCATCGAATAGARACRTTAGC – 3’). PCR reactions were performed, as described above, except that the conditions of the 34 cycles were modified to: 93°C for 25 s, 48C for 50 s, and 68C for 90 s. Digestion reactions were carried out using 20 μL mixtures of 1X buffer, 1 μg (approximately 12 μL) of PCR product and 1μL of Cla I restriction enzyme. Products were incubated for 60 min at 37C and the enzyme inactivated at 65C for 20 min. Products were visualized on 1.5% agarose gels run for approximately 80 volt-hr to determine presence (two fragments) or absence (one fragment) of the restriction site in the ND2 sequence from each male. References 1. Barrett, R. D. H. & Hebert, P. D. N. Identifying spiders through DNA barcodes. Can. J. Zool. 83, 481-491 (2005). 2. Cook, C. E. The complete mitochondrial genome of the stomatopod crustacean Squilla mantis. BMC Genomics 6, 105 (2005). 6 figure S1. Female mitochondrial haplotype and sexual receptivity at first and second mating. Ahaplogroup females (striped bars) and B2-haplogroup females (black bars) were equally likely to accept a sperm packet in their first mating. However, the propensity of B2-haplogroup females to engage in polyandry was significantly lower (*P = 0.05) than that of A-haplogroup females. 7