GM Glasshouse - University Wiki
advertisement
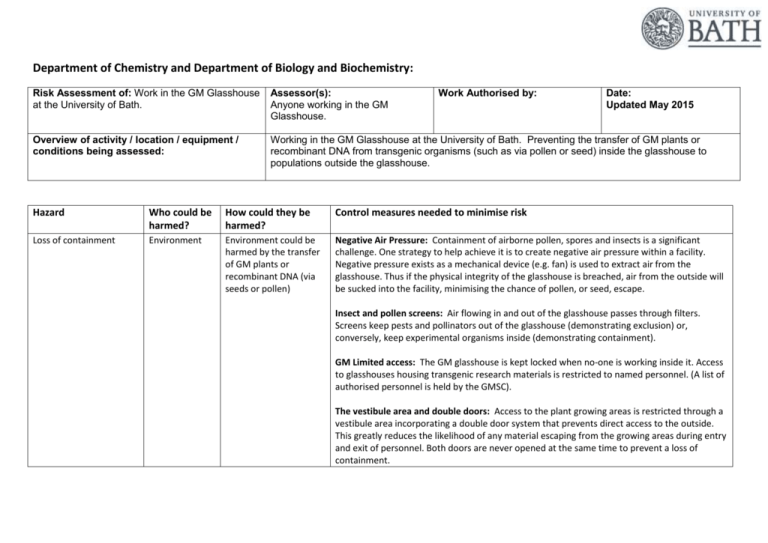
Department of Chemistry and Department of Biology and Biochemistry: Risk Assessment of: Work in the GM Glasshouse at the University of Bath. Assessor(s): Anyone working in the GM Glasshouse. Work Authorised by: Date: Updated May 2015 Overview of activity / location / equipment / conditions being assessed: Working in the GM Glasshouse at the University of Bath. Preventing the transfer of GM plants or recombinant DNA from transgenic organisms (such as via pollen or seed) inside the glasshouse to populations outside the glasshouse. Hazard Who could be harmed? How could they be harmed? Control measures needed to minimise risk Loss of containment Environment Environment could be harmed by the transfer of GM plants or recombinant DNA (via seeds or pollen) Negative Air Pressure: Containment of airborne pollen, spores and insects is a significant challenge. One strategy to help achieve it is to create negative air pressure within a facility. Negative pressure exists as a mechanical device (e.g. fan) is used to extract air from the glasshouse. Thus if the physical integrity of the glasshouse is breached, air from the outside will be sucked into the facility, minimising the chance of pollen, or seed, escape. Insect and pollen screens: Air flowing in and out of the glasshouse passes through filters. Screens keep pests and pollinators out of the glasshouse (demonstrating exclusion) or, conversely, keep experimental organisms inside (demonstrating containment). GM Limited access: The GM glasshouse is kept locked when no-one is working inside it. Access to glasshouses housing transgenic research materials is restricted to named personnel. (A list of authorised personnel is held by the GMSC). The vestibule area and double doors: Access to the plant growing areas is restricted through a vestibule area incorporating a double door system that prevents direct access to the outside. This greatly reduces the likelihood of any material escaping from the growing areas during entry and exit of personnel. Both doors are never opened at the same time to prevent a loss of containment. Sticky mats: A sticky mat (TakMat) inside the glasshouse at the outer doorway of the vestibule is deigned to trap any material such as pollen, seeds, etc adhering to foot-ware. The topmost layer of the mat is regularly removed and disposed as contaminated waste. Protective clothing: Within the vestibule are designated lab coats. These must be worn at all times by staff in the facility. These should be taken off and left in the vestibule before leaving the facility. This will minimize the chance of spread of GM material. Labelling: All GM plant material is labelled. Where known, end dates are recorded on plant trays and also on the external doors of the temperature controlled rooms. Cleanliness: All areas of the glasshouse should be cleaned regularly and maintained in a tidy condition. Hygiene facilities such as a washbasin are available and must be made use of by users of the facility. All re-usable materials such as pots, trays etc should be disinfected by a validated method after each use. Deep cleaning should take place at the end of experimental work. Collection and treatment of drainage water: All liquid effluent is collected into a trap and disinfected by a validated method prior to discharge to the drains. Secure transport of plants and materials between GM Glasshouse, temperature controlled rooms and Laboratory: Plants and materials containing transgenes being transported between the GM glasshouse, temperature controlled rooms or laboratory areas must be in double sealed containers with lids that minimize the risk of escape. Waste materials: All waste plant material, growth media, floor sweepings, TakMat layers and disposable materials should be segregated into clearly marked bags and treated by a validated means before disposal to kill any residual organisms and destroy any rDNA. Where possible waste materials should be autoclaved. The surrounding area and contingency plan for dealing with escapes: A programme of regular ground maintenance in the area immediately surrounding the glasshouse is employed to ensure that cross-fertile plants are not within the pollen dispersal range of the experimental plant. On a two-weekly basis the area surrounding the glasshouse will be checked for weedy plants that may include potential escapees. The area is grassed and any non-grass plants (except trees) should be up-rooted and deposited in an autoclave bag for disposal. Minimizing the amount of pollen and seed: Pollen and seed represent the most significant risk to the environment from transgenic plants within the GM glasshouse. This risk is minimized by preventing flowering or seed set where possible; otherwise, the number of flowers or seed pods are kept to a minimum consistent with the demands of the experiment Seed storage: Seeds are not to be stored in the GM glasshouse apart from in the designated fridge in the vestibule area. Seeds should be sown in the vestibule area and any excess returned to secure storage within the fridge, laboratories or temperature controlled rooms. Transfer of transgenic plants between glasshouse and lab: Closed, double-sealed containers are available for secure transport of plants. These should be used irrespective of the life-cycle stage of the plants. Take care to avoid contaminating the exterior surface of the containers with pollen or seeds. Residual risk rating: (3) Moderate x (2) Unlikely = (6) LOW Untrained staff in facility Environment Environment could be harmed by the transfer of GM plants or recombinant DNA (via seeds or pollen) Providing information, training and supervision: Before entering the glasshouse, all staff working around transgenic organisms should be fully informed by the project supervisor about the containment measures applicable to a given research project. Prescribed procedures and practices should be appropriate for the assigned Containment level; those that appear excessive for the needed level of containment may discourage compliance. Relevant GM project risk assessments (and the Local Rules): These must be read by all those involved and signatures obtained from them. Appropriate supervision of workers must be provided. Induction material: No staff are allowed to start work until they have been given an induction. The induction includes instruction to all staff to register all GMO work with the GM committee, that they cannot use the glasshouse facilities without receiving training first and that all work must be Risk Assessed. Recently all staff were sent out an induction summary to remind them of the induction details and to send any new staff to the Technical Team. Disposal: Re-usable pots must be disinfected by immersing in a solution of bleach (1,000ppm available chlorine) for at least 8 hours (usually overnight). (1,000ppm Cl solution can be made by diluting commercial bleach [nominal 5% Cl] by a factor of 1 in 50.) All plant material, compost, floor sweepings, TakMat layers and disposable plastic ware, such as pots, should be placed in the autoclave bags provided in the blue skips. These bags (and skips) will be removed periodically for sterilization by autoclaving (minimum of 126oC for 30 minutes) in Building 4S. The floor (including under the floor drain covers) should be cleaned with a vacuum cleaner every week. Material from the vacuum cleaner bag must be disposed of in the autoclave bag. Residual risk rating: (3) Moderate x (2) Unlikely = (6) LOW Unauthorised access to facility Environment Environment could be harmed by the transfer of GM plants or recombinant DNA (via seeds or pollen) Signs are on door inducting that access is restricted. Access to glasshouses housing transgenic research materials is restricted. Only personnel approved by the GMSC should enter the GM glasshouse. A list of approved personnel is available from the UBSO and the secretary to the GMSC. Visitors should be accompanied by an approved person who should explain the operation of the facility. The outer and inner doors should be locked when personnel are not present in the facility. The keys to the glasshouse must be kept in a secure place. The inner door should be closed when workers are in the growing area. The vestibule area is an important part of the containment strategy. Before entering the vestibule from outside it is therefore very important to ensure that the inner door is closed. Check before entering and if the inner door is open communicate with the person(s) working inside to close the door before you enter. Make sure you close and lock all doors if you are last to leave the facility. There is CCTV covering the pathway alongside 4S and the glasshouses, and the path up towards the pond and Parade, and recordings are kept for 14 days before being overwritten. Security also regularly patrol the area during the evening and night. Residual risk rating: (3) Moderate x (2) Unlikely = (6) LOW GM plants/ plant material on glasshouse users Glasshouse users Consumption by humans/ contact with humans. Slips and Trips Glasshouse users Slipping on floors/ tripping on hazards Chemicals Glasshouse users Hazardous chemicals affecting human health All work must be risk assessed and the nature of the plant species being used will be detailed in this. All users must read, understand and sign a Risk Assessment for their work before starting. Any risks to human health will be noted in this Risk Assessment and the methods put in place to reduce risk will be noted and followed. Floors are regularly cleaned with algal-biocide to prevent them becoming slippery. Good house-keeping means that clutter is removed. A glasshouse technician is employed to ensure best practice. All hazardous chemicals to be stored in a chemicals cabinet. Instructions on cleaning products containing chemicals should be followed. Electrical Glasshouse users Electrical shock Biological Glasshouse users Possible infection. Manual Handling Glasshouse users Glasshouse users Injury to glasshouse users. Temperature in hot weather may cause discomfort to users. Environment All glasshouse electrical equipment should be PAT tested. Care should be taken to keep electrical items away from any water sources. Do a visual inspection before use – if any damage or defects are seen – do not use until rectified. Ensure good housekeeping and good hygiene to decrease the risk of microorganisms on the skin. No eating or drinking is allowed in any of the glasshouses. Hand-washing facilities are available. The University Manual Handling guidelines should be followed. Users should make sure to take regular breaks outside in the fresh air and keep hydrated. Risk Assessment Guidance The assessor can assign values for the hazard severity (a) and likelihood of occurrence (b) (taking into account the frequency and duration of exposure) on a scale of 1 to 5, then multiply them together to give the rating band: Hazard Severity (a) Likelihood of Occurrence (b) Moderate Serious Fatal (eg discomfort, slight bruising, self-help recovery) (eg small cut, abrasion, basic first aid need) (eg strain, sprain, incapacitation > 3 days) (eg fracture, hospitalisation >24 hrs, incapacitation >4 weeks) (single or multiple) Minor Trivial 1 – Trivial 2 – Minor 3 – Moderate 4 – Serious 5 – Fatal 2 3 4 5 1 – Remote 2 – Unlikely 3 – Possible 4 – Likely 5 – Very likely (almost never) (occurs rarely) (could occur, but uncommon) (recurrent but not frequent) (occurs frequently) The risk rating (high, medium or low) indicates the level of response required to be taken when designing the action plan. Rating Bands (a x b) Remote 1 2 4 6 8 10 3 6 9 12 15 Likely 4 8 12 16 20 Very likely 5 10 15 20 25 Unlikely Possible LOW RISK (1 – 8) MEDIUM RISK (9 - 12) HIGH RISK (15 - 25) Continue, but review periodically to ensure controls remain effective Continue, but implement additional reasonably practicable controls where possible and monitor regularly -STOP THE ACTIVITYIdentify new controls. Activity must not proceed until risks are reduced to a low or medium level