Grading procedures
advertisement
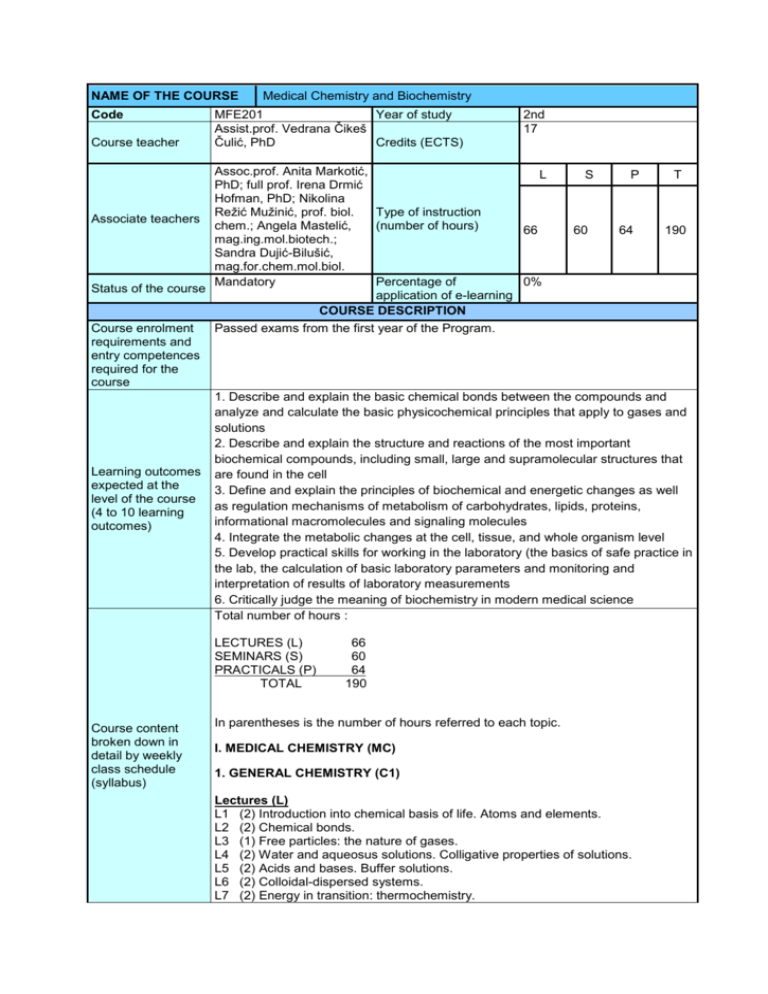
NAME OF THE COURSE Code Course teacher Medical Chemistry and Biochemistry MFE201 Year of study Assist.prof. Vedrana Čikeš Čulić, PhD Credits (ECTS) 2nd 17 Assoc.prof. Anita Markotić, L S P T PhD; full prof. Irena Drmić Hofman, PhD; Nikolina Režić Mužinić, prof. biol. Type of instruction Associate teachers chem.; Angela Mastelić, (number of hours) 66 60 64 190 mag.ing.mol.biotech.; Sandra Dujić-Bilušić, mag.for.chem.mol.biol. Mandatory Percentage of 0% Status of the course application of e-learning COURSE DESCRIPTION Course enrolment Passed exams from the first year of the Program. requirements and entry competences required for the course 1. Describe and explain the basic chemical bonds between the compounds and analyze and calculate the basic physicochemical principles that apply to gases and solutions 2. Describe and explain the structure and reactions of the most important biochemical compounds, including small, large and supramolecular structures that Learning outcomes are found in the cell expected at the 3. Define and explain the principles of biochemical and energetic changes as well level of the course as regulation mechanisms of metabolism of carbohydrates, lipids, proteins, (4 to 10 learning informational macromolecules and signaling molecules outcomes) 4. Integrate the metabolic changes at the cell, tissue, and whole organism level 5. Develop practical skills for working in the laboratory (the basics of safe practice in the lab, the calculation of basic laboratory parameters and monitoring and interpretation of results of laboratory measurements 6. Critically judge the meaning of biochemistry in modern medical science Total number of hours : LECTURES (L) SEMINARS (S) PRACTICALS (P) TOTAL Course content broken down in detail by weekly class schedule (syllabus) 66 60 64 190 In parentheses is the number of hours referred to each topic. I. MEDICAL CHEMISTRY (MC) 1. GENERAL CHEMISTRY (C1) Lectures (L) L1 (2) Introduction into chemical basis of life. Atoms and elements. L2 (2) Chemical bonds. L3 (1) Free particles: the nature of gases. L4 (2) Water and aqueosus solutions. Colligative properties of solutions. L5 (2) Acids and bases. Buffer solutions. L6 (2) Colloidal-dispersed systems. L7 (2) Energy in transition: thermochemistry. L8 (1) Reactions at equilibrium. L9 (1) The rate of chemical change. L10 (1) The natural direction of change: the second law. L11 (1) Chemical energy and electrical energy: electrochemistry. Seminar practicals (SP) and practicals (P) SP1+P1 (1+3) Basic stoichiometry. Preparation of solutions. SP2+ P2 (1+3) Optical methods in medical chemistry. SP3+ P3 (1+3) Gas laws. Ions in solution. Osmotic pressure. SP4+ P4 (1+3) Volumetry: neutralization methods. SP5+ P5 (1+3) Volumetry: oxidation and reduction method. SP6+ P6 (1+3) Acids and alkalis, pH and buffers. SP7+ P7 (1+3) Energetics and kinetics of chemical reaction. 2. ORGANIC CHEMISTRY (C2) Lectures (L) L12 (2) Introduction to organic chemistry. Saturated and unsaturated hydrocarbons; physical and chemical properties. Isomers. L13 (2) Halogenalkanes; nucleophilic substitution, elimination. Optical isomerism; relative and absolute configuration. L14 (1) Alcohols. Ethers. Aldehydes. Ketones. L15 (2) Carboxylic acids and their derivates. L16 (2) Cyclic and aromatic hydrocarbons. Sulfuric and heterocyclic compounds. L17 (1) Amino acids. Carbohydrates. Seminars organic chemistry (SO) SO1 (3) Resonant structures. Isomers. Alkanes, alkenes, alkynes. Alkaloids, phenols, esters, aldehydes. Ketones SO2 (3) Amines. Sulfur heterocyclic compounds. Amino Acids. Carbohydrates. Carboxyl compounds Seminar practicals (SP) and practicals (P) SP8+ P8 (1+3) Qualitative analysis of some organic compounds SP9+P9 (1+3) Potentiometric titration of amino acids II. MEDICAL BIOCHEMISTRY (MB) Lectures (L) and seminars biochemistry (SB) BIOCHEMISTRY (B1) 1. STRUCTURE AND FUNCTION OF PROTEINS AND ENZYMES L18 (2) Amino acids and peptides. Proteins: determination of primary structure. L19 (2) Proteins: higher orders of structure. Proteins: myoglobin and hemoglobin; collagen. SB19 (1) Sickle cell anemia. Scurvy. L20 (2) Enzymes: mechanism of action, kinetics, regulation of activities. SB20 (2) Izoenzymes. Enzymes in medicine. 2. BIOENERGETICS AND METABOLISM OF CARBOHYDRATES AND LIPIDS L21 (2) Bioenergetics: the role of ATP. Biologic oxidation. The respiratory chain and oxidative phosphorylation. SB21 (2) Regulation of respiratory chain. L22 (1) The citric acid cycle: the catabolism of acetyl-CoA. SB22 (1) Pyruvate dehydrogenase deficiency. L23 (1) Glycolysis and the oxidation of pyruvate. SB23 (1) Glycolysis regulation. L24 (1) Metabolism of glycogen. SB24 (1) Regulation of glycogenesis and glycogenolysis. L25 (1) Gluconeogenesis. SB25 (1) Control of blood glucose. L26 (1) The pentose phosphate pathway. SB26 (1) Other pathways of hexose metabolism. L27 (2) Oxidation of fatty acids: ketogenesis. SB27 (1) Metabolic ketoacidosis. L28 (1) Biosynthesis of fatty acids and eicosanoids. SB28 (1) Lipid transport and storage. L29 (1) Cholesterol synthesis, transport and excretion. SB29 (1) Hypercholesterolemia and synthesis of bile salts and vitamin D. BIOCHEMISTRY (B2) 3. METABOLISM OF PROTEINS AND AMINO ACIDS L30 (1) Biosynthesis of the nutritionally nonessential amino acids. L31 (2) Catabolism of proteins and of amino acid nitrogen. Catabolism of the carbon skeletons of amino acids. SB31 (1) Regulation of amino acid and protein catabolism. L32 (2) Conversion of amino acids to specialized products. Porphyrins and bile pigments. SB32 (1) Porphyrine metabolism disorders. 4. STRUCTURE, FUNCTION AND REPLICATION OF INFORMATIONAL MACROMOLECULES L33 (2) Nucleotides. Metabolism of purine and pyrimidine nucleotides. SB33 (1) Regulation of purine and pyrimidine metabolism. L34 (1) DNA organization, replication and repair. SB34 (2) DNA mutations and chemotherapy. L35 (1) RNA synthesis, processing and modification. Protein synthesis and the genetic code. SB35 (1) Protein synthesis regulation and inhibition. L36 (1) Regulation of gene expression. SB36 (1) Gene expression regulation. L37 (2) Molecular genetics, recombinant DNA and genomic technology. 5. BIOCHEMISTRY OF EXTRACELLULAR AND INTRACELLULAR COMMUNICATION L38 (2) Membranes: structure, assembly and function. SB38 (1) Protein conformational changes in membrane. L39 (2) The diversity of the endocrine system. SB39 (1) Hormone synthesis disorders. L40 (2) Hormone action and signal transduction. SB40 (1) Signal transduction disorders. 6. SPECIAL TOPICS L41 (2) Nutrition, digestion and absorption. SB41 (1) Micronutrients: vitamins and minerals. L42 (2) Integration of metabolism. SB42 (1) Integration of tissue metabolism. Special topics. Additional seminars SB43 (2) Biochemistry of extracellular matrix. Plasma proteins and immunoglobulins. SB44 (2) Hemostasis and thrombosis. SB45 (2) Metabolisam of xenobiotics. Seminar practicals (SP) and practicals (P) SP10+ P10 (1+3) SP11+ P11(1+2) SP12+ P12(1+2) SP13+ P13(1+3) Serum proteins electrophoresis Urease: determination of inhibitor Alkaline phosphatase: effect of pH on enzyme activity Alkaline phosphatase: determination of Km and Vmax in the SP14+ P14(1+2) SP15+ P15(1+3) SV16+ P16(1+2) SP17+ P17(1+2) SP18+ P18(1+2) SP19+ P19(1+3) SP20+ P20(1+2) SP21+ P21(1+3) SP22+ P22(1+2) SP23+ P23(1+2) P24 (4) Format of instruction Student responsibilities Screening student work (name the proportion of ECTS credits for each activity so that the total number of ECTS credits is equal to the ECTS value of the course) presence of inhibitors Amylase: determination in saliva sample Determination of HbA1c by ion-exchange chromatography Lipids: separation of skin lipids by thin-layer chromatography Determination of lipoproteins Determination of conjugated and total bilirubin in serum Determination of creatinine and the pathological compounds in urine Determination of iron and iron binding capacity in serum Immmunochemical analysis. ELISA. Determination of vitamin C Hemostasis- clotting time and bleeding time tests Comprehensive final exam (laboratory practicals) ☒ lectures ☒ seminars and workshops ☒ exercises ☐ on line in entirety ☐ partial e-learning ☐ field work ☐ independent assignments ☐ multimedia ☒ laboratory ☐ work with mentor ☒ consultations In accordance to Rules of studying and Deontological code for USSM students. Class attendance Experimental work 2 Essay Research Practical training 2 Report (Other) Seminar essay (Other) Tests 6 Oral exam (Other) Written exam 7 Project (Other) Grading is based on an absolute scale, with a (minimum) course total of 90 points. Scores in four partial written exams (C1, C2, B1, B2) and a comprehensive final exam (practicals) are the principal means of accumulating points. Students who attended lectures and practicals could write partial written exams, while final exam from practicals is prerequisite for attending B2. Points can be earned in outside of class activities as well at instructor discretion. Instructor has the right to deduct points for missed exams, late work, disruptive classroom behaviour, or lack of effort or participation. Grades are assigned according to percentage of possible points earned. Grading and evaluating student Grading procedures work in class and at Total accumulated points will be comprised of the following: the final exam pass Physical chemistry (C1) 40 23 Organic chemistry (C2) 15 9 Biochemistry 1 (B1) 50 28 Biochemistry 2 (B2) 40 24 Final exam-practicals 15 9 160 93 Total number of points possible Final grade in this course will be determined by calculation of mean grade of the following: 1) C1+C2+ final exam-practicals (max 70 points) 2) B1+B2 (max 90 points) Point interval of grade 1) 41-48 sufficient (2) 49-56 good (3) 57-63 very good (4) 64-70 excellent (5) Point interval of grade 2) 52-61 sufficient (2) 62-71 good (3) 72-81 very good (4) 82-90 excellent (5) Title Required literature (available in the library and via other media) 1. Murray RK, Bender DA, Boatham KM, Rodwell VW, Weil PA: Harpers’ Illustrated Biochemistry, 29th edition. McGraw Hill, 2012. 1. Atkins PW, de Paula J. Physical Chemistry, 10th edition. Macmillian Education, Oxford, 2014. 2. McMurry JE. Fundamentals of Organic Chemistry, 7th edition. Cornell University, 2011. 3. Lieberman M, Marks AD. Mark's Basic Medical Biochemistry-a Clinical Approach, 4th edition. Lippincott Williams & Wilkins, 2013. Optional literature (at the time of submission of study programme proposal) Quality assurance methods that ensure the acquisition of exit competences Other (as the proposer wishes to add) Number of Availability via copies in other media the library 2 Teaching quality analysis by students and teachers Exam passing rate analysis Committee for control of teaching reports External evaluation