Supplementary Information Stream Water Collection and Processing
advertisement

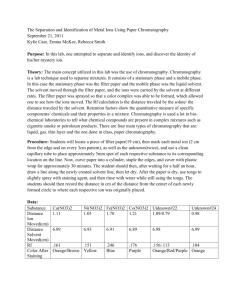
Supplementary Information Stream Water Collection and Chemistry? Water samples were taken using triple rinsed 500ml high-density polyethylene (HDPE). Samples were filtered using 0.45 μm glass fiber filters within 24 hours. Samples were stored on ice in the field until transported back to the lab where they were refrigerated at 4oC until analysis. Ion chromatography was used to analyze the samples for fluoride [F-], chloride [Cl-], nitrate [NO3-], and sulfate [SO4-2], with detection limits of 0.2 mg/l, 0.25 mg/l, 0.25 mg/l, and 2.5 mg/l, respectively. Constituents were measured in compliance with the U.S. Environmental Protection Agency Method #0300.1, which provides the procedure for determining inorganic ions in drinking water by ion chromatography (USEPA 1997). Soil Collection and Processing Soil samples were tested for nitrates using established methodology by Griffin et al. (1995). This method relies on the assumption that nitrate in soils with low anion exchange capacity is water soluble and can therefore be extracted from the soils. Nitrate is extracted from the soils and measured by the same method as the water samples. Each sample was sieved (2 mm) and oven-dried at low heat (60oC) overnight. 5 g of the sample was then placed in a 125 ml Erlenmeyer flask and combined with 50 ml of a prepared extractant, 0.01 M Calcium Sulfate (CaSO4) in this case. Calcium Sulfate was used due to its compatibility with ion chromatography and its ionic strength, resulting in a clear filtrate. The solution was placed on a reciprocating shaker at 200 oscillations per minute for 15 minutes. Medium grade filter paper (Whatman No. 2) was used to filter the soil suspension without contributing measurable amounts of nitrate. Ion chromatography was used to analyze the resultant filtrate.