Exp. 5
advertisement

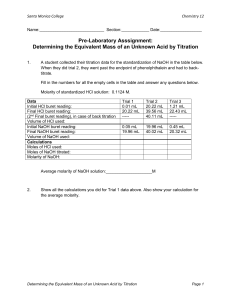
Exp. 5: Determination of the Formula of a Compound Required Locker Materials: - 50 ml Erleneyer flask 100ml volumetric flask 200ml beaker (2) 250ml Erlenmeyer Eye dropper Safety: - Eyewear MUST be worn at all times Waste: - This Lab produces a lot of liquid waste. Make sure to dispose all waste in the waste containers. Introduction Salts dissolved in aqueous solution always behave like strong electrolytes, meaning salts when dissolved always dissociate into hydrated positive and negative ions. For example calciumchloride dissolved in water will dissociate into one calcium and two chloride ions. In the following experiment you will work with two salts and both will dissociate according to the following equations: 1 2 CuCl2(s) Cu2+(aq) + 2 Cl-(aq) NaOH(s) Na+(aq) + OH-(aq) Metal cations such as copper(II) can form complex compounds with certain anions. In the following experiment you will investigate and determine the formula of a complex compound of copper and hydroxide ions according to the following equation: x Cu2+ + y OH- Cux(OH)y(2x-y) Experimental Objective: The objective of the lab is to determine the formula of a copper complex compound correctly through a titration. Students need to learn to measure the volumes of liquids correctly by using different measuring tools such as burets and pipets. In addition students will need to apply mol-calculations to determine the correct outcome. Procedure For this experiment record all observations and measurements in your notebook: Preparation of copper solution The copper solution is prepared only once and is used for all following titrations! 1. Determine what type of copper salt is available for the titration. Write down the correct formula in your notebook and calculate the formula mass of the compound. 2. In your notebook show the calculation of how you will prepare a 100.0ml 0.100M Cu 2+ solution. Before you move on to step 3 show your calculation to the instructor! 3. In a 50ml beaker weigh out approximately the amount of Cu(II)salt calculated in step 2. Write down your exact mass. 4. Carefully transfer the salt to 100ml volumetric flask. With a squirt bottle fill the volumetric flask half with DI water. Swirl the volumetric flask until all salt is dissolved. Fill the flaks to the mark carefully with DI water; the last drops should be added with an eye dropper. Record the measured volume of you solution in your notebook. 5. Transfer the well mixed solution from the volumetric flask to a 250ml Erlenmeyer. Exp. 5: Page 1 of 3 Leo Truttmann – Fall 13 Titration of copper solution 6. On the Chemical Bench each student (or group) takes 150ml of a standard NaOH solution in a clean and dry beaker. Record the molarity of the NaOH solution in your notebook. 7. Organize a buret from the equipment bench and set it up with a buret stand. Rinse it a few times with 5 to 10 mL portions of the standard NaOH solution and fill the buret. (Don't forget to rinse out the tip.) 8. Rinse a 25 mL pipet two or three times with dilute ammonia cleaning solution, then with a few portions of distilled water; a squirt bottle is of use in this operation. Finally, rinse the pipet a few times with 3-5 mL portions of the prepared Cu2+ solution. 9. To a 250 mL Erlenmeyer flask add about 50 ml of distilled water and 10 drops of phenol red-Nile blue indicator. 10. Pipet about 20 ml of Cu2+ solution into the Erlenmeyer flask, and record the exact volume used (Depending on the type of pipet used, you need to write down initial and final volume). 11. Record the initial buret reading. Titrate the solution in the Erlenmeyer with standard NaOH. Keep the contents of the flask mixed during the titration by swirling the Erlenmeyer constantly. As one approaches the end point-a persistent and homogeneous violet color-the NaOH should be added drop wise in order to avoid over titrating. 12. Record the final buret reading and determine the exact volume of NaOH solution added. 13. Repeat the titration two more times. 14. Clean up and dispose all waste into the liquid container. Rinse buret multiple times with DI water and return it to the equipment bench. Calculations For the calculation start a new section in your notebook. Remember you need to show every calculation you do by writing down the equation and an example calculation. The example calculation only needs to be done for one trial. The results of the remaining trials can be written down in a table in your notebook. Calculation of copper(II) salt concentration Concentration of Cu2+ in prepared solution It is important to calculate the concentration of Cu2+ in the prepared copper solution. In a first step calculate the amount of moles of Cu2+ in the solution and then divide the numbers of moles by the volume given by the volume of the volumetric flask. Titration of copper solution Volume of added Cu-solution If you used a ‘to deliver’ pipet then the volume added will be equal the measured initial volume in the pipette. In case you have a ‘not to deliver’ pipette, you need to calculate the added volume by taking the difference between initial and final volume reading. Moles of Cu2+ (added) This is calculated by using the added volume and the calculated concentration of your copper(II) salt solution. Exp. 5: Page 2 of 3 Leo Truttmann – Fall 13 Volume and moles of NaOH added First you need to calculate the amount of NaOH solution added in the titration and then you calculate how many moles of NaOH were added. Mol ratio The mole ratio between hydroxide and copper ions is calculated by taking the ratio of the corresponding moles in each trial. Average mole ratio and Average Deviation Calculate the Average and Average Deviation of your best three trials. Questions and Discussions After you finished all the calculations write a short answer for each of the following questions and discuss your answer. 1. According to the rules of valence, is this formula electrically neutral? Explain. 2. If not, what species present could provide charge neutrality? 3. What would the formula of the compound be if this species were included? 4. Explain what would happen to the calculated ratio, moles OH- to moles Cu2+, if you carried out the titration with a buret in which drops of the NaOH solution stuck to the sides. Template for Measurements Measurements Trial 1 Formula weigh of Cu-salt [g/mol] mass of Cu-salt (weighed) [g] Volume of prepared Cu-solution [ml] Initial Vol of Cu2+solution (pipet) [ml] Final Vol of Cu2+solution (pipet) [ml] Initial Vol of NaOH solution (buret) [ml] Final Vol of NaOH solution (buret) [ml] Exp. 5: Page 3 of 3 Trial 2 Trial 3 Leo Truttmann – Fall 13