Dietary Considerations Of Protein Quality
advertisement

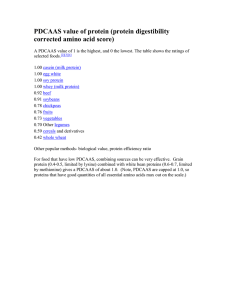
Dietary Considerations of Protein Quality Lynne Kammer, MSE, MA The University of Texas at Austin Protein is a key macronutrient that has direct effects on overall health. Although it is most commonly associated with muscle, it also provides structure for other cells and is involved in biochemical processes. Protein forms include skin, hair, hormones, enzymes, cell receptors, transporters, immune system cells and plasma. Proteins consist of amino acids (AAs) that are either synthesized in the body (non-essential amino acids) or obtained in the diet (essential amino acids, or EAAs) (Table 1). Cysteine and tyrosine are synthesized from methionine and phenylalanine respectively, and will be deficient if these primary EAAs are not consumed. Children cannot synthesize arginine, which must be included in their diet. Availability and rate of appearance of EAAs vary by protein source and must be considered when developing a dietary plan. Most amino acids are digested in the small intestine and processed by the liver, but isoleucine, leucine and valine, the branched-chain amino acids (BCAAs) bypass the liver and are processed by muscle. Table 1 Amino Acids Histidine Isoleucine Leucine Lysine Essential amino acids Methionine Cysteine Phenylalanine Tyrosine Threonine Tryptophan Valine Non-essential amino acids Alanine Glutamine Arginine Glycine Asparagine Proline Aspartic acid Serine Cysteine Tyrosine Glutamic acid Excess EAAs are not stored, but are converted into other amino acids, used for energy, or stored as fat. In the latter two situations, the nitrogen portion of the molecule is excreted from the body, primarily as urea. Biochemical processes that depend upon EAAs will not occur if EAAs are not available; therefore protein must be consumed multiple times during the day. Daily dietary protein requirements depend upon age, activity type and level, weight and energy balance (Table 2). Protein intake should match the metabolic demand, but there may be variation between individuals and even within individuals. The current protein RDA for adults is 0.8 g/kg body weight (Institute of Medicine, 2005) and no additional allowance was made for the needs of exercise. However, this position is in contrast with the views of most sports nutrition researchers. As shown in Table 2, the general consensus is that athletes require up to double the RDA and sometimes more still if energy and/or carbohydrate intake is restricted. 1 Table 2 Daily protein requirements1 1 Population Institute of Medicine RDA for Protein (g/kg body weight/day) Infants to 1 yr 1.5 Children age 1-3 yr 1.1 Children age 4-13 yr 0.95 Children age 14-18 yr 0.85 Adults age 19-59 yr 0.8 Adult endurance athletes 0.8 1.2-1.4 Adult strength athletes 0.8 1.2-1.7 Vegetarian adult athletes 0.8 1.3-1.8 Elderly 60 0.8 1.0-1.3 Dieting adults (goal is fat loss while maintaining lean) 0.8 1.2-1.6 Post-diet maintenance (goal is maintenance of fat loss) 0.8 1.0-1.6 First half pregnancy 0.8 Second half pregnancy (based on pre-pregnancy weight) 1.1 Lactation 1.3 Range of Protein to Optimize Body Composition and Health (g/kg body weight/day) De Souza, 2010; FAO/WHO/UNU, 2007; Institute of Medicine, 2005; Layman, 2009; Lejeune, 2005; MSSE, 2009; Mojtahedi, 2011 The amount of protein consumed each day should meet daily requirements to maintain or improve health while minimizing excretion. It is available from animal sources such as meat, dairy and eggs and also vegetable sources including grains, rice, nuts and beans; however, quality, or the availability of EAAs, varies by source. The Food and Agriculture Organization of the United Nations (FAO) and World Health Organization (WHO) define protein quality by assigning a Protein Digestibility – Corrected Amino Acid Score (PDCAAS) to protein sources. PDCAASs are accepted internationally and are derived by assessing digestibility and comparing the protein source to an ideal mix of EAAs, termed a reference protein (FAO/WHO/UNU, 2007). The first quality component within the PDCAAS is digestibility, or the percentage of protein that is extracted from a protein source. It is determined by measuring nitrogen loss in feces during both protein-only and protein-free diets. The second component of each PDCAAS is an amino acid score (AAS), which is calculated by dividing the limiting EAA in a protein source by the amount of this EAA contained in the reference “ideal” protein and multiplying by 100. For example, if the protein source contains 98% of leucine contained in the reference protein and 86% of lysine, the AAS is 86 because the amount of lysine in the protein would limit protein synthesis. Note that the AAS is a function of the relation of the protein source to the reference protein, not the total percentage of EAAs in the protein source. Table 3 shows the PDCAAS 2 (Percent digestibility x AAS) for various protein sources. Since digestibility is always less than 100%, FAO/WHO truncates the PDCAAS, which clusters many protein sources. Table 3 PDCAAS values of protein sources1 Protein Source Beans Egg Beef Cow Milk Brown Rice Soy protein Wheat 1 Digestibility (%) 78 97 98 95 88 95 86 AAS 102 121 94 127 66 96 40 PDCAAS (Truncated) 78 97 92 95 58 91 34 PDCAAS (Not Truncated) 80 118 92 121 58 91 34 FAO/WHO/UNU, 1985; FAO/WHO/UNU, 2007; Schaafsma, 2000; Sarwar, 1997 Although both animal and vegetable sources may be identified that provide all EAAs with respect to the reference protein (AAS = 100 for each EAA) (Table 4), digestibility affects the total volume that must be consumed. For example, to supply the same net amount of available protein in an all-bean versus all-milk diet, the bean eater would have to consume 1.2 times (digestibility of milk divided by digestibility of beans) the amount of milk protein. This difference must be considered to ensure that net available protein meets dietary goals; however, typical diets contain a mixture of different protein sources, affecting both total digestibility and total AAS, which may balance the relative deficiencies of some proteins. Using the PDCAAS scoring model is sufficient for healthy adults who perform moderate exercise but probably not for individuals who participate in regular high-volume endurance or resistance exercise (Millward et al., 2008). One of the BCCAs, leucine, is not only important for protein structure, but also triggers pathways within the muscle that are necessary to build muscle. Additional leucine beyond the amount in the reference protein may be necessary to support muscle development, and except soy, plant proteins generally contain less leucine as compared to animal proteins, especially when the lower digestibility of most plant proteins is considered. Finally, leucine oxidation during hard exercise can rise, particularly when carbohydrate stores are low. Muscle recovery is optimized when protein is consumed 30-60 minutes after exercise, but not all highly-digested proteins are tolerated after a long and/or intense workout. Supplement manufacturers have recognized this and include protein that is quickly absorbed such as soy and/or whey into drinks, bars and powders. Since muscle building continues beyond the shortterm recovery window, proteins that are digested more slowly will further increase muscle. For example, milk can be separated into fast-acting whey and slow-acting casein. Similar to soy and whey, casein has a high PDCAA, but it does not provide as many BCAAs as soy or whey and digests more slowly. While studies comparing the effects of soy and whey are inconsistent, the additional leucine in whey appears to stimulate greater muscle anabolism after exercise, at rest and in the elderly (Wilkinson et al., 2007; Millward et al., 2008; Tang et al., 2009). Alternatively, soy may also affect total antioxidant status and reducing oxidative stress after exercise (Paul, 2009). A combination of both fast-acting and slow-acting protein sources in supplements or recovery foods can supply EAAs for both immediate and extended muscle building while providing other health benefits (Wilkinson et al., 2007; Paul, 2009; Tang et al., 2009). 3 Table 4 Essential amino acid content in selected foods and supplements1 Whole Wheat Flour (g/100g) 0.357 Dry Black Beans (g/100g) 0.601 5.174 4.100 0.336 0.443 0.954 3.542 8.326 6.653 0.657 0.898 1.725 5.327 2.868 7.345 5.694 0.303 0.359 1.483 1.130 0.907 1.618 2.013 0.179 0.228 0.325 0.682 1.168 Dried Egg White (g/100g) Beef Ground 95% Lean (g/100g) Soy Protein Isolate (g/100g) Dry Nonfat Milk (g/100g) Histidine 0.618 1.830 0.714 2.303 Isoleucine 1.342 4.581 0.939 Leucine 2.172 6.838 Lysine 1.824 Methionine 0.760 AA Whey Protein Powder (g/100g) Caesin Protein Powder (g/100g) 0.981 1.391 4.253 2.188 1.671 6.783 5.515 1.785 2.790 0.565 Phenylalanine 1.360 4.736 0.823 4.593 1.746 2.461 3.575 0.410 Threonine 1.112 3.685 0.846 3.137 1.632 5.441 3.247 0.291 0.367 0.909 Tryptophan 0.334 0.999 0.127 1.116 0.510 1.332 0.913 0.101 0.174 0.256 Valine 1.716 5.164 1.050 4.098 2.420 4.678 5.028 0.466 0.564 1.130 24 100 30.5 23 30.4 32 50 100 100 Amount per serving size Serving size (g) 100 1 2.150 Brown Rice Raw (g/100g) 0.202 Whole Egg 2 large (g/~100g) Histidine 0.618 0.439 0.714 0.702 0.226 0.423 0.688 0.101 0.357 0.601 Isoleucine 1.342 1.099 0.939 1.297 0.503 1.573 1.312 0.168 0.443 0.954 Leucine 2.172 1.641 1.671 2.069 0.815 2.531 2.129 0.329 0.898 1.725 Lysine 1.824 1.324 1.785 1.625 0.660 2.233 1.822 0.152 0.359 1.483 Methionine 0.760 0.670 0.565 0.345 0.209 0.492 0.644 0.090 0.228 0.325 Phenylalanine 1.360 1.137 0.823 1.401 0.402 0.748 1.144 0.205 0.682 1.168 Threonine 1.112 0.884 0.846 0.957 0.375 1.654 1.039 0.146 0.367 0.909 Tryptophan 0.334 0.240 0.127 0.340 0.117 0.405 0.292 0.051 0.174 0.256 Valine 1.716 1.239 1.050 1.250 0.557 1.422 1.609 0.233 0.564 1.130 USDA nutrient database, Optimum Nutrition, Inc. 4 There is criticism regarding the methodology that FAO/WHO has used to create PDCAASs for each protein source, but it would be difficult to verify the model in multiple populations while also considering activity level, activity type, body type and gender (Schaafsma, 2000; FAO/WHO/UNU, 2007; Millward et al., 2008). Instead, PDCAAs can be used as a starting point to determine baseline protein intake. The best way to ensure that protein requirements are met is to periodically check weight and body composition, which are related to caloric intake and dietary protein, respectively (Table 5). Understanding digestibility, EAA content and digestion rate of various protein sources is necessary to determine total dietary intake while meeting weight and lean mass goals. Table 5 Effect of protein and caloric intake on weight and lean mass goals Lean Mass Goal Maintain Weight Goal Increase % Protein Energy intake % Protein Energy intake Maintain or 1 Increase Lose 1 Building muscle is an expensive metabolic process. If normal daily caloric intake is insufficient, an increase in energy intake will be necessary to support muscle anabolism. 5 References Abete, I., Astrup, A., Martinez, J. A., Thorsdottir, I., & Zulet, M. A. (2010). Obesity and the metabolic syndrome: role of different dietary macronutrient distribution patterns and specific nutritional components on weight loss and maintenance. Nutrition Reviews, 68(4), 214-231. Campbell, W. W., Trappe, T. A., Wolfe, R. R., & Evans, W. J. (2001). The Recommended Dietary Allowance for Protein May Not Be Adequate for Older People to Maintain Skeletal Muscle. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 56(6), M373-M380. De Souza, P. G., & Martini, L. A. (2010). Effect of protein intake on bone and muscle mass in the elderly. Nutrition Reviews, 68(10), 616-623. FAO/WHO/UNU Expert Consultation (1985) Energy and protein requirements. Technical report series 724. World Health Organization, Geneva. FAO/WHO/UNU Expert Consultation (2007) Protein and Amino Acid Requirements in Human Nutrition. Technical report series 935. World Health Organization, Geneva. Institute of Medicine (2005), Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids (Macronutrients), National Academy Press, Washington, DC Food and Nutrition Board. Layman, D. K., Evans, E. M., Erickson, D., Seyler, J., Weber, J., Bagshaw, D., et al. (2009). A Moderate-Protein Diet Produces Sustained Weight Loss and Long-Term Changes in Body Composition and Blood Lipids in Obese Adults. The Journal of Nutrition, 139(3), 514-521. Lejeune, M., Kovacs, E. M. R., & Westerterp-Plantenga, M. (2005). Additional protein intake limits weight regain after weight loss in humans. British Journal of Nutrition, 93, 281-289. Millward, D. J., Layman, D. K., Tomé, D., & Schaafsma, G. (2008). Protein quality assessment: impact of expanding understanding of protein and amino acid needs for optimal health. The American Journal of Clinical Nutrition, 87(5), 1576S-1581S. Mojtahedi, M. C., Thorpe, M. P., Karampinos, D. C., Johnson, C. L., Layman, D. K., Georgiadis, J. G., et al. (2011). The Effects of a Higher Protein Intake During Energy Restriction on Changes in Body Composition and Physical Function in Older Women. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences. Nutrition and Athletic Performance. (2009). Medicine & Science in Sports & Exercise, 41(3), 709-731. Paul, G. L. (2009). The rationale for consuming protein blends in sports nutrition. Journal of the American College of Nutrition, 28(4 Supplement 1), 4645-4725. Sarwar, G. (1997). The Protein Digestibility‚ÄìCorrected Amino Acid Score Method Overestimates Quality of Proteins Containing Antinutritional Factors and of Poorly 6 Digestible Proteins Supplemented with Limiting Amino Acids in Rats. The Journal of Nutrition, 127(5), 758-764. Schaafsma, G. (2000). The Protein Digestibility-Corrected Amino Acid Score. The Journal of Nutrition, 130(7), 1865S-1867S. Tang, J. E., Moore, D. R., Kujbida, G. W., Tarnopolsky, M. A., & Phillips, S. M. (2009). Ingestion of whey hydrolysate, casein, or soy protein isolate: effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. Journal of Applied Physiology, 107(3), 987-992. Wilkinson, S. B., Tarnopolsky, M. A., MacDonald, M. J., MacDonald, J. R., Armstrong, D., & Phillips, S. M. (2007). Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage. The American Journal of Clinical Nutrition, 85(4), 1031-1040. USDA National Nutrient Database for Standard Reference, n.d., US Department of Agriculture, Agriculture Research Service, Nutrient Data Laboratory, Retrieved June 20, 2011 at http://www.nal.usda.gov/fnic/foodcomp/search/. 7