Kitchen Chemicals LAB
advertisement

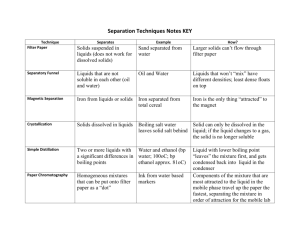
Name __________________________ lab partner(s) ___________________________ pd __ Kitchen Chemicals LAB The chemical and physical properties of a substance make up a sort of fingerprint that characterizes the substance. In this lab, you will test four unknown solids using three different liquids. The unknowns are common materials that you’d probably find in your kitchen. The results of your tests will give you the information you need to unravel the compositions of mixtures of two and three solids. Objectives: Observe the chemical and physical reactions of four common kitchen solids with three test reagents. Compare and interpret the reactions of the test reagents with five different mixtures of the four common kitchen solids (three mixtures will contain two different solids, two mixtures will contain three different solids). Infer the composition of each of the five unknown mixtures by comparing their reactions with those of the known materials. Materials: Spot plate White paper Pen/pencil solids “A, B, C, & D” solid mixtures “#1, 2, 3, 4, & 5” liquid reagents I, II, and III Procedure: 1. Place the spotplate on a white piece of paper so that the shorter side is on the top and bottom. 2. Label three long columns of wells I, II & III across the top. Label nine rows along the left side A, B, C, D, 1, 2, 3, 4, & 5. I II III A B C D 1 2 3 4 5 3. Add a SMALL amount of each solid to the row of three wells with the appropriate letter or number. 4. Observe and record the color and texture of each of the nine solids in the data and observations table. 5. Add 3 drops of liquid I to each of the nine solids in column I. 6. Observe any changes that take place (fizzing, color change to black, dissolving or no reaction) and record them in the data table. 7. Repeat steps 5 & 6 using liquid II & III Data and Observations Solid (pure) A (pure) B (pure) C (pure) D (2 solid mix) 1 (2 solid mix) 2 (2 solid mix) 3 (3 solid mix) 4 (3 solid mix) 5 Color Texture Reaction with liquid I Reaction with liquid II Reaction with liquid III Analyze and conclude 1. What properties and reactions characterize each of the four pure kitchen solids? A - _____________________________________________________________________ B - _____________________________________________________________________ C - _____________________________________________________________________ D - _____________________________________________________________________ 2. Can you positively identify the solids that are contained in any of the five mixtures? If so, identify which pure solids (A, B, C, or D) are definitely in each mixture and explain how you know. Mixture Contains # this/these solids: (A, B, C &/or D) 1 (2 solid mix) 2 (2 solid mix) 3 (2 solid mix) 4 (3 solid mix) 5 (3 solid mix) Explanation of conclusion 3. If you are unable to conclusively identify the solids in any of the mixtures, what are their LIKELY identities? Explain. Mixture # PROBABLY Contains this/these solids: (A, B, C &/or D) Explanation of conclusion 1 (2 solid mix) 2 (2 solid mix) 3 (2 solid mix) 4 (3 solid mix) 5 (3 solid mix) 4. Two of the four original solids, baking powder and baking soda, are often used in making baked goods. What characteristic probably makes them useful in baking (hint: it helps rise the dough)? Which two solids (A, B, C, or D) display this characteristic? 5. Baking powder is a mixture of two or more compounds, and it reacts with water or ANY other liquid that contains water. Baking soda is a single compound that reacts with acidic solutions but NOT with water. Which of the solids (A, B, C, or D) do you think is baking powder? EXPLAIN why. 6. One of the solids is starch, an organic compound. It produces a characteristic color when combined with iodine. Which solid (A, B, C, or D) gave this reaction?