Study Guide Exam 1
advertisement

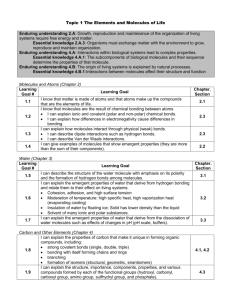
Matter consists of chemical elements in pure form an in combinations called compounds. Elements – Substances that cannot be broken down to other substances. Compounds – Substance composed of two or more elements. Matter – Anything that takes up space. *An Element’s properties depend on the structure of its atoms Atom – Smallest unit of matter that still retains the properties of an element Proton (+), Neutrons (no charge), and Electrons (-) are subatomic particles. Isotope – A different atomic form of an element (# of neutrons change) Energy – The capacity to cause change Electron Shell – An energy level of electrons at characteristic average distance from the nucleus of an atom *The formation and function of molecules depend on chemical bonding between atoms Chemical Bonds – Attraction between two atoms results from sharing of outer shell electrons or the present of opposite charges on atoms *The strongest chemical bonds are covalent and ionic. Covalent bonds – STRONG chemical bonds in which 2 atoms share 1 or more pairs of valence electrons. *Valence equals the # of unpaired electrons required to complete the atom’s outermost shell. Polar Covalent Bond – Electons NOT shared equally Nonpolar Covalent Bond – Electrons shared equally Ion – Charges Atom, (+) = Cation, (-) = Anion Ionic Bond – Chemical bond resulting from the attraction between oppositely charged ions Ionic Compounds – Compound resulting from formation of ionic bonds (Salts) Hydrogen Bonds – WEAK chemical bonds formed when slightly (+) hydrogen atoms of a polar covalent bond in one molecule is attached to the slightly (-) atom of a polar covalent bond in another molecule (Water has hydrogen bonds) *Chemical Reactions make and break chemical bonds *96% of all living matter is made of carbon, hydrogen, nitrogen and oxygen The polarity of water molecules result in hydrogen bonding. Polar Molecule – Two ends of a molecule have opposite charges (polar loves water) *Water is the solvent of Life Hydrophilic – Water loving, dissolves in water Hydrophobic – Water fearing, doesn’t dissolve in water Aqueous Solution – Solution where water is the solvent. *Acidic and Basic conditions affect living organisms Acid – Substance that increases the hydrogen concentration of a solution Basic – Reduces the hydrogen ion concentration of a solution pH Scale – Ranges from 0 – 14 (0 = acidic, 7 = neutral, 14 = basic) Buffer – Substances that minimize changes in the concentration of H+ (acidic) and OH- (basic) solutions *Accepts H+ ions when solution is too acidic *Donates H+ ions when solution is too basic Carbon is the backbone of life *What carbon can build is unlimited *Organic Chemistry is the study of carbon compounds *Carbon Atoms can form diverse molecules by bonding to four other atoms Isomers – Compounds that have the same # of atoms of the same elements but different structure and different properties Hydrocarbons – Organic molecules consisting of only carbon and hydrogen. *A Small # of chemical groups are key to the functioning of biological molecules Functional Groups – A specific configuration of atoms commonly attached to the carbon skeletons of organic molecules and usually involved in chemical reactions Adenosine Triphosphate (ATP) – An adenine + ribose sugar + three phosphates that releases energy when its phosphate bonds are hydrolyzed (broken off): This energy is used to drive chemical reactions Macromolecules – A giant molecule formed by joining of smaller molecules, usually by a condensation reaction. Polysaccharides, proteins and nucleic acids are macromolecules Polymer – Long molecule consisting of many similar or identical building blocks linked by covalent bonds Monomer – The repeating units that serve as building blocks Condensation Reaction – 2 molecules become covalently bonded to each other through the loss of water (Aka dehydration reaction) Hydrolysis – When polymers are disassembled to monomers. Reverse of above Enzymes – Specialized macromolecules that speed up chemical reactions in cells. Substrate, Active Site, Product Enzymes change shape using a Phosphate *Carbohydrates serve as fuel and building material Carbohydrates – Both sugars and polymers of sugars Monosaccharides – Simplest Carbs Disaccharides – 2 monosaccharides joined Polysaccharides – 100 to 1000’s of monosaccharides joined together Starch in plants Glycogen in animals *Lipids are a diverse group of hydrophobic molecules *Triglycerides/Phospholipids/Steroids *Phospholipids make up cell membranes *Steroids are a group of hormones made from cholesterol *Triglycerides – Saturated, Unsaturated, Trans Fat, Omega 3 Fatty Acids *Proteins have many structures, resulting in a wide range of functions (repair, catalyze, provide structural support…) Monomers - amino acids Polymers – polypeptides 4 levels of structure (Primary, Secondary, Tertiary, Quaternary *Denaturing Proteins – Protein wears down/becomes inactive High temperatures, low pH *Cellular Organelles Ribosomes - Serve as “Protein factories” of the cell Mitochondria – Site of cellular respiration (Powerhouse of the cell), produces ATP aerobically Lysosome – Produces Digestive enzymes Peroxisome – Breaks down Hydrogen Peroxide/ Catalase Golgi Apparatus – Vesiculation and Glycosylation Centrioles – Helps in cellular division by dividing the cells Rough Endoplasmic Reticulum – Stores Calcium, Helps in Protein Synthesis Smooth Endoplasmic Reticulum – Stores Calcium, Produces Triglycerides, Phospholipids and Steroid Hormones *The plasma membrane of the cell exhibits selective permeability *Phospholipids and proteins move laterally within the membrane *Membrane Proteins Integral Proteins – Trans membrane protein with hydrophobic regions that extend into and often span the hydrophobic interior of the membrane and the hydrophilic region in contrast with the aqueous solution on either side of the membrane Peripheral Proteins – A protein loosely bound to the surface of a membrane or to part of an integral protein and not embedded in the phospholipid bilayer *Membrane structure of the cell, results in semi-permeable or selective permeability *A cell must exchange molecules and ions with its surrounding, a process controlled by the plasma membrane *Hydrophobic substances can pass through the cell’s lipid bilayer rapidly. *To cross a cell’s membrane substances generally require transport proteins. *Passive Transport is diffusion of a substance across a membrane that DOES NOT require energy Diffusion – The movement of molecules of any substance from high concentration to low concentration, so that they spread out evenly into available space. Passive Transport – The diffusion of a substance across a biological membrane Osmosis – The diffusion of water across a semi-permeable membrane *Active Transport USES ENERGY to move solutes against their concentration gradient, from low concentration to high concentration. Primary – Movement of one type of substance at a time across the membrane Secondary – Movement of two types of substances at a time across the membrane. Symport/Antiport *Metabolism – The totality of an organism’s chemical reactions, consisting of catabolic and anabolic pathways, which manage the material and energy resources of the organism *Aerobic Respiration – A catabolic pathway that consumes oxygen and organic molecules, producing ATP. *Cellular Respiration has 3 stages Glycolysis – Splitting of glucose into pyruvate (occurs in the cytoplasm and is anaerobic) Citric Acid Cycle – Completes the metabolic breakdown of glucose molecules begun in glycolysis by oxidizing pyruvate to carbon dioxide (occurs in the mitochondria and is aerobic) Oxidative Phosphorylation – The production of ATP using energy derived from an electron transport chain. Uses NAD and FAD carriers of Hydrogen (Occurs in the mitochondria and is aerobic) *DNA is the genetic material *Four nitrogenous bases in DNA Adenine (A) Thymine (T) Guanine (G) Cytosine (C) *These nitrogenous base pairs in a DNA double helix are held together by hydrogen bonds. Base pairs are complementary: *Adenine bonds with Thymine *Guanine bonds with Cytosine *A chromosome consists of a DNA molecule packed together with proteins *Nucleic Acids store and transmit hereditary information *2 types: DNA and RNA DNA: (deoxyribose) (C, G, A, T) (Double-Stranded) Stores all hereditary information RNA: (Ribose) (C, G A, U) (Single-Stranded) *Nucleic Acids carry protein-coding instructions from DNA to protein synthesizing machinery. *Nucleus contains most of the genes in the cell *Nuclear envelope encloses the nucleus Chromosomes – Units of DNA that carries the genetic information Gene Expression – The process by which DNA directs the synthesis of proteins (or in some cases, just RNAs) *Genes specify proteins via the processes of TRANSCRIPTION and TRANSLATION Transcription – The synthesis of RNA under the direction of DNA (occurs in the nucleus) Translation – The synthesis of a polypeptide, which occurs under the direction of mRNA and a ribosome (occurs in the cytoplasm) *Genetic information is encoded as a sequence of non-overlapping base codons. A codon in messenger RNA either is translated into an amino acid or serves as a stop signal. Codons must be read in the correct order *In RNA uracil (U) takes the place of thymine (T) *RNA is modified after transcription RNA Processing – Modification of RNA transcripts, including splicing out introns, joining together exons, and adding a methylated cap and a poly A tail Introns – A non-coding, intervening sequence within a primary transcript that is removed from the transcript after RNA polymerase reads the mRNA; also refers to the regions of DNA from which this sequence was transcribed Exons – Coding regions. A sequence within a primary transcript that remains in the RNA after RNA processing; also refers to the region of DNA from which this sequence was transcribed *More on Translation *A cell translates mRNA message into protein using tRNA. After binding up specific amino acids, tRNA lines up via their anticodons at complimentary codons on mRNA. Ribosomes help facilitate this coupling with binding sites for mRNA and tRNA *Point mutations can affect protein structure and function *A point mutation is a change in one DNA base pair, which may lead to production of a non-functional protein. Base-pair substitutions can cause missense or nonsense mutations. Base-pair insertions or deletions may produce frameshift mutations *Mutagens – A chemical of physical agent that can interact with DNA and cause mutation such as Benzene found in gasoline