DSMC Annual Renewal Form
advertisement

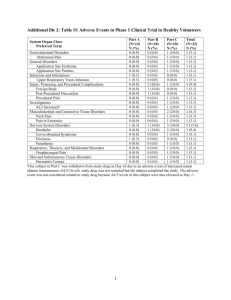
Data and Safety Monitoring Committee (DSMC) Annual Renewal Form Investigator-Initiated Studies without a DSMB Protocol #: Date form completed: Protocol Title: Principal Investigator: The information provided in this form should be in accordance with the Data and Safety Monitoring Plan written in the protocol. Submit completed form and attachments to Clinical Research Support at submissions@fredhutch.org Submission due date: A. Dose Escalation: Dose Escalation (or multi dose level) definition: For each subject to date, provide cohort and dose assignment. Add rows to table as needed so that each subject enrolled to date is displayed (Attachments in the following format are acceptable) If a subject experienced a dose modification per protocol, indicate dose modification. If this is a single dose level study state “not applicable” in the first subject line and skip this table. Subject # Cohort Study Drug Dose Level/Modification B. Dose Limiting Toxicities (DLT) (as per protocol): Dose limiting toxicity (DLT) definition: Provide the definition of DLT as written in the protocol. If protocol does not define DLTs, note “not applicable” and skip this table. Total # of Patients at dose level Dose Level Total # of Patients Experienced DLT Identify DLTs: 01312 DSMC Annual Renewal Form Page 1 of 3 Version: 07/28/2015 Data and Safety Monitoring Committee (DSMC) Annual Renewal Form Investigator-Initiated Studies without a DSMB C. Stopping Rules (as per protocol): Stopping rules: Provide the stopping rules as written in the protocol. If protocol does not have stopping rules, note “not applicable” and skip this table. How are stopping rules followed? Stopping rule(s) per patient Have any stopping rules been met? Yes Stopping rule(s) per event No Stopping rule: # of Patients/Events met Stopping Rule Identify events that count toward stopping rule: D. Were there any premature terminations from study due to toxicity not otherwise listed in this form? Yes No If yes, please describe. If no, note “not applicable” and skip this table. E. Adverse Events: If there are no Adverse Events being reported, please provide explanation: If there are Adverse Events to report, provide cumulative tabulation of adverse events by type and grade as described in the data and safety monitoring plan outlined in your protocol. Examples of Adverse Event tables are below. Events must be reported in tabular format Report must include the category; toxicity; grade and number of events For multicenter studies where the Cancer Consortium is not the coordinating center, report only local events. For multicenter studies where the Cancer Consortium is the coordinating center, report study wide events. Include Grade 5 events for reporting number of deaths on study. 01312 DSMC Annual Renewal Form Page 2 of 3 Version: 07/28/2015 Data and Safety Monitoring Committee (DSMC) Annual Renewal Form Investigator-Initiated Studies without a DSMB Examples of Adverse Event Tables Example Table 1: Adverse Events Category (CTCAE) Gastrointestinal Disorders Gastrointestinal Disorders Vascular disorders Example Table 2: Adverse Event/Toxicity 1 N Nausea Pancreatitis Hypertension Grade and Number (N) of events 2 3 4 5 N N N N 1 1 1 Adverse Events Category Gastrointestinal Disorders Gastrointestinal Disorders Vascular Disorders 01312 DSMC Annual Renewal Form CTC Term Nausea Pancreatitis Hypertension Page 3 of 3 Grade Number of Events 3 4 2 8 1 4 Maximum Attribution Unlikely Possibly Unrelated Version: 07/28/2015