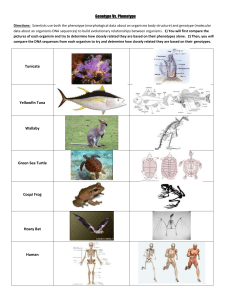
CYP3A4 phenotype: EXTENSIVE METABOLIZER
advertisement

PGXL Laboratories 201 E. Jefferson Street Louisville, KY 40202 Phone: (502) 569-1584 Laboratory Director: Linder PhD CLIA#: 18D0983143 KY State License #: 200251 info@pgxlab.com Mark W. Sex: M PGXL Laboratories Name: Test, Patient Example, Physician DOB: 7/15/1955 Age: 59 Accession Client ID #: #: PG-14-01698 Client: PGXL Laboratories Collected: 11/14/2014 Received: 11/17/2014 Reported: 11/17/2014 Specimen Type : Buccal Swab Reason for Referral : Depression Final PGXL Interpretive Report Therapeutic implications adapted from published resources: CYP2D6 phenotype: POOR METABOLIZER Avoid: Alternative Considerations: Adjust Dosage: Adjustment: Codeine** Morphine, non-opioid Aripiprazole† decrease 50% Hydrocodone** Hydromorphone, non-opioid Clomipramine† decrease 50% Oxycodone** Oxymorphone, non-opioid Doxepin† decrease 60% Tramadol** Consider active drug, non-opioid Flecainide† decrease 50% Tamoxifen** Anastrozole, exemestane, letrozole Haloperidol† decrease 50% Amitriptyline† Citalopram, sertraline Imipramine† decrease 70% Venlafaxine† Citalopram, sertraline Nortriptyline† decrease 60% Risperidone† Quetiapine, olanzapine, clozapine Propafenone† decrease 70% Metoprolol† decrease 75%, or atenolol, bisoprolol Vortioxetine† maximum 10mg/day Zuclopenthixol† decrease 50%, or flupenthixol quetiapine, olanzapine, clozapine CYP2C19 phenotype: POOR METABOLIZER Avoid: Alternative Considerations: Adjust Dosage: Adjustment: Clopidogrel** Prasugrel, Ticagrelor Citalopram† Maximum 20mg/day Imipramine† decrease 30% Sertraline† decrease 50% CYP2C9 phenotype: INTERMEDIATE METABOLIZER Decreased metabolic clearance Adjust Dosage: Adjustment: expected. Phenytoin† decrease 25% This report continues... (Final) 1 Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: ent name: Test, Patient PG-14-01698 Warfarin† Adjust based on multiple factors VKORC1 phenotype: INTERMEDIATE WARFARIN SENSITIVITY Average VKORC1 enzyme expression and average warfarin dose requirement expected. Warfarin Dose Information: Estimated warfarin maintenance dose requirement: 5.5 mg/day‡ Estimated time to warfarin steady-state: Delayed, 11-15 days The time to reach steady-state affects the timing of INR measurement for optimal interpretation. Time to reach steady-state is the time period required for the dosage to consistently yield reproducible blood concentrations of warfarin. INR measurements are most reliable when measured after steady-state has been achieved. INR measurements before steadystate blood concentrations have been achieved should be interpreted with caution, as they may not be indicative of stable therapy. CYP3A4 phenotype: EXTENSIVE METABOLIZER Normal metabolic clearance expected. Common CYP3A4 medications below. CYP3A5 phenotype: DECREASED METABOLIZER Genotype consistent with reduced CYP3A5 enzymatic activity and represents the majority (60-80%) of the population. For DMs, maintenance dosages for most CYP3A drugs are lower than extensive metabolizers. Common CYP3A5 medications below. CYP1A2 phenotype: HYPERINDUCER Rapid metabolism expected, especially in smokers. Consider dose increases for medications inactivated by CYP1A2 particularly in smokers, or alternative medications not metabolized by CYP1A2. Common CYP1A2 medications below. SLC6A4 phenotype: POOR RESPONDER Decreased serotonin transporter expression expected. Risk of decreased response to SSRI-based therapies and increased risk of adverse events. Consider non-SSRI antidepressant therapies, such as SNRIs or tricyclic antidepressants alternatives. SLCO1B1 phenotype: 2.6 FOLD INCREASED MYOPATHY RISK Reduced OATP1B1 transporter function. Adjust Dosage: Adjustment: Increased risk of statin-induced myopathy. Simvastatin max 10-20 mg and consider monitoring CK levels, or consider alternative statin if also taking verapamil or diltiazem. Consider low 10-20 mg dose of alternative statin if patient also CYP3A4*22 carrier. Atorvastatin max 10-20 mg Factor V Leiden phenotype: NO INCREASED THROMBOSIS RISK Factor II phenotype: 2-3 FOLD INCREASED THROMBOSIS RISK MTHFR phenotype: INCREASED RISK This report continues... (Final) Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 2 ent name: Test, Patient PG-14-01698 Increased risk of hyperhomocysteinemia, coronary artery disease, and thrombosis when folate deficiency is present. Consider folate supplementation. COMT phenotype: HIGH ACTIVITY The COMT Val/Val genotype results in increased COMT activity and decreased dopamine levels in the prefrontal cortex. Compared to patients with the Met allele, Val/Val patients with depression are less likely to achieve remission when treated with SSRI antidepressants, and Val/Val patients with schizophrenia are less likely to demonstrate improved cognitive effects when treated with antipsychotics. Val/Val patients may benefit from agents that increase dopamine availability including methylphenidate and amphetamine. OPRM1 phenotype: Adjust Dosage: INTERMEDIATE OPIOID RESPONDER / NORMAL NALTREXONE RESPONDER Morphine, increase up to 10% Opioid response: Higher than average doses of morphine typically required (may also apply to other active opioids, eg, hydromorphone, oxymorphone). Note: Formation of active opioid metabolites (e.g., morphine) from prodrugs (eg, codeine) is dependent on CYP2D6 activity. Naltrexone response: In patients treated with naltrexone for alcohol dependence, those with the AG genotype have a 15% lower average relapse rate compared to the typical on-treatment relapse rate. Overall, relapse rate among G allele carriers is 25% compared to 54% in patients with AA genotype. **Prodrug: PM lack of efficacy due to failure to produce active metabolite, UM at risk of toxicity from increased active metabolite; †Active drug: PM risk of adverse events due to diminished drug clearance, UM risk of subtherapeutic response. CYP2D6 *4/*4 Poor Metabolizer (PM): This patient’s genotype is consistent with a lack of CYP2D6 enzymatic activity. PMs are at increased risk of drug-induced side effects due to diminished drug elimination of active drugs or lack of therapeutic effect resulting from failure to generate the active form of the drug, as is the case with pro-drugs. CYP2C19 *2/*2 Poor Metabolizer (PM): This patient’s genotype is consistent with significantly reduced CYP2C19 enzymatic activity. PMs are at increased risk of drug-induced side effects due to diminished drug elimination of active drugs. Patients with no CYP2C19 function (PMs) taking clopidogrel lack adequate antiplatelet response and remain at risk for cardiovascular events, including thrombosis, myocardial infarction, stroke, and death. CYP2C9 *1/*3 Intermediate Metabolizer (IM): This patient’s genotype is consistent with reduced CYP2C9 enzymatic activity. Reduced CYP2C9 activity leads to lower dose requirement (e.g., warfarin) due to decreased clearance, increased elimination half-life, and increased time to reach steady-state blood concentrations. VKORC1 Result: GA Intermediate Warfarin Sensitivity: ‡The warfarin maintenance dose estimate was derived using a published formula that accounts for age, gender, weight, and CYP2C9 and VKORC1 genotypes. This estimate should be viewed as an example of how this information can be taken into consideration by the physician as part of the overall patient management strategy. CYP3A4 *1/*1 Extensive Metabolizer (EM): This patient’s genotype is consistent with normal CYP3A4 enzymatic activity. In general, extensive metabolizers can be administered drugs which are substrates of the CYP3A4 enzyme following standard dosing practices. CYP3A5 *3/*3 Decreased Metabolizer (DM): This patient's genotype is consistent with reduced CYP3A5 enzymatic activity and represents the majority (60-80%) of the population. For DMs, maintenance dosages for most CYP3A drugs are lower than extensive metabolizers. CYP1A2 *1F/*1F Hyperinducer: This report continues... (Final) Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 3 ent name: Test, Patient PG-14-01698 Presence of the -163 C>A variant (present in *1F, *1L, *1J hyperinducer alleles) results in the hyperinduction phenotype. Hyperinduction may yield 20-40% higher CYP1A2 activity compared to the normal *1A allele in the presence of an inducer, such as tobacco smoke. Patients who are homozygous for hyperinducer alleles may exhibit even higher rates of CYP1A2 enzymatic activity and have been described as ultra-rapid metabolizers for olanzapine. As an example, carriers of CYP1A2*1F with the hyperinduction phenotype may exhibit as much as 50% lower than expected plasma levels of olanzapine and clozapine, which could lead to sub-therapeutic response. Hyperinducers may require increased dosages of CYP1A2 substrates due to higher than normal rates of drug metabolism in the presence of an inducer. SLC6A4 Result: S/S Poor Responder: Patients with S/S, S/LG, and LG/LG genotypes may be less likely to achieve remission of depression, more likely to have a higher number of antidepressant trials, and are more likely to experience adverse effects when treated with selective serotonin reuptake inhibitors (SSRIs) compared to non-SSRIs. SLCO1B1 *1/*5 Moderate Myopathy Risk: This patient's genotype is consistent with decreased OATP1B1 transporter function and is associated with an increased risk of simvastatin-induced myopathy at 40 mg doses. The FDA recommends avoiding 20mg simvastatin dose when the patient is co-prescribed verapamil or diltiazem. Consider low dose alternatives to simvastatin in patient who also carry the CYP3A4*22 variant allele. Factor V Leiden Result: GG, no variant Normal Thrombosis Risk: This genotype result revealed that the patient does not possess the Factor V Leiden (1691G>A) variant, which is consistent with no increased risk of thromboembolic events. Factor II Result: GA, heterozygous Moderate Thrombosis Risk: This genotype result revealed that the patient is heterozygous for (has one copy of) the Factor II (Prothrombin) 20210 G>A variant, which has been associated with an increased risk of thromboembolic events. This variant is found in approximately 2% of individuals in the U.S. Presence of the Factor II 20210G>A variant increases the risk of venous thromboembolism (VTE) by 2-3 fold in heterozygous carriers and >3 fold in homozygous carriers. MTHFR 677(C>T): TT, homozygous variant MTHFR 1298(A>C): AC, heterozygous Increased Risk: Presence of the 677 C>T polymorphism of MTHFR leads to decreased MTHFR enzymatic activity and elevated homocysteine. This patient’s genotype is consistent with an increased risk of hyperhomocysteinemia, atherosclerotic heart disease, myocardial infarction, cerebrovascular disease, and venous thrombosis. Additionally, associations between the 677 C>T polymorphism and increased risk for methotrexate toxicity, increased 5-fluorouracil chemosensitivity, and increased risk of fetal neural tube defects in pregnant women have also been reported in states of folate deficiency. COMT Result: Val/Val High activity: COMT is an enzyme that degrades dopamine and norepinephrine, primarily in the prefrontal cortex. The Val allele has higher enzymatic activity resulting in higher dopamine degradation and lower dopamine concentrations as compared to the Met allele. Lower prefrontal dopamine in Val/Val patients is associated with impaired cognitive function and working memory deficits. Val/Val homozygotes are less likely to achieve remission of symptoms when treated with psychotropic medications and may benefit from agents that increase dopamine availability such as methylphenidate and amphetamine. OPRM1 Result: AG Intermediate Opioid Responder/Normal Naltrexone Responder: This patient's genotype is consistent with higher than average doses of active opioids (e.g., morphine, hydromorphone, oxymorphone) for therapeutic effect. Formation of active opioid metabolites (e.g., morphine) from prodrugs (e.g., codeine) is dependent on CYP2D6 activity. In patients treated with naltrexone for alcohol dependence, those with the AG genotype have a 15% lower average relapse rate compared to the typical on-treatment relapse rate. Overall, relapse rate among G allele carriers is 25% compared to 54% in patients with AA genotype. The AG genotype is found in approximately 30% of Caucasians, 20% of African Americans, and 50% of Asians. This test should not be used as the sole means of treatment decision making, and should be regarded by the ordering physician as adjunctive to the overall patient management strategy. These genotyping results do not eliminate the necessity to account for non-genetic factors that can influence dose requirements for or responses to medications that are metabolized by these enzyme systems. Drug-drug and drug-gene interactions that lead to enzymatic inhibition or induction may lead to altered metabolism. Results should always be interpreted in context with the clinical picture and all co-administered medications. This report continues... (Final) Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 4 ent name: Test, Patient PG-14-01698 CYP2D6 common medications Psychiatry Antidepressants Amitriptyline Atomoxetine Clomipramine Desipramine Doxepin Duloxetine Fluoxetine Fluvoxamine Imipramine Maprotiline Mirtazapine Nortriptyline Paroxetine Trimipramine Venlafaxine Vortioxetine Elavil, Levate Strattera Ananfranil Norpramin Sinequan Cymbalta Prozac Luvox Tofranil Ludiomil Remeron Pamelor, Aventyl Paxil Surmontil Effexor Brintellix Antipsychotics Amphetamine Aripiprazole Chlorpromazine Haloperidol Iloperidone Lisdexamfetamine Perphenazine Risperidone Thioridazine Zuclopenthixol Adderall, Dexedrine Abilify Thorazine Haldol Fanapt Vyvanse Trilafon Risperdal Mellaril Cisordinol, Clopixol Pain Management Codeine** Hydrocodone** Oxycodone** Tramadol** Various brands Lortab, Vicodin, Norco Oxycontin, Percocet Tramal, Ultram Cardiology Carvedilol Flecainide Metoprolol Nebivolol Propafenone Propranolol Timolol Coreg Tambocor Toprol-XL Bystolic Rythmol Inderal, InnoPran Blocadren Urology Darifenacin Fesoterodine Tamsulosin Tolterodine Enablex Toviaz Flomax Detrol Other Dextromethorphan Diphenhydramine Donepezil Doxorubicin Gefitinib Loratadine Meclizine Promethazine Tamoxifen** Robitussin D, Delsym Benadryl Aricept Adriamycin Iressa Claritin Antivert Phenergan Soltamox (**indicates prodrug) CYP2C19 common medications Amitriptyline Elavil, Levate Lansoprazole Prevacid Carisoprodol** Soma Methadone (active portion) Various Brands Citalopram Celexa Nelfinavir Viracept Clopidogrel** Plavix Omeprazole Prilosec Dexlansoprazole Dexilant Pantoprazole Protonix Diazepam Valium Rabeprazole Aciphex Escitalopram Lexapro Sertraline Zoloft Esomeprazole Nexium Voriconazole Vfend Imipramine (**indicates prodrug) Tofranil CYP2C9 common medications Celecoxib Celebrex Losartan Cozaar Diclofenac Cataflam, Voltaren XR Meloxicam Mobic This report continues... (Final) Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 5 ent name: Test, Patient PG-14-01698 Fluvastatin Lescol Naproxen Aleve Glimepiride Amaryl Phenytoin Dilantin Glipizide Glucotrol Rosuvastatin Crestor Glyburide Diabeta Tolbutamide Orinase Ibuprofen Advil, Motrin Warfarin Coumadin CYP3A4/CYP3A5 common medications PSYCHIATRY Benzodiazepines Alprazolam Xanax Midazolam Versed Triazolam Halcion Antipsychotics Buspirone Carbamazepine Lurasidone Quetiapine Ziprasidone Buspar Tegretol Latuda Seroquel Geodon Antidepressants Desvenlafaxine Levomilnacipran Nefazodone Reboxetine Trazodone Vilazodone Pristiq Fetzima Serzone Edronax Desyrel Viibryd CARDIOLOGY Amiodarone Cilostazol Dronedarone Quinidine Ranolazine Rivaroxaban Ticagrelor Cordarone Pletal Multaq Quinaglute, Quinidex Ranexa Xarelto Brilinta Statins Atorvastatin Lovastatin Mevastatin Simvastatin Lipitor, Caduet Mevacor, Advicor Compactin Zocor, Vytorin, Simcor Ca Channel Blockers Amlodipine Diltiazem Felodipine Lercanidipine Nifedipine Nisoldipine Nitrendipine Verapamil Norvasc Cardizem Plendil Zanidip Adalat Sular Various brands Calan, Isoptin, Verelan ONCOLOGY Docetaxel Doxorubicin This report continues... (Final) Taxotere Adriamycin PAIN MANAGEMENT Alfentanil Buprenorphine Cyclobenzaprine Fentanyl Alfenta Subutex, Suboxone Flexeril Actiq, Duragesic UROLOGY Alfuzosin Avanafil Darifenacin Doxazosin Dutasteride Finasteride Oxybutynin Sildenafil Silodosin Solifenacin Tamsulosin Tadalafil Vardenafil Uroxatral Stendra Enablex Cardura Avodart Proscar Ditropan Viagra Rapaflo Vesicare Flomax Cialis Levitra OTHER Antimicrobials Clarithromycin Erythromycin Indinavir Nelfinavir Ritonavir Saquinavir Telithromycin Biaxin E-Mycin Crixivan Viracept Norvir Fortovase Ketek Steroids Beclomethasone Budesonide Estradiol Hydrocortisone Progesterone Testosterone QVAR Symbicort, Uceris Various brands Various brands Various brands Various brands Immunosuppressants Cyclosporine Sirolimus Tacrolimus Gengraf Rapamune Prograf Other Bromocriptine Colchicine Eletriptan Guanfacine Parlodel Colcrys Relpax Intuniv Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 6 ent name: Test, Patient Erlotinib Gefitinib Imatinib Pazopanib Regorafenib Vincristine CYP1A2 common medications Other Cyclobenzaprine Flutamide Frovatriptan Lidocaine Rasagiline Ropinirole Ropivacaine Tacrine Theophylline Tizanidine Triamterene Zolmipitran Caffeine 17-beta estradiol PG-14-01698 Tarceva Iressa Gleevec Votrient Stivarga Loperamide Saxagliptin Zolpidem Flexeril Eulexin Frova Xylocaine Azilect Requip Naropin, Polyamp Cognex Aerolate Zanaflex Dyrenium Zomig Various Various Psychiatry Asenapine Clozapine Duloxetine Olanzapine Promazine Imodium Onglyza Saphris Clozaril Cymbalta Zyprexa Sparine Methods: Laboratory specimens were analyzed by a real-time PCR method (TaqMan SNP Genotyping, Life Technologies) developed by PGXL Laboratories. Alleles tested may include up to all of the following, depending on the clinical order, as reported above: CYP2D6: active *1,*2; partially active *9,*10,*17,*29,*41; inactive *3,*4,*5 (deletion),*6,*7,*11,*12; gene duplication *1,*2,*4 (inactive),*6 (inactive), *9,*10,*17,*29 or *41; CYP2C19: active *1; inactive *2,*3,*4,*6,*8,*9,*10; hyperactive *17; CYP2C9: active *1; decreased function alleles *2,*3,*4,*5,*6,*8,*11,*12; CYP3A4: active: *1; decreased activity *3,*17,*22; CYP3A5: active *1; decreased activity: *2,*3,*6,*7; CYP1A2 normal *1A,*1E; decreased inducer *1C,*1K; hyperinducer based on presence of -163 C>A variant *1F,*1J,*1L; VKORC1 high sensitivity variant -1639 G>A; SLCO1B1 normal *1, variant *5; OPRM1 118A>G poor responder variant; COMT 472G>A (158 Val>Met) low activity variant; Factor V Leiden 1691 G>A; Factor II (prothrombin) 20210 G>A; MTHFR 677C>T and 1298A>C; SULT4A1 rs763120 T>C and rs5764010 C>T. SULT4A1-1 positive haplotype is defined by presence of rs76210 C and rs5764010 T, as these variants are in linkage disequilibrium with the complete SULT4A1-1 haplotype previously described. As applicable, testing for SLC6A4 was performed by PCRRFLP analysis, including normal transporter expression allele LA, decreased expression alleles S, LG. Performance characteristics were validated by PGXL Laboratories with analytical specificity and sensitivity of >99% for detection of the above variants. Other known variants that are not listed are not detected. These methods have not been cleared or approved by the U.S. FDA, however the FDA has determined that such clearance or approval is not necessary. This test is used for clinical purposes and should not be regarded as investigational or for research. This laboratory is certified under the Clinical Laboratory Improvement Amendments (CLIA) as qualified to perform high complexity clinical laboratory testing. All testing was performed at PGXL Laboratories, CLIA No. 18D0983143, KY State License No. This report continues... (Final) Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 7 Patient name: Test, Patient PG-14-01698 200251, 201 E. Jefferson Street, Suite 309, Louisville, KY 40202, (502)569-1584. References available upon request Electronically Signed on 11/17/2014 at 5:09 PM by Mark Borgman, PhD Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 8 Patient name: Test, Patient PG-14-01698 END OF REPORT (Final) Copyright 2012-2014 Pharmacogenetics Diagnostic Laboratory, LLC Page: 9