PorcineIgG_Purification_Porcine
advertisement

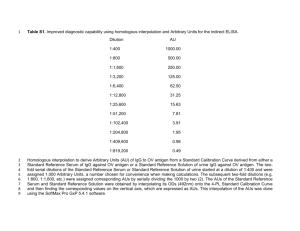
Porcine IgG Purification and Extraction from Porcine Serum Catherine Su 2012-2013 (Adapted from Juan Afanador’s procedure 2011-2012) Background: In blood, serum is a collection of organic components that don’t include clotting factors such as white blood cells or red blood cells, and it is often referred to as plasma. This collection of proteins is the source of most electrolytes, antigens, antibodies and hormones. Out of all this organic material, Immunoglobulin G (IgG), an antibody, is the most prominent protein. This fundamental component of the immune system acts like any other antibody in detecting antigens (components of allergens or other hazards) and setting off a response from the immune system. IgG is unique in the sense that it composes 75% of serum, and it is known for its hypersensibility to fungi, bacteria and viruses. IgG is a “Y” shaped antibody that has a molecular weight of 150 kilodaltons (kDa). It is composed of two sets of parallel branches that are known as chains. In each of these sets, there is a light chain (~25 kDa) and a heavy chain (~50 kDa). Since IgG is a protein, and we know its molecular weight, we are able to visualize it via Polyacrylamide Gel Electrophoresis (PAGE). PAGE gel visualization is an easy method of visualizing IgG, yet it isn’t completely accurate as there are many other proteins that could have similar molecular weights. Affinity chromatography is a method used to separate proteins based on the interactions between antigen and antibody, enzyme and substrate, or receptor and ligand. IgG binds to the protein A in the column while the solution is washed and cycled through the column. After the IgG has bind to protein A, an elution buffer with a very low pH can be used to elute the IgG from the protein A, leaving purified IgG from pig serum. This is the most efficient way to purify IgG. A new form of column for small quantities of IgG purification is called a Protein A Phytip developed by PhyNexus. These 1 mL tips are packed with Protein A on the bottom of the tip. Purpose: To extract Immunoglobulin-G from porcine serum and optimize its purification with PAGE gels and spectrophotometry. Materials: 1X PBS Buffer at pH 7.4 Porcine Serum Spectrophotometer 0.45 µm filter bottle 0.22 µm filter bottle 60 cc syringe PhyNexus PhyTip Columns with Protein A affinity resin 1.5 L Laemmli Buffer Sample Prep Loading Dye NuSep Tris-Glycine NN 10% 10 well gel 10-250 kDa sizing standard protein ladder Centrifuge 15 mL centrifuge tubes 13 x 100mm glass tubes P1000, P100, P20 Micropipettes 10 mL, 5 mL, 2 mL pipets Acetic acid (1M, 0.15M) 1.7 mL tubes 20% ethanol Coomassie blue PAGE gel loading tips 1M Tris solution 1 mL glass pipets (for acids) Procedures: PART I: Sterilize Porcine Serum 1. Let porcine serum thaw. 2. Filter the porcine serum across filter paper and collect in beakers. 3. Continue filtering and sterilizing the filtered serum by vacuum pump filtering it with 0.45µm then 0.22µm filter bottles. 4. Aliquot the filtered serum into 15 mL conical tubes, label and freeze at -20°C until use. PART II: Characterizing IgG by PAGE 1. Prepare 1.5 L of Laemmli buffer (16.5g of TRIS base, 9g of boric acid, 15 mL of 10% SDS). First dissolve in 1.3 L, then fill up to 1.5 L with deionized water. 2. Prepare 50 mL of PAGE Sample Prep Loading dye without BME (5g sucrose, 0.05g of bromophenol blue, 30 mL of Laemmli buffer). Note: To make 1 mL of SPLD w/ BME, add 950 uL of SPLD with 50 uL of BME in the chemical fume hood. 3. Create 1:10, 1:30, 1:60, and 1:90 dilution of serum in Laemmli buffer. Heat samples on heat block at 80oC for 5 minutes before loading samples onto gel. 4. Run a 10 well Tris-glycine gel with multiple porcine serum dilutions. 5. Sample preparation: 25 µL of loading dye and 25 µL of the porcine serum per sample 6. Run empty gel for 10 minutes 7. Load 30 µL of each sample into each well, 10 µL of standard marker in well 5 and 15 µL of marker in well 10. Run gel at 170 volts for 60 minutes or when the samples are in the bottom quadrant. 8. Photograph the gel and store in dH2O for storage. The band at 150 kDa will help determine the best concentration of the pig serum. Put gel in NuSep Fast Staining for 30 minutes or Coomassie Blue staining for at least 3 hours. Pour off Coomassie Blue staining and add destaining solution for at least 7 hours. Note: Staining and destaining should be done on an orbital shaker. PART III: Purification of IgG via Protein A Affinity Chromatography (modified from PhyNexus (2007) procedure) 1. Prepare a 1X PBS solution, pH 7.4, for serum dilution 1:60 (Dissolve 8g NaCl, 0.2 g KCl, 1.44g Na2HPO4, 0.2g KH2PO4 in 800 mL of dH2O, adjust pH, then fill up to 1L with dH2O). Autoclave to sterilize. Preparation of PhyTip: 1. Flush Phytip with water to remove ethanol. Add water (about 0.6 mL) to the top of the PhyTip column, then discard the water. Repeat 3 times. 2. Equilibrate Phytip with PBS. Add PBS (about 0.6mL), then discard the PBS. Repeat 3 times. Purification of IgG: 1. Bind pig IgG onto the Protein A resin inside the Phytip. Put 0.4 mL pig serum in the column and expel into a tube. Add the 0.4 mL of pig serum to the column again and expel into a second tube. Repeat this again and expel into a third tube. These are the fractions collected for the removed substances that are not IgG. 2. Wash unbound components off the Protein A resin. Pour about 5 mL PBS into a tube or beaker. Add 0.6 mL PBS, then expel into a tube. Repeat 5 times, each time expelling into a new tube. These are also fractions collected of removed substances in the serum that are not IgG. 3. Adjust the IgG solution to pH 5.5 – 6.5. Predetermine the volume of 1 M Tris base (in our case, 70 uL) needed to change the 0.6 mL elution buffer (0.15M acetic acid, pH 3.7) to pH 6.0. 4. Elute pig IgG. Add 0.6 mL 0.15M acetic acid (pH 3.7); expel the solution into a clean tube that contains the predetermined amount of 1M Tris base necessary to change the pH to 6.0. 5. Determine the IgG concentration of this solution by diluting 0.1 mL of this solution with 0.9 mL of PBS, then measure the absorbance at 280 nm. Determine the purity of this solution by SDS PAGE. Cleaning and Storage of PhyTip: 1. Clean the PhyTip with acid to remove remaining bound material. Add 0.6 mL of 1 M acetic acid, then discard the acid. Repeat 3 times. 2. Clean the PhyTip with water. Add 0.6 mL water, then discard. Repeat 3 times. 3. Store the Phytip in 20% ethanol. Add 0.6 mL of 20% ethanol then store the PhyTip at 2-8ºC. Results: Lanes are loaded with increasing volumes of the eluted IgG sample. IgG heavy chain 53 kDa IgG light chain 25 kDa 52 kDa 29 kDa 21 kDa Note: This was conducted with a reused PhyTip so it is a follow-up to make sure the PhyTips are reusable and can produce the same results a second time. Conclusion: The ultimate purpose of my lab is to extract IgG from porcine serum. These results may be visualized on a vertical gel to determine the purity of the protein. In this experiment, I was able to use the 1:1 and 1:60 dilution of filter sterilized porcine serum from Juan’s previous experiments. Then, I ran a gel of 1:10, 1:30, 1:60, and 1:90 dilutions of the filtered porcine serum to determine which dilution was the best for purification. Since the 1:60 had the best visualized bands, I decided to use this serum dilution for the purification of IgG. At the beginning of the fall semester, I had around 60 Protein A PhyTips that have been used and stored from Genentech. Each time, I would yield results of 3 bands at around 115 kDa, 50 kDa, and 25 kDa. I wasn’t sure whether this was denatured and undenatured IgG, or it was other protein impurities. Other times, I would get very streaky bands, which I discovered was due to too high of a voltage. After the old PhyTips became contaminated, I obtained new Protein A PhyTips from Genentech. These PhyTips yielded a band at 53 kDa and a band at 25 kDa, which represent the heavy chain and the light chain of IgG. There is very strong evidence that I have purified IgG from the porcine serum. The new PhyTips were able to yield good results and when I reused the PhyTip, I was able to obtain consistent results. Possible errors include some sort of contamination of the old PhyTips. Since there was soon growth of some type of fungus or bacteria, it is possible that they were previously contaminated before I received them that may have influenced its function. If I had obtained new PhyTips earlier, I would have gotten more successful trials of purified IgG. Another possible error of why I kept getting streaky gels was that there may have been precipitation of the sample or the voltage/current was too high. Therefore, after I began to centrifuge the samples before loading and using a lower voltage of 170, there were less streaks of protein bands on the gel. This experiment is useful because often times, pure IgG must be used in ELISAs to determine the protein or antibody concentrations of a particular sample. Therefore, in order to detect only the protein that reacts with the IgG, it must be pure. There can’t be any other proteins in the sample that may affect with the results of the ELISA. Follow up experiments can include testing the purity of the IgG using ELISAs or Western Blots. One may also optimize the serum concentration used for purification and the number of cycles through the Protein A PhyTip. Another follow up could be to prepare Protein A Agarose columns from scratch and to compare the purification techniques.