More Info - Vivione Biosciences

susceptiSTAT – UTI™
Overview
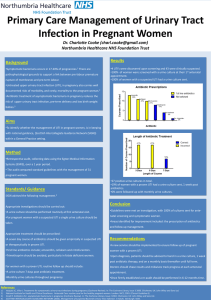
susceptiSTAT - UTI™ is a rapid antimicrobial susceptibility assay meant to rapidly diagnose urinary tract infections (UTI) and optimize upfront empiric therapy in UTI patients by use of the RAPID-B platform.
Current Methods
Urinary tract infection represent the highest volume microbiologic test performed in U.S. and European hospitals and is estimated to be approximately 35% of all microbiology performed. Epidemiology has determined approximately 7 million cases of cystitis in the U.S., with 1 million ED visits and 100,000 hospitalizations.
Internal marketing defined that an average medium to large sized hospital will perform 100-150/day. The cost to the U.S. healthcare system has been estimated at
$1.6 billion annually (Wilson CID, 2004:38 1150).
Traditional microbiologic plating methods are the predominant technology used to quantitatively determine the presence of bacteria in normally sterile urine samples.
This process is both laborious and slow in providing results, taking between 24-48 hours. Drug susceptibility testing takes, on average, another 18-48 hours. There is currently no rapid antibiotic susceptibility technology on the market for UTI. susceptiSTAT – UTI
The susceptiSTAT – UTI™ test aims to provide clinicians rapid, quantitative assessment of bacteria in urine as well as a rapid antibiotic susceptibility profile of the most common empiric antibiotics used to treat UTI in under 3 hours. The test will provide quantitative reductions in bacterial growth due to standard empiric antimicrobial therapies relative to a control sample with no antibiotics in the growth media. These results will help choose appropriate therapy for the patient and lower the risk of creating drug-resistant pathogens in both the hospital and community settings.
Therapies Assessed by susceptiSTAT- UTI™
Class
Sulfa
Nitrofuran
Fluoroquinolone
Β-lactam
Aminoglycoside
Drug
TMP-SMX
Ciprofloxacin
Nitrofurantoin
Amoxacillin
Gentamycin
Market Landscape
The market opportunity for urinary tract infections screening/testing is divided into two major areas: outpatient care and the nosocomial, or hospitalized patient. In most instances the outpatient is treated empirically, or simply with minimal or no testing performed. Treatment is based on general knowledge regarding efficacy of the major causative pathogens seen in UTI (e. coli in >80% of cases).
Hospitalized patients, many who are catheterized, represent a different approach.
Confirmatory testing via culture plating is the predominant methodology used for diagnosing UTI. There are several competing technologies that count bacteria, albeit without any drug susceptibility investigation. Susceptibility testing, in most circumstances, is performed via disc diffusion methodologies or with newer overnight automated systems such as the VITEK system manufactured by
Biomerieux.
While the disposable cost of culture media and plates is low, the expense and time of personnel adds a significant expenditure to pathology lab budgets. Estimates on total expenditures, both in the U.S. and the U.K. for urine testing is around $50.00/ sample.
The ability to have rapid, quantifiable results that incorporate drug susceptibility testing opens the market to the outpatient clinic due to the increasing rates of resistance for the traditional empiric antibiotics.
Competition
Currently there are no technologies that provide both rapid, quantifiable bacteria analysis and antibiotic susceptibility screening/testing.