CHEM162PracticeExamI..
advertisement

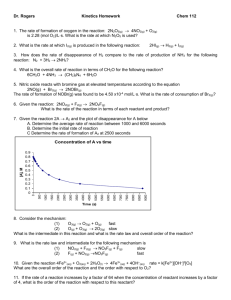
Practice Exam I (Tro chapters 10, 11, 13) Dr. Overway CHEM 162 Here are some things you may need. Formal Charge = # of valence e - - # of non-bonding e - - # of bonds Formal Charge = # of valence e - - # of non-bonding e - - ½ (# of bonding e - ) R= 8.3145 J/(K mol) R=0.08206 (Latm)/(Kmol) Psolv = Xsolv Posolv Kb (water) = 0.512 C kg/mol Kf (water) = 1.858 C kg/mol T = Kf cm ·i T = Kb cm ·i = MRT·I 𝑙𝑛[A] = 𝑙𝑛[𝐴]0 − 𝑘𝑡 𝑘 𝑅 𝑙𝑛 ( 2 ) 𝑘1 𝐸𝑎 = 1 1 − 𝑇1 𝑇2 𝐸 1 1 𝑘2 [ 𝑎 ( − )] = 𝑒 𝑅 𝑇1 𝑇2 𝑘1 E=hc/λ h=6.62610-34 Js 𝑡1/2 = c=2.9979108 m/s Avogadro’s number = 6.022×1023 things/mol 𝑡1/2 = 1 𝑙𝑛 (2) −𝑘 1 𝑘[A]0 −𝐸𝑎 𝑘 = 𝐴𝑒 𝑅𝑇 1 1 = + 𝑘𝑡 [A] [A]0 𝐸𝑎 1 ln 𝑘 = ln 𝐴 − ( ) ( ) 𝑅 𝑇 CHEM 162 Practice Exam 1. Match each of the following terms with the correct definitions. Answer Term Definition polymorph A a highly ordered solid allotrope B a solid substance whose molecules are held together by covalent bonds ionic solid C a solid substance whose molecules are held together by metallic bonds network covalent solid D the different crystal structures in which an element can exist metallic solid E a crystal structure the containing an atom at each of the corners of the cube and an atom at each face of the cube molecular solid F a solid substance whose molecules are held together by ionic bonds crystalline solid G a substance that has very little order amorphous solid H a substance that bursts into flame when in contact with the air I the different crystal structures in which a compound can exist J a solid substance whose molecules are held together by H-bonds, dipole-dipole forces, or dispersion forces 2. In each of the following comparisons, circle the correct compound. highest boiling point: CH4 CH3CH2OH lowest viscosity: H2O CH3CH2CH3 highest vapor pressure at 25 C: PH3 NH3 highest boiling point: H2S H2O lowest freezing point: SF6 CH3CH2CH2CH2CH3 3. Determine whether each of the following statements are true (T) or false (F) concerning the two phase diagrams shown below. Substance C is less dense as a liquid than a solid. At pt. 1 substance D is a supercritical fluid. Going from pt. 2 to pt. 3 in the phase diagram for substance D involves a phase change from gas to solid. At 1 atm of pressure substance D melts and substance C sublimes if the temperature is increased. 4. Of the intermolecular forces listed in the textbox to the right, which would be the strongest force that would have to be overcome in order to convert the following molecules from a liquid to a gas? a. CCl4 ______________________________. A) Ionic b. HBr ______________________________. B) Covalent C) Dispersion c. H2O ______________________________. D) Dipolar d. H2NCH2CH2CH3 ______________________________. E) Metallic e. CH3CH2CH2CH3 F) Hydrogen bonding ______________________________. 5. Circle the aqueous solution that should have the highest boiling point. Explain your answer. A. 1.0 M KNO3 (electrolyte) B. 0.75 M NaCl (electrolyte) C. 0.75 M CuCl2 (electrolyte) D. 2.0 M C12H22O11 (sucrose, non-electrolyte) 6. (8 pts) Dimethylglyoxime, DMG, is an organic compound used to test for aqueous nickel(II) ions. A solution prepared by dissolving 65.0 g of DMG (a non-electrolyte) in 375 g of ethanol boils at 80.3°C. What is the molar mass of DMG? (Kb = 1.22 C /molal, boiling point of pure ethanol = 78.5°C) 7. Lysine is an amino acid that is an essential part of nutrition but which is not synthesized by the human body. What is the molar mass of lysine if 750.0 mL of a solution containing 8.60 g of lysine (a non-electrolyte) has an osmotic pressure of 1.918 atm? (T = 25.0°C) 8. Draw the band gap structure for 1) an insulator and 2) a conductor. Label all of the parts. 9. Given a Rate Law of Rate=k[A]2[B]1, if you wanted to increase the rate of the reaction in the most efficient way, which chemical would you increase in concentration knowing that both A and B are very expensive? Explain. 2 N2O5 (g) 4 NO2 (g) + O2 (g) 10. The following data was measured for the reaction: Time (s) [N2O5] (M) 0 0.100 50.0 0.0707 100.0 0.0500 From the above data, the following graphs were made: A) Write the rate law for the reaction B) Calculate the rate constant (k) and provide the correct units. C) Propose a mechanism that is consistent with the rate law 200.0 0.0250 300.0 0.0125 400.0 0.00625 11. Radioactive materials, such as 60Co, are often used in biological studies. A radiation biologist studied the rate of decomposition of 60Co and obtains the following data: Time (min) 0 30.0 60.0 90.0 180.0 60 Co (µg) 15.4 11.66 8.84 6.69 2.91 A) What reaction order do nuclear reactions follow? B) Determine the rate constant with proper units. C) Determine the half-life of 60Co. 12. True/False (T or F) The slowest step in a mechanism has the smallest activation energy (Ea) The solid catalyst in an exhaust system of a car is a heterogeneous catalyst As the activation energy (Ea) decreases, the rate (speed) of a reaction decreases Transition states (activated complexes) are fairly stable and can be found in the valleys of a reaction diagram If the equilibrium constant (Keq ) of a reaction is large, the reaction is very fast If the rate constant (k) of a reaction is large, the reaction is very fast 13. Determine the rate law for each of the following overall reactions: A) C2H4 (g) + Br2 (g) C2H4Br2 (g) Rate = ____________________ B) 2 ClO2 (aq) + 2 OH-(aq) ClO3-(aq) + ClO2-(aq) + H2O (l) Rate = ____________________ 14. For the overall reaction of C2H4 (g) + H2 (g) C2H6 (g) , draw a reaction diagram for the uncatalyzed and catalyzed reactions - that makes two reaction paths on the same chart. Label the E, the activation energy (Ea), reaction intermediate(s), and the transitions state(s), axes labels, reactants, products. uncatalyzed reaction: step 1: C2H4 (g) + H2 (g) C2H6 (g) Ea uncat = 200 kJ/mol Erxn uncat = + 60 kJ/mol catalyzed reaction: step 1 (slow): C2H4 (g) + Pd(s) C2H4Pd (ad) Ea cat 1 = 80 kJ/mol Erxn cat1 = + 30 kJ/mol step 2 (fast): C2H4Pd (ad) + H2 (g) C2H6 (g) + Pd(s) Ea cat 2 = 40 kJ/mol Erxn cat 2 = + 30 kJ/mol 15. Write the Rate Law for the following mechanisms A) step 1: step 2: NO2 (g) + F2 (g) NO2F(g) + F(g) NO2(g) + F(g) NO2F(g) fast slow Rate Law = ______________________________ B) step 1: step 2: NO2 (g) + F2(g) NO2F(g) + F(g) NO2(g) + F(g) NO2F(g) slow fast Rate Law = ______________________________ C) step 1: step 2: F2(g) F(g) + F(g) 2 NO2(g) + 2 F(g) 2 NO2F(g) slow fast Rate Law = ______________________________ 16. For the reaction: H2O2 (aq) + 3 I-(aq) + 2 H+(aq) I3- (aq) + 2 H2O(l) The following initial rate data were obtained: Initial Concentrations Exp. # [H2O2] (M) [I-] (M) [H+] (M) Initial Rate (M/sec) 1 0.010 0.010 0.00050 1.1510-6 2 0.020 0.010 0.00050 2.3010-6 3 0.010 0.020 0.00050 2.3010-6 4 0.010 0.010 0.00100 1.1510-6 Write the specific rate law for the reaction. What is the rate constant k (with proper units)? 18. Answer True (T) or False (F) for the statements below. ___ a catalyst increases the activation energy of a reaction ___ a catalyst decreases the equilibrium constant (Keq) of a reaction ___ a catalyst appears in the overall chemical reaction ___ the solid catalytic converter of an automobile that converts NO(g) and CO(g) to N2 (g) and CO2 (g) is a homogeneous catalyst 19. List two ways in which a reaction rate can be affected. Provide specific detail and give an example (for example: if you increase the __, the reaction rate will increase (or decrease) because...)