Lec 32: Citric Acid Cycle & Fatty Acid Oxidation.
advertisement

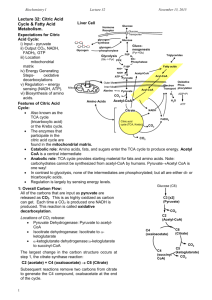
Biochemistry I, Spring Term Lec 32: Citric Acid Cycle & Fatty Acid Oxidation. Expectations: Lecture 32 Liver Cell O O Glycogen ii) Output Glucose Transporter glycogen synthase O O Gluconeogenesis F6P Glycolysis iii) Location Lactate Transporter Glucose glycogen phosphorylase (Glu - Pyr) Lactate (from anerobic met) Glucose Hormone Receptor O i) Input April 10, 2013 (Pyr-Glu) Triglycerides bisPhosphatase PFK Fatty acids F16P iv) Energy Gen Steps NAD+ NAD+ NADH v) Regulation NADH Acyl-CoA Pyruvate Outer Mem Mitochondria General Features; Inner Mem Alanine Acyl-CoA O Oxidative Phosphorylation ADP+Pi 2 CO2 Fatty Acid Also known as the Oxidation H2O TCA cycle Acetyl-CoA Amino Acids ATP (tricarboxylic acid) or the Krebs cycle. NADH Citrate The enzymes that participate in the citric FADH2 Citric Acid acid cycle are found in CO2 Cycle the mitochondrial matrix. Catabolic role: CO2 Amino acids, fats, and sugars enter the TCA cycle to produce energy. Acetyl CoA is a central intermediate Anabolic role: TCA cycle provides starting material for fats and amino acids. Note: carbohydrates cannot be synthesized from acetyl-CoA by humans. PyruvateAcetyl CoA is one way! In contrast to glycolysis, none of the intermediates are phosphorylated; but all are either di- or tricarboxylic acids. Regulation is largely by sensing energy levels. 1: Overall Carbon Flow: All of the carbons that are input as pyruvate are released as CO2. This is as highly oxidized as carbon can get. Each time a CO2 is produced one NADH is produced. This reaction is called oxidative decarboxylation. Locations of CO2 release: 1. Pyruvate Dehydrogenase: Pyruvate to AcetylCoA 2. Isocitrate dehydrogenase: Isocitrate to ketoglutarate 3. -ketoglutarate dehydrogenase:-ketoglutarate to succinyl-CoA The largest change in the carbon structure occurs at step 1: The citrate synthase reaction catalyzes the following reaction: C2 (acetate) + C4 (oxaloacetate) C6 (Citrate). Subsequent reactions remove two carbons from citrate to generate the C4 compound. Note that these carbons are not the same as those added. 1 Pyruvate Electron Transport Glucose (C6) C3 [x2] (Pyruvate) CO2 C2 (Acetyl-CoA) C4 (oxaloacetate) C4 (succinyl CoA) C6 (Citrate) CO2 C5 (ketoglutarate) CO2 Biochemistry I, Spring Term Lecture 32 April 10, 2013 2. Energetics of the TCA Cycle: Glucose In contrast to glycolysis, most of the energetic currency is in the form of redox reactions, only a single ATP is produced/pyruvate while four NADH and one FADH2 are produced. Most of the energy from oxidation is of glucose is harvested in the TCA cycle. The TCA cycle is a slower but richer source of energy. 2a: Oxidative decarboxylations: These occur at three locations, leading to the loss of the three carbons from pyruvate. 1. Pyruvate dehydrogenase 2. Isocitrate dehydrogenase 3. -ketoglutarate dehydrogenase Pyruvate dehydrogenase: This reaction, as with α-ketoglutarate dehydrogense, produces a thio-ester as the oxidized species. The reaction proceeds in two steps, loss of the CO2 group, followed by oxidation of the aldehyde and formation of the thioester (The latter step is equivalent to the scheme for oxidation of glyceraldehyde-3-P in glycolysis, but occurs by a different mechanism here.) The thio-ester is formed between the oxidized product and Coenzyme A, to form acetyl-CoA. The thio-ester is the same oxidation state as a carboxylate. F6P PFK-1 bisPhosphatase-1 F 1,6 P O Pyr O H3C O NAD+, CoA CO2 C Oxaloacetate HO Malate C C O CoA Citrate CoA O Go=-31 kJ/mol OH OH OH C C Fumarate C C COOH C HO NADH OH O OH O 8 NADH =24 ATP 2 FADH2= 4 ATP 2 GTP = 2 ATP FADH2 OH O S C C O O Acetyl-CoA C [citrate synthase] OH C H3C C O O OH [Pyruvate dehydrogenase] NADH O O 2 ATP 2 NADH C C COOH OH O CO2 ATP NADH CoA O NADH C C COOH OH C [a-ketoglutarate OH C dehydrogenase] O succinyl-CoA S-CoA O O a-ketoglutarate C O OH [isocitrate dehydrogenase] CO2 GTP [succinate OH O dehydrogenase] C C C Succinate OH iso-citrate OH NH2 O CO2 O NAD+ Pyruvate H3C N O S H OH O O H3C O H3C O NADH NH2 N N H H N O O HS O N N H H OH O P O coenzymeA (CoA) O O P O O P N O O N N O OH O3PO N O O3PO Thioesters in Biochemical Reactions: i) The relatively weak thioester bond facilitates the transfer of the acetyl group to other compounds, such as transfer of the acetyl group to oxaloacetate in the citrate synthase reaction. 2 N P O OH O Acetyl-CoA N O O OH O O C O C OH C C OH O Oxaloacetate H3C S CoA O C HO C OH C C H2 C OH O Citrate O OH Biochemistry I, Spring Term Lecture 32 April 10, 2013 ii) Acetyl-CoA or succinyl CoA are high energy compounds, capable of driving the synthesis of GTP, such as in the succinyl CoA synthetase in the citric acid cycle. GTP can be converted to ATP at no energy cost. HO O O P O O P O O CH2 O O O P O CH2 O O O O N O N O OO P O NH NH2 N O P O O O O O P O O N NH2 N OH OH CH2 O O FADH2 O OH CH3 N H N CH3 CH3 N N H COH COH COH CH2 O O P NH2 O O N N O P H O O 2 C O N N O H H O OH Succinate N HN COH COH COH 2e- + 2 H+ CH2 O O P NH2 O O N N O P H O O 2 C O N N REDOX HH O CH3 N HN 1. Oxidation of succinate to fumarate reduces FAD to FADH2. Alkane → Alkene FAD Succinate GTP Alkane → Alkene → Alcohol → Ketone OH OH OH OH GDP REDOX O CH2 O 2b. The remaining section of the pathway, from succinate to oxaloacetate follows a classic three step oxidation scheme that is also seen in fatty acid oxidation: HH HO NH S-CoA Succinyl-CoA O N HO O OH Fumarate OR OR HO FADH2 (Reduced) FAD (Oxidized) 2. Addition of water to the double bond, to make the alcohol. Alkene → Alcohol O OH O H2O H H Glucose OH H H HO PFK-1 Fructose-1,6-P O OH Malate O OH Fumarate Fructose-6-P bisPhosphatase-1 O Pyr + 3. Oxidation of Malate to Oxaloacetate reduces NAD to NADH. Alcohol → Ketone O OH NAD+ NADH O CO2 O CH2 Oxaloacetate O O OH Malate [Pyruvate dehydrogenase] NADH OH O CH2 O NAD+, CoA H HO O H3C O OH Oxaloacetate C C O OH O Acetyl-CoA C CoA H3C S CoA O C OH [Citrate synthase] HO OH C C COOH C Citrate O 3. Regulation of the TCA Cycle: OH COOH 1. High energy, irreversible steps are regulated. Succinyl-CoA 2. Regulated reactions are at the "top" of the pathway. Examples of: 1. Product Inhibition. 2. Allosteric inhibition by feedback inhibition by products 'down stream' in the pathway. Energy sensing is the most important regulatory control of the TCA cycle (I=inhibited) High Energy OTHER Step NADH ATP Compound Prod. Inh Feedback Inh Pyruvate I I Inhibited by Acetyl CoDehydrogenase A Citrate Synthase I I Inh. by succinyl-CoA CH2 CH2 O S-CoA Inhibited by citrate Role of citrate in regulation of glycolysis: Citrate stabilizes the tense-form of PFK, shutting down glycolysis. 3 Biochemistry I, Spring Term Lecture 32 April 10, 2013 Fatty acids Fatty Acid Oxidation (-Oxidation): A. Formation of Acyl-CoA (Cytosol): The fatty acids in the Acyl-CoA cytosol are coupled to coenzyme A to form acyl-CoA. The activation reaction is catalyzed by acyl-CoA synthetase and involves the hydrolysis of ATP to AMP, i.e. the equivalent of two high energy ATP molecules (60 kJ/mol). The released pyrophosphate is hydrolyzed to inorganic phosphate, making the overall ΔG negative for the rection (indirect coupling) Note: it is only necessary to utilize ATP once in the activation of the fatty acid. O O O O HS O O P N N O O P O Fatty Acid activation electron transport Acyl-CoA Pyruvate CO2 Fatty Acid Oxidation Acetyl-CoA FADH2 NADH Citrate Citric Acid Cycle Adenine CO2 O O OH H H O triglycerides CO2 CoA S ATP AMP O O P O Acyl-CoA: Length N O P H2O H O H O O O 2ATP 2ADP O OH O3PO O O O H3C O P S H H O FAD O O P O B. Transport into mitochondria: The H acyl-CoA is transported into the mitochondrial matrix for oxidation. This location is ideal for funneling the products of oxidation (NADH and FADH2) to E. transport. H3C FADH2 O CoA S H H2O C. -Oxidation (Mito. matrix): Alkane → Alkene → Alcohol → Ketone REDOX OH O H3C S REDOX O O P O O HO CoA Additional Cycles NAD+ O Adenine CoA O P O O O O OH N N H H O Coenzyme A O3PO NADH SH O H3C Acyl-CoA is shortened 2 carbons at a time from the carboxyl end of the fatty acid using the following steps: S CoA O N-2 carbons (shrinking H3C chain) O CoA 1. Formation of trans - double + HC S S 3 bond by acyl-CoA dehydrogenase, an FAD enzyme. 2. Addition of water to the newly formed double bond to generate the alcohol by enoyl-CoA hydratase 3. Oxidation of the alcohol by NAD+ to give the ketone, catalyzed by 3-L-hydroxyacyl-CoA dehydrogenase. 4. Cleavage reaction by -ketoacy-CoA thiolase (thiolysis), generates acetyl-CoA and an acyl-CoA that is two carbons shorter. The acetyl-CoA enters the TCA cycle. 5. Steps 1-4 are repeated until only acetyl-CoA remains. H3C H3C O O S CoA H3C CoA 4 O O Oxidation of C6 Fatty Acid CoA S O S CoA CoA H3C H3C O O S CoA H3C CoA O S CoA O S CoA H3C S CoA Biochemistry I, Spring Term 5 Lecture 32 April 10, 2013