Water Isotopes Time Machine

Water Isotopes Time Machine
Part of:
Inquiry Science with Dartmouth
Developed by: Gifford Wong, PhD candidate,
Department of Earth Sciences
Inspired by: David Noone (Oregon State University) and his
Summit Station (Greenland) Summer Science Lecture (2014)
Overview
Ice cores are cylinder-shaped samples of ice drilled from a glacier. Similar to tree rings, ice core records provide a direct and detailed way to study past climate. One reason ice cores present such great records of past climate is the accuracy by which we can date an ice core. Scientists can analyze for soluble and insoluble chemical constituents as well as the isotopic composition of the ice, all of which typically vary depending upon the season in which the snow fell. Stable isotope ratios of water (hydrogen and oxygen), which vary depending on temperature, can reveal warmer (summer) and colder (winter) periods of the year.
In this lesson, students will review the structure of an atom, molecule and isotope. Students will then be introduced to the physical factors controlling heavier and lighter isotopes, developing (or augmenting) a conceptual understanding of the hydrological cycle. Finally, students will apply their newly acquired understanding to interpret stable water isotope data from an ice core taken from the Greenland Ice Sheet.
This last exercise can be done by hand (or in Excel) to practice making plots using real data.
Science Standards
Next Generation Science Standards (NGSS):
MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures.
MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance when thermal energy is added or removed.
MS-ESS2-4. Develop a model to describe the cycling of water through Earth’s systems driven by energy from the sun and the force of gravity.
MS-ESS3-5. Ask questions to clarify evidence of the factors that have caused the rise in global temperatures over the past.
New Hampshire Curriculum Framework:
SPS2-4. Patterns of change
SPS3. Collaborations in scientific endeavors
SPS4-2. Communication skills
Focus Question
What information do stable water isotope ratios (measured) in ice cores tell us, and how can we use their behavior to ask questions about how climate has changed in the past?
Objectives
Through this lesson, students will:
Develop conceptual models of stable isotopes of water.
Observe how the properties of stable isotopes of water impact how readily they move through the water cycle.
Receive “hands-on” experience creating graphs that illustrate the cyclical nature of water isotopes in a Greenland ice core record.
Understand the magnitude of change seen in the seasons, and compare that with changes seen in stable isotopes of water going back through the last ~2000 years.
Background
Students should have a basic understanding of matter (e.g., atom, molecule, compound), as well as have had an introduction to the periodic table (e.g., element, atomic number, atomic mass) and the states of matter (e.g., solid, liquid, gas). Students will build upon this foundation as this lesson introduces the concept of an isotope.
Originally presented on December 5, 2014, students were reminded of matter (atom, molecule), states of matter (liquid, gas, solid), and the periodic table (# protons characterize an element, # electrons balance the charge). Here we introduced the concept of an isotope (# neutrons). We outlined our intended “three take-aways”: 1) heavier things (water) require more energy (sun) to move compared with lighter things,
2) scientists can take advantage of cyclical phenomena (e.g., seasons) to refine their understanding of the system they are studying (e.g., climate in Polar region), and 3) small changes (in average annual ratio of stable water isotopes) can represent very big changes in climate.
The first part of this lesson can be expanded to fill an entire day. Similarly, the incorporation of real-time weather data as a secondary component could enhance the graphing segment of this activity. The goals of this lesson are to introduce two ways the ratios of stable water isotopes are used in ice core science. They respond to temperature, with one isotope more representative of environmental conditions of the ice core site (δ 18 O) and another more representative of source environmental conditions (δ 17 O). In analyzing the
δ 18 O record (which this activity focuses on), you can assign accurate ages to ice core depth based on the number of summers (higher values of δ 18 O) you count in your data. Similarly, over longer reaches of time, you can see periods of time when your site was relatively more warm or cold, such as the time when
Vikings occupied Greenland (Medieval Warm Period) and when they decided to vacate (Little Ice Age).
Materials
1.
graph paper (Excel is an option, but not necessary; Google Sheets is also great)
2.
spreadsheet with the δ 18 O data. We split the class into teams of 2 or 3 students, and assigned each team a row of ~25 data points. You can also parse out the data further (or procure a longer record) and have each student graph their own portion of an ice core.
a.
After all groups are done, we had students tape the plots in sequential order and observe/discuss (and possibly count) all of the peaks (or valleys) in the signal. i.
Q: “Are they all summers?” If not, what else could a peak be interpreted as?
[possibly a “warm” storm in the middle of a usually-cold month]
3.
weights (approximately 10-20 pounds for each hand, and likely 4 such weights would be sufficient); more would allow more students the opportunity to “experience” the effect a.
I made mine from canned goods and fabric shopping bags
Preparation
No specific preparation is required for this activity (other than printing out enough copies), but it is highly recommended that teachers run through the lab and presentation ahead of time so they can get a feel of the timing of the lab.
Procedure
1.
Introduction: Begin class with a refresher on matter (atoms and molecules). The accompanying
PowerPoint presentation provides one option for this that also references the Periodic Table, or feel free to utilize your own previous material. a.
Introduce the concept of an isotope using water as the model molecule. i.
If desired, introduce (or refresh) how the listed atomic mass of an element is really an average atomic mass that incorporates all of the known isotopes of an element and is based on the isotopic mass and percent abundance. b.
Water may have one or two “heavy” hydrogens ( 2 H) and/or one “heavy” oxygen ( 17 O or 18 O). c.
While abundances of the heavier water molecules are quite small, sensitive instruments allow us the opportunity to observe differences in relative abundances of stable isotopes of water due to kinetic fractionation (see http://en.wikipedia.org/wiki/Kinetic_fractionation ) occurring during evaporation/condensation. These changes occur at many temporal scales: daily to seasonal to decadal/centennial. d.
Demonstration : Recruit 2 volunteers (more if you have enough “weights”). Pass out weights (e.g., shopping bags with canned goods, reams of paper, dumbbells) with one student(s) representing “heavier” water (e.g., H
2
18 O) and the other(s) representing
“regular” water (H
2
16 O). The weights should be heavy enough such that the student(s) will tire out if they held them away from their bodies with straight arms. i.
Option 1 : Have all the volunteer students sit on the ground. On cue, have the students all stand up and raise the weights so that their straightened arms are parallel to the ground. Ideally, the heavier weights will take a little longer to stand up (kinetic fractionation). If anyone’s arms start to droop, they must sit down. Again, ideally the heavier weights will not last as long as the “regular” water (kinetic fractionation). ii.
Option 2 : Have all the volunteer students stand with their arms parallel to the ground. Play music (something danceable) and have the volunteers dance.
While they can bounce up and down, they must “piston” their arms keeping them raised (parallel to the ground). Ideally, the heavier weights will need to stop dancing before the “regular” water dancers. iii.
Explanation : Both options demonstrate how water molecules respond to external stimuli (energy via the sun). The first more explicitly describes the evaporation/condensation tendencies of heavier v. “regular” water molecules.
The second implicitly covers that, but more directly demonstrates the condensation component (see http://www.scientificamerican.com/article/how-are-past-temperatures/ ). e.
Concept from demonstration : The ratio of heavier to “regular” water molecules
(δ 18 O) can vary, and that δ 18 O can be used to reflect relative temperatures at the site of deposition. This is because water molecules are well-preserved in polar regions by way of ice cores, and ice cores are the medium by which we extract climate information for analysis and interpretation. A higher relative ratio of heavier to regular (δ 18 O) water molecules generally indicates warmer temperatures for the ice core site in question due to the correlation between temperature and evaporation. By studying the variation in these ratios over time, scientists can assign specific years to
specific depths (i.e., count the “summers” contained in an ice core) as well as demonstrate when in our history were temperatures relatively cooler or warmer to now. f.
“Now that we know (approximately) what controls δ 18 O, let’s look at some data.”
2.
Visualizing data: The provided data are from a shallow ice core drilled in northwest
Greenland. The core, drilled in 2011, is ~21 m deep and goes back to 1990. The data are split such that 8 groups of students (paired-up) can plot ~50 data points. a.
Have students plot their subset of the data on a piece of graph paper. For the sake of consistency, have student groups space their x-axis “sample number” on the same scale as well as their y-axis (δ 18 O value). Isotope ratios range from -20 (δ 18 O; max) and -40 (δ 18 O; min). Once all groups are done, have the students “stitch” the separate plots together so that the class can visualize the whole of the stable water isotopes dataset. b.
Notice the regular pattern of relatively high and low ratios of δ 18 O. Discuss with the class what environmental signal produces regular patterns. There would be a diurnal cycle, but these data are too coarse to possible visualize that pattern. There is a seasonal cycle (summer/winter), and that pattern is being shown with the ice core data. c.
Be aware : time moves “backwards” along the x-axis. The original data (δ 18 O) is plotted against depth, which approximates time. The deeper you go in an ice sheet the further back in time you “travel”. In the example plot (Excel spreadsheet), the δ 18 O is plotted against depth, with “0m” representing the surface. If you were to label each peak as a summer, then the first peak would be the summer of 2010, and each subsequent peak would be the previous summer, hence the “time moving backwards” x-axis.
3.
Going back in time [how does knowledge of current water isotope trends help us interpret water isotope data going back thousands and tens of thousands of years ago?]: The remainder of the included PowerPoint presentation shows how measured δ 18 O in a deep ice core
Greenland changed through time. a.
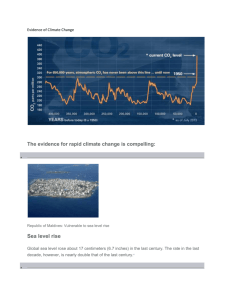
The figure shows how δ 18 O has changed over the last ~80,000 years. Higher values of
δ 18 O indicate for warmer times. b.
Agrarian societies first developed ~10,000 years ago, when it appears the δ 18 O trend is flat or “stable”. c.
Zooming in on the most recent ~2,000 years, you can see that in spite of the relatively stable δ 18 O data, there are still discernible higher and lower values, which equate to relatively warmer or cooler periods. d.
A specific example is the period of time when Vikings first arrived in Greenland, and when they were forced to abandon Greenland. These events coincided with warm
(Medieval Warm Period) and cold (Little Ice Age) times, as recorded by δ 18 O (stable isotopes of water).
4.
Wrap-up: Have students discuss what the consequences might be if the world became much warmer or cooler relative to now. How does the experience of the Vikings compare with modern stories of civilizations, such as the Carteret Islanders of Papua New Guinea, having to move because of environmental changes? (see http://earthfirstjournal.org/newswire/2014/04/06/first-official-climate-change-refugeesevacuate-their-island-homes-for-good/ )
Assessment
Ask students to describe an isotope. Ask students to describe how stable isotopes of water (specifically
H
2
18 O) allow scientists to “go back in time” and explain relative changes in the Earth’s temperature.
Extensions
Groups of students may “count the years” (i.e., count how many summers there are in this record by counting the number of peaks) and compare them with the class (and my assertion this record extends from summer 2011 down to 1990). Longer records of δ 18 O exist from both hemispheres, such as those hosted at the National Snow and Ice Data Center ( link to data ), and more advanced students may download these data and graph them using Excel.
If part of a larger climate section, this module can be linked with Samantha Roberts’ “Using Real Data to
Test Hypotheses by Graphing in Excel” to expose students to other long-term climate data sets while also learning to visualize data using Excel.