Quant Lab 4
advertisement

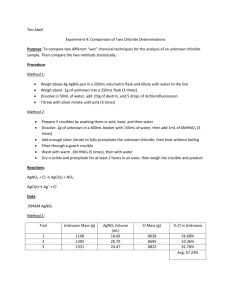
Experiment 4: Comparison of Two Chloride Determinations Introduction: To compare two different “wet” chemical techniques for the analysis of an unknown chloride sample. The first analysis is a volumetric chloride determination and the second is a gravimetric determination. Procedure: Volumetric Chloride Determination 1. 4.5g of AgNO3 was dissolved and diluted in a 250 mL volumetric flask. 2. Standardize the AgNO3 using oven dried sodium chloride. Weigh enough sodium chloride for a 25 mL titration. (0.155g NaCl) 3. Add 0.03 g of dextrin and 5 drops of dichlorofluoresein as an indicator. Titrate solution to a pink endpoint. Obtain three good trials. Gravimetric Chloride Determination 1. Clean three Gooch crucibles with an acid solution (0.1M HNO3) and then (0.1M NH3) basic. 2. Follow washes with distilled water and dry one hour in 120°C oven. Let crucibles cool and fit each crucible with a glass fiber filter. 3. Accurately weigh 0.2 g unknown chloride and dissolve each sample in a 400 mL beaker with 150 mL distilled water. Add 1 mL of 6M HNO3. 4. Add silver nitrate solution to completely precipitate the unknown chloride. Heat the solution on a hot plate without boiling and frequently stir until the supernatant is clear. 5. Vacuum filter the samples through weighed Gooch crucibles. Use warm 0.1M HNO3 to wash the precipitate five times, and then finally wash the precipitate with distilled water. 6. Dry the crucibles and precipitate in a 120°C oven for at least two hours before final weighing. Reactions: AgNO3 + Cl- AgCl (s) + NO3AgCl (s) Ag+ + ClData: Mass AgNO3= 4.5006g Amount dextrin 0.0328g 0.0336g 0.0390g Unknown chloride 0.1041g 0.1002g 0.1078g Amount AgNO3 solution 16.00mL 14.60mL 15.00mL Part 2 Data: Mass unknown chloride Amount AgNO3 added Mass crucible and filter paper Mass crucible, filter paper, and product Mass product Trial 1 0.2016g Trial 2 0.2028g Trial 3 0.2014g 7.3mL 21.1403g 9.5mL 22.3275g 8.4mL 22.6890g 21.3759g 22.6776g 22.9440g 0.2356g 0.3501g 0.2550g Calculated Data(Volumetric) Trial 1 2 3 M AgNO3 0.106 0.106 0.106 Mol AgNO3 & Cl0.0017 0.0015 0.00159 Mass Cl- in Precipitate (g) 0.0601 0.0532 0.0564 Mass % Cl- in Uknown 57.7% 53.1% 52.3% Calculated Data (Gravimetric & Corrected) Trial Mass Clin precip. (g) Mass % Clin unknown 1 2 3 0.0583 0.0866 0.0631 28.9% 42.7% 31.3% Theroetical AgNO3 needed (mL) 30.4 30.4 30.4 Fraction AgCl obtained Total mass AgCl (g) Total mass Cl- (g) 0.2401 0.3125 0.2763 0.9813 1.1203 0.9229 0.2427 0.2771 0.2283 Mass % Clin Unknown (corrected) 120.4% 136.6% 113.4% Calculations: Volumetric Determination Molarity of AgNO3 1 𝑚𝑜𝑙 𝐴𝑔𝑁𝑂3 𝑀 = 𝑚𝑎𝑠𝑠𝐴𝑔𝑁𝑂3 × ÷𝑉 169.873𝑔𝐴𝑔𝑁𝑂3 Moles of AgNO3 & Cl𝑀 𝐴𝑔𝑁𝑂3 × ∆𝑉 𝐴𝑔𝑁𝑂3 = 𝑚𝑜𝑙 𝐴𝑔𝑁𝑂3 & 𝐶𝑙 − Mass of Cl- in Precipitate 35.453𝑔𝐶𝑙 − 𝑚𝑜𝑙 𝐶𝑙 − × = 𝑚 𝐶𝑙 − 1 𝑚𝑜𝑙 𝐶𝑙 − Mass Percent of Cl- in Unknown 𝑚 𝐶𝑙 − × 100% = 𝑚𝑎𝑠𝑠 % 𝐶𝑙 − 𝑚 𝑖𝑛𝑖𝑡. 𝑠𝑎𝑚𝑝𝑙𝑒 Molarity of AgNO3 1 𝑚𝑜𝑙 ÷ 0.250𝐿 169.873𝑔 Moles of AgNO3 & Cl- Trial 1 0.106𝑀 × 0.01600𝐿 = 0.0017 𝑚𝑜𝑙 Mass of Cl- in Precipitate Trial 1 35.453𝑔𝐶𝑙 − 0.0017𝑚𝑜𝑙𝐶𝑙 − × = 0.0601𝑔𝐶𝑙 − 1 𝑚𝑜𝑙 𝐶𝑙 − Mass Percent of Cl- in Unknown Trial 1 0.0601𝑔 𝐶𝑙 − × 100% = 57.7% 𝐶𝑙 − 0.1041𝑔 𝑈𝐾 0.106𝑀 = 4.5006𝑔 × Gravimetric Determination Mass of AgCl (precipitate) 𝑚𝑓𝑖𝑛𝑎𝑙 − 𝑚𝑐𝑟𝑢𝑐. − 𝑚𝑓 𝑝𝑎𝑝𝑒𝑟 = 𝑚 𝐴𝑔𝐶𝑙 Mass of Cl- in precipitate 1 𝑚𝑜𝑙 1 𝑚𝑜𝑙 𝐶𝑙 − 𝑚 𝐴𝑔𝐶𝑙 × × 143.321𝑔 1 𝑚𝑜𝑙 𝐴𝑔𝐶𝑙 35.453𝑔 × = 𝑚 𝐶𝑙 − 1 𝑚𝑜𝑙 Mass Percent of Cl- in Unknown 𝑚 𝐶𝑙 − × 100% = 𝑚𝑎𝑠𝑠 % 𝐶𝑙 − 𝑚 𝑖𝑛𝑖𝑡. 𝑠𝑎𝑚𝑝𝑙𝑒 Correction Gravimetric Determination Data Theoretical amount of AgNO3 needed ̅̅̅̅ AgNO3 × 2 ≅ 𝑉 𝐴𝑔𝑁𝑂3 𝑛𝑒𝑒𝑑𝑒𝑑 ∆V Fraction of AgCl obtained 𝑉 𝐴𝑔𝑁𝑂3 𝑢𝑠𝑒𝑑 𝑉 𝐴𝑔𝑁𝑂3 𝑛𝑒𝑒𝑑𝑒𝑑 = 𝑓𝑟𝑎𝑐𝑡𝑖𝑜𝑛 𝑜𝑓 𝐴𝑔𝐶𝑙 𝑜𝑏𝑡𝑎𝑖𝑛𝑒𝑑 Total mass of AgCl 𝑚 𝐴𝑔𝐶𝑙 𝑜𝑏𝑡𝑎𝑖𝑛𝑒𝑑 = 𝑡𝑜𝑡𝑎𝑙 𝑚 𝐴𝑔𝐶𝑙 𝑓𝑟𝑎𝑐𝑡𝑖𝑜𝑛 𝑜𝑓 𝐴𝑔𝐶𝑙 𝑜𝑏𝑡𝑎𝑖𝑛𝑒𝑑 Total mass of Cl1 𝑚𝑜𝑙 1 𝑚𝑜𝑙 𝐶𝑙 − 𝑚𝑡𝑜𝑡 𝐴𝑔𝐶𝑙 × × 143.321𝑔 1 𝑚𝑜𝑙 𝐴𝑔𝐶𝑙 35.453𝑔 × = 𝑚 𝐶𝑙 − 1 𝑚𝑜𝑙 Mass Percent of Cl- in Unknown 𝑚 𝐶𝑙 − × 100% = 𝑚𝑎𝑠𝑠 % 𝐶𝑙 − 𝑚 𝑖𝑛𝑖𝑡. 𝑠𝑎𝑚𝑝𝑙𝑒 t calc = Mass of AgCl (precipitate) Trial 1 21.3759𝑔 − 21.1403𝑔 = 0.2356𝑔 𝐴𝑔𝐶𝑙 Mass of Cl- in precipitate Trial 1 1 𝑚𝑜𝑙 1 𝑚𝑜𝑙 𝐶𝑙 − 0.2356𝑔 × × 143.321𝑔 1 𝑚𝑜𝑙 𝐴𝑔𝐶𝑙 35.453𝑔 × = 0.0583𝑔 𝐶𝑙 − 1 𝑚𝑜𝑙 Mass Percent of Cl- in Unknown Trial 1 0.0583𝑔 𝐶𝑙 − × 100% = 28.91% 𝐶𝑙 − 0.2016𝑔 𝑈𝐾 Theoretical amount of AgNO3 needed Trial 1 15.20mL AgNO3 × 2 ≅ 30.40𝑚𝐿 𝐴𝑔𝑁𝑂3 Fraction of AgCl obtained Trial 1 0.0073𝐿 𝐴𝑔𝑁𝑂3 𝑢𝑠𝑒𝑑 0.03040𝐿 𝐴𝑔𝑁𝑂3 𝑛𝑒𝑒𝑑𝑒𝑑 = .2401 𝐴𝑔𝐶𝑙 𝑜𝑏𝑡𝑎𝑖𝑛𝑒𝑑 Total mass of AgCl Trial 1 0.2356𝑔 𝐴𝑔𝐶𝑙 𝑜𝑏𝑡𝑎𝑖𝑛𝑒𝑑 = .9813𝑔 𝐴𝑔𝐶𝑙 0.2401 𝐴𝑔𝐶𝑙 𝑜𝑏𝑡𝑎𝑖𝑛𝑒𝑑 Total mass of Cl- Trial 1 1 𝑚𝑜𝑙 1 𝑚𝑜𝑙 𝐶𝑙 − 0.9813𝑔 𝐴𝑔𝐶𝑙 × × 143.321𝑔 1 𝑚𝑜𝑙 𝐴𝑔𝐶𝑙 35.453𝑔 × = 0.2427𝑔 𝐶𝑙 − 1 𝑚𝑜𝑙 Mass Percent of Cl- in Unknown Trial 1 0.2427𝑔 𝐶𝑙 − × 100% = 120.4% 𝐶𝑙 − 0.2016𝑔 |𝑑̅ | √n 𝑠 Volumetric % Gravimetric % Difference 57.7 28.9 28.8 53.1 42.7 10.4 52.3 31.3 21.0 20.07 × √6 = 5.32 9.235 ttable for at 95% confidence interval is = 2.776 5.32 > 2.776 Discussion: 1. We compared data with the student’s t test. Both methods are fairly accurate and give similar results. However, the gravimetric data seems to be more precise than the volumetric. The mass percent of our Cl- is well over 100% which means we could have made an error during our experiment, and more trials should be done to test the unknown. 2. We performed a direct titration because the titrant was added directly to the known analyte. 3. The gravimetric determination was indirect because the analyte concentration was unknown. Conclusion: 1. To improve this experiment we could use a better indicator that gives a more distinctive color change to make sure we don’t over titrate. Another improvement could be to make sure that the crucibles were measured at the exact same temperature at the final weighing.