Supplementary Appendix Circulating presepsin (soluble CD14
advertisement

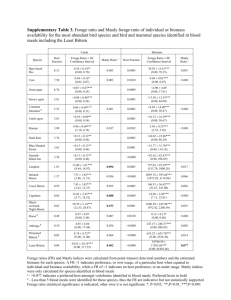
Supplementary Appendix Circulating presepsin (soluble CD14 subtype) as a marker of host response in patients with severe sepsis or septic shock. Data from the multicenter, randomized ALBIOS trial. Serge Masson; Pietro Caironi; Caterina Fanizza; Ralf Thomae; Roberto Bernasconi, Andrea Noto, Roberto Oggioni, Giovanni Stefano Pasetti, Marilena Romero; Gianni Tognoni; Roberto Latini; Luciano Gattinoni; on behalf of the ALBIOS Study Investigators§ 1 Supplementary Methods Study design, data collection, and definitions Inclusion and exclusion criteria, and study design of the ALBIOS trial have been published elsewhere [1], and are briefly summarized here. Patients aged 18 years or older who met clinical criteria for severe sepsis within the previous 24 hours and at any time during ICU stay, were enrolled in the ALBIOS trial. During the early phase of volume resuscitation (6-24 hours after study enrollment), fluids were administered in both groups according to the “early goal-directed therapy” protocol [2]. Primary outcome was all-cause mortality 28 days after randomization, and principal secondary outcome was all-cause mortality at 90 days. Additional secondary outcomes included the number and severity of organ dysfunctions, respectively defined as the number of both novel organ dysfunction and organ failure occurring during study period (based upon the SOFA score), and the average of the daily SOFA scores for each individual patient during the study period, ICU and hospital length of stay. Tertiary outcomes included the use of renal replacement therapy, the incidence of acute kidney injury, the duration of mechanical ventilation, the time to suspension of vasopressor or inotropic agents, the clinical resolution and duration of the primary infection, and the incidence of secondary infections, all during the study period. Clinical resolution of the primary infection, if any, was established by the treating physician according to standard clinical signs. Acquired secondary infections were defined as new infections other than the primary infection responsible for the development of severe sepsis, arising during the study, and evaluated by the treating physician. Immunodeficiency at the time of randomization was defined as the exposition to immunosuppressive therapy before study enrollment (such as immunosuppressive agents, steroids, chemotherapy, and radiative therapy), or the presence of a co-morbidity able to suppress defenses of the immune system (neoplasia, blood tumor, AIDS, etc.). Demographic, clinical, laboratory and microbiological data were collected on a daily basis. Overall severity of the systemic illness was assessed at study entry with the Simplified Acute Physiology Score SAPS II [3]. Organ function was rated daily using the Sequential Organ Failure Assessment (SOFA) score [4], including sub-scores from 0 to 4 for five components (lungs, coagulation, liver, circulation, kidney), with high scores indicating a high degree of organ dysfunction. Microbiological sampling, as well as routine surveillance cultures, were done at the discretion of the attending physician, and according to standard practice in each center. All the treatments applied with the exception of fluid management were at the discretion of the attending physician [1], who usually did not coincide with the local investigator. 2 Inclusion in the pre-defined biomarkers study A subset of 997 patients, recruited in 40 centers, participated to the predefined biomarkers sub-study. About 70% of the patients enrolled in the original ALBIOS trial from each of those centers participated also into the biomarker sub-study. Venous blood samples were serially collected 1, 2 and 7 days after enrolment (or at ICU discharge, whichever came first), centrifuged, and plasma was shipped on dry ice to a central repository and stored at -70°C until assayed. Additional statistical methods Multivariable linear regression analysis with backward elimination was performed to identify the clinical determinants independently associated with a high log-transformed concentration of presepsin on day 1. The analysis included continuous variables (age, heart rate, mean arterial pressure, central venous pressure, urine output, lactate, serum albumin, hemoglobin, serum creatinine, white blood cells, serum bilirubin, platelet count) and categorical variables (chronic renal failure, immunodeficiency, shock, primary site of infection, type of infection according to site culture, type of infection according to blood culture). The relation between presepsin concentration and mortality was first assessed with univariate logistic models entering log-transformed concentration of the biomarker as a continuous variable. The following covariates were then introduced into a multivariable model if they had a simple association with outcome with p<0.20: age, sex, body mass index, reason for admission in ICU, SAPS II score, SOFA score at baseline, appropriate antibiotic therapy, heart rate, mean arterial pressure at baseline or after 6 hours, central venous pressure, PaO 2/FiO2, urine output, serum concentration of lactate, albumin, hemoglobin, creatinine or bilirubin, platelet count, mechanical ventilation, COPD, chronic renal failure, immunodeficiency, congestive or ischemic heart disease, liver disease, shock. The relations between tertiary outcomes and presepsin concentration were analyzed with a multivariate logistic regression model and a linear regression model for categorical and continuous variables, respectively. The variables included in the model were age, immunodeficiency, shock, mechanical ventilation and SOFA score at baseline. Statistical analysis was done using SAS software, version 9.3 (SAS Institute). A two-sided p value of <0.05 was deemed statistically significant. 3 Supplementary References 1. Caironi P, Tognoni G, Masson S, Fumagalli R, Pesenti A, et al (2014). Albumin Replacement in Patients with Severe Sepsis or Septic Shock. N Engl J Med 370: 1412-1421. 2. Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, et al (2001). Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 345: 1368-1377. 3. Le Gall JR, Lemeshow S, Saulnier F (1993). A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 270: 2957-2963. 4. Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, et al (1996). The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22: 707-710. 4 Supplementary Table 1: Presepsin levels on day 1 and infection Variable Primary site of infection Lung Abdomen Urinary tract Other site More than one site Type of infection according to site culture Bacterial infection Purely gram-negative bacteria Purely gram-positive bacteria Mixed bacteria Culture negative Culture not obtained Other infection Type of infection according to blood culture Bacterial infection Purely gram-negative bacteria Purely gram-positive bacteria Mixed bacteria Culture negative Culture not obtained Other infection No. (%) 332 (34.7) 94 (9.8) 91 (9.5) 334 (34.9) 107 (11.2) 411 (42.9) 211 (22.0) 133 (13.9) 67 (7.0) 314 (32.8) 60 (6.3) 173 (18.0) 229 (23.9) 98 (10.2) 112 (11.7) 19 (2.0) 581 (60.6) 106 (11.1) 42 (4.4) Presepsin P (median [Q1-Q3], ng/L) <0.0001 958 [518-1832] 1200 [630-3309] 1224 [553-2821] 762 [397-1340] 982 [589-2405] 0.0003 1025 [558-2197] 1062 [557-2319] 972 [577-1863] 1097 [558-2160] 773 [388-1767] 735 [443-1529] 943 [549-1962] <0.0001 1178 [657-2623] 1398 [763-2774] 963 [559-2297] 1887 [813-3714] 868 [454-1589] 850 [357-1846] 1141 [549-2440] 5 Supplementary Table 2: Presepsin levels on day 1 and achievement of hemodynamic goals Presepsin on Day 1 All Tertile 1 (<597 ng/L) Tertile 2 (597-1397 ng/L) Tertile 3 (>1397 ng/L) No. (%) At randomization Mean arterial pressure ≥ 65 mmHg – no. (%) Vasoactive drugs – no. (%) Dopamine Norepinephrine Epinephrine Dobutamine Vasopressin 958 (100.0) 320 (33.4) 320 (33.4) 318 (33.2) 719 (75.1) 609 (63.6) 248 (25.9) 462 (48.2) 44 (4.6) 63 (6.6) 10 (1.0) 266 (83.1) 168 (52.5) 70 (21.9) 123 (38.4) 9 (2.8) 8 (2.5) 2 (0.63) 242 (75.6) 219 (68.4) 85 (26.6) 170 (53.1) 15 (4.7) 29 (9.1) 5 (1.6) 211 (66.4) 222 (69.8) 93 (29.3) 169 (53.1) 20 (6.3) 26 (8.2) 3 (0.9) <0.0001 <0.0001 0.03 0.0002 0.04 0.004 0.7 Two or more vasoactive drugs – no (%) Doses of vasoactive drugs - µg/kg/min 201 (21.0) 41 (12.8) 79 (24.7) 81 (25.5) <0.0001 8.27 ± 4.52 0.28 ± 0.27 0.15 ± 0.21 8.25 ± 3.96 0.25 ± 0.26 0.15 ± 0.18 7.52 ± 4.33 0.28 ± 0.27 0.11 ± 0.08 8.96 ± 5.00 0.29 ± 0.27 0.18 ±0.28 0.70 0.05 0.69 822 (85.8) 298 (93.1) 281 (87.8) 243 (76.4) <0.0001 205 (22.6) 398 (43.9) 304 (33.5) 612 (75.5) 79 (25.7) 140 (45.6) 88 (28.7) 217 (77.8) 71 (23.6) 137 (45.5) 93(30.9) 213 (77.7) 55 (18.4) 121 (40.5) 123 (41.1) 182 (70.5) 852 (89.9) 562 (60.9) 652 (68.8) 234 (24.7) 514 (54.2) 57 (6.0) 134 (14.1) 9 (0.9) 304 (95.6) 237 (76.7) 179 (56.3) 64 (20.1) 131 (41.2) 10 (3.1) 28 (8.8) 1 (0.3) 289 (91.2) 185 (59.9) 228 (71.9) 74 (23.3) 178 (56.2) 19 (6.0) 49 (15.5) 4 (1.3) 259 (82.8) 140 (45.9) 245 (78.3) 96 (30.7) 205 (65.5) 28 (9.0) 57 (18.2) 4 (1.3) <0.0001 <0.0001 <0.0001 0.002 <0.0001 0.002 0.0007 0.21 265 (28.0) 51 (16.0) 88 (27.8) 126 (40.3) <0.0001 7.44 ± 3.95 0.28 ± 0.30 0.13 ± 0.12 6.82 ± 3.38 0.24 ± 0.27 0.08 ± 0.05 7.23 ± 3.63 0.29 ± 0.33 0.11 ± 0.07 8.01 ± 4.46 0.28 ± 0.31 0.16 ± 0.16 0.15 0.14 0.13 Dopamine Norepinephrine Epinephrine After 6 hours Mean arterial pressure ≥ 65 mmHg – no. (%) Central venous pressure – no. (%) < 8 mmHg 8-12 mmHg > 12 mmHg Central venous oxygen saturation ≥ 70% - no. (%) On day 1 Mean arterial pressure ≥ 65 mmHg – no. (%) Lactate < 2 mmol/L – no (%) Vasoactive drugs – no. (%) Dopamine Norepinephrine Epinephrine Dobutamine Vasopressin Two or more vasoactive drugs – no (%) Doses of vasoactive drugs - µg/kg/min Dopamine Norepinephrine Epinephrine P across tertiles 0.001 0.05 P for trend across tertiles were evaluated with the Cochran-Armitage test or one-way analysis of variance with tertiles of presepsin as a continuous variable. 6 Supplementary Table 3: Presepsin and clinical outcomes Outcome Presepsin at day 1 All Tertile 1 (<597 ng/L) Tertile 2 (597-1397 ng/L) Tertile 3 (> 1397 ng/L) Adjusted p Acute kidney injury (no. (%)) 200 (21.3) 46 (14.6) 54 (17.2) 100 (32.3) <0.0001 Renal replacement therapy (no. (%)) 213 (22.2) 17 (5.3) 54 (16.9) 142 (44.7) <0.0001 Length of stay in ICU – days (mean±SD) 15.4 ± 16.5 12.2 ± 14.1 16.3 ± 16.1 17.8 ± 18.5 0.0001 Length of stay in hospital – days (mean±SD) 29.1 ± 27.8 26.2 ± 24.1 30.6 ± 29.3 30.5 ± 29.7 0.04 Duration of mechanical ventilation – days (median [Q1-Q3]) 7 [2-15] 4 [1-10] 7 [3-17] 10 [3-18] <0.0001 Time to suspension of vasopressor or inotropic agents– days (median [Q1-Q3]) 3 [2-7] 2 [1-5] 3 [2-8] 5 [2-9] 0.006 Clinical resolution of the primary infection – (no. (%)) 488 (61.4) 190 (72.8) 169 (63.30) 129 (48.3) 0.001 Duration of the primary infection – days (median [Q1-Q3]) 8 [4-13] 6.5 [4-10] 9 [4-15] 9 [5-15] 0.0006 Incidence of secondary acquired infection during the study (no. (%)) 246 (25.7) 61 (19.1) 89 (27.8) 96 (30.2) 0.10 The relations between tertiary outcomes and presepsin concentration were analyzed with a multivariate logistic regression model and a linear regression model for categorical and continuous variables, respectively. The covariates included in the model were age, immunodeficiency, shock, mechanical ventilation and SOFA score at baseline. 7 Supplementary Table 4: Prognostic discrimination and reclassification of presepsin at day 1 ICU mortality Patients Model AUC [95%CI] p IDI [95%CI] Continuous NRI p Clinical model All Shock No shock Events correctly reclassified Non-events correctly reclassified 20% 34% 0.76 [0.74-0.80] 0.80 [0.77-0.83] Clinical model 0.74 [0.69-0.78] 0.80 [0.76-0.84] Clinical model 0.75 [0.69-0.80] IDI [95%CI] Continuous NRI p value Events correctly reclassified Non-events correctly reclassified 0.04 [0.03-0.06] p<0.0001 0.55 [0.41-0.69] p<0.0001 0.09 [0.06-0.11] p<0.0001 0.61 [0.43-0.79] p<0.0001 0.03 [0.01-0.05] p=0.01 0.39 [0.16-0.62] p=0.0009 0.001 0.04 [0.03-0.05] p<0.0001 0.51 [0.38-0.63] p<0.0001 18% 33% 0.002 0.06 [0.04-0.08] p<0.0001 0.58 [0.42-0.75] p<0.0001 21% 38% 0.39 0.003 [-0.0040.01] p=0.36 0.21 [0.00-0.41] p=0.05 5% 15% 0.81 [0.78-0.84] 23% 38% 0.81 [0.78-0.85] 0.81 [0.77-0.85] 0.17 0.77 [0.72-0.82] p 0.78 [0.74-0.82] <0.0001 Clinical model + presepsin AUC [95%CI] 0.79 [0.76-0.81] 0.002 Clinical model + presepsin Clinical model + presepsin 90-day mortality 15% 24% 0.81 [0.77-0.86] The variables included in the clinical model are listed in the legend of Table 3. AUC: area under the curve; IDI: integrated discrimination improvement; continuous NRI: category-free net reclassification improvement. Discrimination was evaluated by C-index comparing the clinical model with the same model plus day-1 concentration of presepsin with the DeLong test. 8 Supplementary Table 5: Interaction between presepsin at day 1 and albumin on outcome ICU mortality (No. events/No. patients (%)) 90 day mortality (No. events/No. patients (%)) Group All Albumin Crystalloids P All Albumin Crystalloids P Presepsin < 946 ng/L 68/479 (14.2) 39/242 (16.1) 29/237 (12.2) 0.22 122/473 (25.8) 67/238 (28.2) 55/235 (23.4) 0.24 Presepsin ≥ 946 ng/L 185/479 (38.6) 89/232 (38.4) 96/247 (38.9) 0.91 247/472 (52.3) 118/229 (51.5) 129/243 (53.1) 0.73 Breslow-Day for interaction between presepsin on day 1 and randomized treatment: p= 0.29 for ICU mortality, p=0.27 for 90-day mortality. 9 Supplementary Table 6: Prognostic value of changes in presepsin concentration over time Outcome Patients Changes from day 1 to day 2 No. events / no. OR [95%CI] Changes from day 1 to day 7 p patients (%) ICU mortality 90-day mortality No. events / no. OR [95%CI] p 146/716 (20.4) 2.74 [2.06-3.65] <0.0001 64/327 /19.6) 3.46 [2.17-5.50] <0.0001 82/389 (21.1) 2.39 [1.64-3.49] <0.0001 patients (%) All 216/870 (24.8) 2.43 [1.62-3.65] <0.0001 Severe sepsis 84/384 (21.9) 3.28 [1.74-6.19] 0.0002 Shock 132/486 (27.2) 2.07 [1.25-3.44] 0.005 All 325/858 (37.9) 1.59 [1.11-2.28] 0.01 242/704 (24.4) 1.90 [1.49-2.42] <0.0001 Severe sepsis 123/375 (43.7) 1.93 [1.09-3.43] 0.02 98/318 (30.8) 1.71 [1.20-2.44] 0.003 Shock 202/483 (41.8) 1.43 [0.91-2.26] 0.12 144/386 (37.3) 1.80 [1.31-2.47] 0.0003 The results of the logistic models for the association between changes in presepsin concentration over time (after log-transformation) and mortality are shown as odd ratio and 95% confidence interval. The covariates, as listed in the legend of Table 3, were introduced in the multivariable logistic models if they had a p<0.20 at univariate analyses and selected in the final model after backward elimination. 10 Supplementary Figure 1: Baseline presepsin and disease severity Baseline presepsin concentrations according to the SOFA score (A) or the numbers of organ dysfunction or failure at randomization (B). Number of patients with organ-specific failure on day 1 (SOFA scores 3-4): respiration (335, 35.9% of those with organ-specific evaluation of SOFA score), coagulation (124, 13.1%), liver (30, 3.2%), cardiovascular (578, 61.0%), kidney (204, 21.6%). P across categories by Kruskal-Wallis test. 11 Supplementary Figure 2: Time course of plasma presepsin levels according to microorganism culture results and appropriateness of antibiotic therapy Median [Q1-Q3] concentrations of presepsin at different times according to microorganism culture results in blood (a) or at the primary site of infection (b) and the appropriateness of empirical antibiotic therapy, as defined in the Methods. The number of patients in each category is shown in the columns. Two-way analyses for repeated measures adjusted for SOFA score at baseline. Time course by blood culture: time (p=0.05), appropriateness of antibiotic therapy (p=0.93), interaction time x appropriateness of antibiotic therapy (p=0.0009). Time course by site culture: time (p=0.57), appropriateness of antibiotic therapy (p=0.53), interaction time x appropriateness of antibiotic therapy (p=0.002). 12 Supplementary Figure 3: Time course of presepsin according to ICU survival Concentrations of presepsin are shown as median [Q1-Q3]. The number of patients is shown above the columns. 13 Supplementary Figure 4: Presepsin concentration on day 1 and mortality Kaplan–Meier survival curves for ICU or 90-day mortality by tertiles of presepsin concentration measured at day 1. The number of patients at risk is shown below the curves. P for log-rank test. 14