Molecular biology Dr. Mohammed Albayati
advertisement

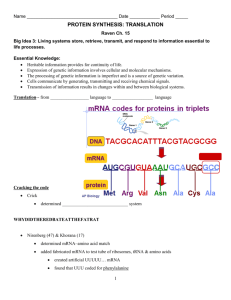
Molecular biology Dr. Mohammed Albayati 2015 Protein Synthesis and Targeting Codon–anticodon interaction In the cleft of the ribosome, an antiparallel formation of three base pairs occurs between the codon on the mRNA and the anticodon on the tRNA. If the 5’ anticodon base is modified, the tRNA can usually interact with more than one codon. Wobble The wobble hypothesis describes the nonstandard base pairs that can form between modified 5’anticodon bases and 3’codon bases. As long as the distances between the ribose units were close to normal. 1. No purine–purine or pyrimidine–pyrimidine base pairs are allowed as the ribose distances would be incorrect. 2. No single tRNA could recognize more than three codons, hence, at least 32 tRNAs would be needed to decode the 61 codons, excluding stop codons. 3. tRNAs can recognize either one, two or three codons, depending on their wobble base (the 5’anticodon base). a) If it is C it will recognize only the codon ending in G as usual. b) If it is G, it will recognize the two codons ending in U or C. c) If U, which is subsequently modified, it will pair with either A or G. d) The wobble nucleoside is never A, as this is converted to inosine which then pairs with A, C or U. Shine–Dalgarno sequence The ribosome binding site is a sequence just upstream of the initiation codon in prokaryotic mRNA which base-pairs with a complementary sequence near the 3’end of the 16S rRNA to position the ribosome for initiation of protein synthesis. Polysomes Polyribosomes (polysomes) form on an mRNA when successive ribosomes attach, begin translating and move along the mRNA. A polysome is a complex of multiple ribosomes in various stages of translation on one mRNA molecule. Page 1 of 8 Molecular biology Dr. Mohammed Albayati 2015 Initiator RNA A special tRNA (initiator tRNA), recognizing the AUG start codon, is used to initiate protein synthesis in both prokaryotes and eukaryotes. In prokaryotes, the initiator tRNA is first charged with methionine by methionyl-tRNA synthetase. The methionine residue is then converted to N-formylmethionine. In eukaryotes, the methionine on the initiator tRNA is not modified. This Initiator RNA differs from the tRNA that inserts internal Met residues. MECHANISM OF PROTEIN SYNTHESIS There are three stages of protein synthesis: initiation – the assembly of a ribosome on an mRNA; elongation – repeated cycles of amino acid delivery, peptide bond formation and movement along the mRNA (translocation); termination – the release of the polypeptide chain. Schematic representation of eukaryotic ribosome and the sites involved in protein synthesis. P (peptidyl) site initially binds initiator tRNA, later binds growing peptide chain. A (aminoacyl) site binds incoming tRNA molecule with activated amino acid (ie, bound amino acid with high-energy bond). E site receives uncharged tRNA once amino acid has been added to polypeptide. Page 2 of 8 Molecular biology Dr. Mohammed Albayati 2015 initiation In prokaryotes, initiation requires the large and small ribosome subunits, the mRNA, the initiator tRNA, three initiation factors (IFs) and GTP. IF1 and IF3 bind to the 30S subunit and prevent the large subunit binding. IF2 + GTP can then bind and will help the initiator tRNA to bind later. This small subunit complex can now attach to an mRNA via its ribosome-binding site. The initiator tRNA can then base-pair with the AUG initiation codon which releases IF3, thus creating the 30S initiation complex. The large subunit then binds, displacing IF1 and IF2 + GDP, giving the 70S initiation complex which is the fully assembled ribosome at the correct position on the mRNA. Elongation involves the three factors (EFs), EF-Tu, EF-Ts and EF-G, GTP, charged tRNAs and the 70S initiation complex (or its equivalent). o It takes place in three steps. o A charged tRNA is delivered as a complex with EF-Tu and GTP. The GTP is hydrolyzed and EF-Tu_GDP is released o Peptidyl transferase makes a peptide bond by joining the two adjacent amino acids without the input of more energy. o Translocase (EF-G), with energy from GTP, moves the ribosome one codon along the mRNA, ejecting the uncharged tRNA and transferring the growing peptide chain to the P-site. Termination Release factors (RF1 or RF2) recognize the stop codons and, helped by RF3, make peptidyl transferase join the polypeptide chain to a water molecule, thus releasing it. Ribosome release factor helps to dissociate the ribosome subunits from the mRNA. In eukaryotes a single release factor, eRF, recognizes all three stop codons and requires GTP for activity. Page 3 of 8 Molecular biology Dr. Mohammed Albayati 2015 Many antibiotics inhibit the protein synthesis at some specific steps: Streptomycin: It is a highly basic trisaccharide. It interferes with the binding of f-met t-RNA to ribosomes and thereby inhibits the initiation process. It also leads to misreading of m-RNA. Puromycin: This inhibits protein synthesis by releasing nascent polypeptide chains before their synthesis is complete. It binds to the A site on ribosome and inhibits the entry of aminoacyl-t RNA. It acts both in bacterial and mammalian cells. Tetracycline: It binds to the 30S subunit and inhibits binding of aminoacyl t-RNA, thus inhibits the initiation process. Chloramphenicol: It inhibits the peptidyl transferase activity of 50S subunit. Thus it inhibits the process of elongation. Cycloheximide: This inhibits peptidyl transferase activity of 60S ribosomal subunit in eukaryotes. It also inhibits elongation. Erythromycin: It binds to the 50S subunit and inhibits translocation. Diphtheria toxin: Corynebacterium diphtheriae produces a lethal protein toxin. It binds with EF-2 in eukaryotes and blocks its capacity to carry out translocation. TRANSLATIONAL CONTROL AND POST- TRANSLATIONAL EVENTS In prokaryotes, the level of translation of different cistrons can be affected by: (i) the binding of short antisense molecules, (ii) the relative stability to nucleases of parts of the polycistronic mRNA, and (iii) the binding of proteins that prevent ribosome access. Page 4 of 8 Molecular biology Dr. Mohammed Albayati 2015 In eukaryotes, protein binding can also mask the mRNA and prevent translation, and repeats of the sequence 5’-AUUUA-3’ can make the mRNA unstable and less frequently translated. A specific and interesting example is how body regulates the amount of iron in cells, iron being essential for the activity of some proteins, but harmful in excess. Iron is transported into cells by the transferrin receptor protein and is stored within cells bound to the storage protein ferritin. The mRNA for each of these proteins contains noncoding sequences that can form stem-loop structures, called the iron response element (IRE) to which an iron sensing protein (ISP) can bind. However, the position of the IRE and the action of the bound ISP is very different. In the transferrin receptor mRNA, the IRE is in the 3’ noncoding region and when the ISP binds, which it does when iron is low, it stabilizes the mRNA and allows more translation. But when iron levels are high the ISP dissociates from the IRE and unmasks destabilizing sequences that are then attacked by nucleases, thus reducing translation due to mRNA degradation. When iron levels are high, not only is the transferrin mRNA destroyed, but the translation of ferritin storage protein mRNA is increased. This occurs because at low iron levels the IRE, which is located in the 5’ noncoding region, binds ISP which in turn reduces the ribosome's ability to translate the ferritin mRNA. When iron levels rise, the ISP again dissociates from the IRE, but in this case causes an increase in translation of ferritin because the ribosome's progress is not repressed. This translational control system rapidly and responsively regulates intracellular iron levels. There are other examples of translational control that work via protein binding in the vicinity of destabilizing sequences and inhibiting them. Other examples of translational control include micro RNAs in eukaryotes that bind to mRNAs to which they are complementary, and either cause degradation or translational repression of the mRNA Polyproteins A single translation product that is cleaved to generate two or more separate proteins is called a polyprotein. Many viruses produce polyproteins. Page 5 of 8 Molecular biology Dr. Mohammed Albayati 2015 Protein targeting Certain short peptide sequences in proteins determine the cellular location of the protein, such as nucleus or mitochondrion. The signal sequence of secreted proteins causes the translating ribosome to bind factors that make the ribosome dock with a membrane and transfer the protein through the membrane as it is synthesized. Usually the signal sequence is then cleaved off by signal peptidase. Protein modification the most common alterations to nascent polypeptides are those of cleavage and chemical modification. Cleavage examples are a) Signal sequences are also usually cleaved off secreted proteins b) Release mature fragments from polyproteins eg. Ubiquitin is made as a polyprotein containing multiple copies linked end-to-end, and this must be cleaved to generate the individual ubiquitin molecules, c) Remove internal peptides as in the case of insulin d) Trimming both N- and C-termini. There are many chemical modifications that can take place on all but six of the amino acid side chains, ( Ala, Gly, Ile, Leu, Met and Val. )The modifications include acetylation, hydroxylation, phosphorylation, methylation, glycosylation and even the addition of nucleotides. Hydroxylation of Pro is common in collagen, and some of the histone proteins are often acetylated. Often phosphorylation controls the activity of the protein. Protein degradation Different proteins have very different half-lives. Regulatory proteins tend to turn over rapidly and cells must be able to dispose of faulty and damaged proteins, modified or inherently unstable proteins, these are marked for degradation by having multiple molecules of ubiquitin covalently attached. The ubiquitinylated protein is then degraded by a 26S protease complex. Page 6 of 8 Molecular biology Dr. Mohammed Albayati 2015 The majority of the degraded proteins are reduced to amino acids that can be used to make new proteins, but random peptide fragments of nine amino acids in length are attached to peptide receptors (called major histocompatibility complex class I molecules) and displayed on the surface of the cell. Cells display around 10 000 of these peptide fragments on their surface and this gives them a unique identity since different individuals display a different set of these peptide fragments on the surface of their cells. These differences explain, not only why transplanted organs are often rejected, but also why cells infected by viruses (which will display foreign peptide fragments on their surfaces) will be destroyed by the immune system. In eukaryotes, it has been discovered that the N-terminal residue plays a critical role in inherent stability. 8 N-terminal aa correlate with stability: Ala Cys Gly Met Pro Ser Thr Val 8 N-terminal aa correlate with short t1/2: Arg His Ile Leu Lys Phe Trp Tyr 4 N-terminal aa destabilizing following chemical modification: Asn Asp Gln Glu Methods of Regulation of Gene Expression in Eukaryotes 1. RNA processing: 2. Gene amplification: the expression of a gene is increased several-fold. This is commonly observed during the developmental stages of eukaryotic organisms. In pateints receiving Methotrexate therapy malignant cells can develop drug resistance by increasing the number of genes for the enzyme dihydrofolate reductase. 3. Gene rearrangement eg. synthesis of light chains of immunoglobulins (Igs). 4. Gene regulation by histones and nonhistone proteins: Page 7 of 8 Molecular biology Dr. Mohammed Albayati 2015 post-translational modifications of the different histones. Such modified histones can regulate gene expression. 5. Class switching: In this process, one gene is switched off and a closely related gene takes up the function. Examples: Class switching is best illustrated by Hb Zeta-eta → α γ → then α β and α δ 6. Binding of regulatory proteins to DNA: • Helix-turn helix • Zinc finger motif, and • Leucine zipper motif. 7. Role of enhancers and silencers Figures and diagrams are present in the ppt of these lectures This will conclude our lectures for this year You are expected to read more from your text book And your lecture hand notes to pass the final exam. Page 8 of 8