Ionic Compound Puzzle: Science 8 Worksheet
advertisement

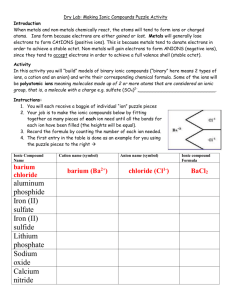
Science 8 – Helgeson Ionic Puzzle Piece Activity Name _________________________________________ Period ________ Introduction: When metals and non-metals chemically react, the atoms will tend to form ions or charged atoms. Ions form because electrons are either gained or lost. Metals will generally lose electrons and form positive ions. This is because metals tend to donate electrons in order to achieve a full valence shell (stable state). Non-metals will gain electrons to form negative ions since they tend to accept electrons in order to achieve a full valence shell (stable octet). Activity: In this activity you will create models of ionic compounds and observe the chemical formula of the ionic compound you have created. You will need at least one positive ion and a negative ion for each compound. To create an ionic compound: 1. Cut out the positive and negative ion pieces. 2. Place the cards together, adding additional cards of the same ion until the heights of the positive ions and negative ions are equal. 3. Count the number of each ion needed for the heights to be equal and record. 4. Record the name of each positive ion and negative ion combined. 5. Write both the formula and name for each ionic compound. 6. Make as many compounds as possible! ClAl3+ Cl- = AlCl3 (Aluminum Chloride) Cl- Positive Ion Name Number of Positive Ions Negative Ion Name Number of Negative Ions Chemical Formula Aluminum +3 Sodium +1 Magnesium +2 1 Chloride -1 Fluoride -1 Chloride -1 3 AlCl3 1 NaF 2 MgCl2 1 1 Ionic Compound Name Aluminum Chloride Sodium Fluoride Magnesium Chloride Positive Ion Name Number of Positive Ions Negative Ion Name Number of Negative Ions Chemical Formula Ionic Compound Name Follow-up Questions: 1. What is the charge for all elements in Group 1? What is the charge for all elements in Group 2? What is the charge for all elements in Group 17? 2. Do you notice a pattern in the charge for elements in each group? Explain. (Hint: relate to number of valence (outer) electrons and the octet rule (stable structure)) 3. Can an ionic compound ever consist of a negative-negative or positive-positive bond? Explain. 4. What is the resulting charge of an atom that loses one or more electrons? What is the resulting charge of an atom that gains one or more electrons? 5. What is the overall charge of ionic compounds? Why?