CHEM111: Final Lab Report - Personal.psu.edu
advertisement

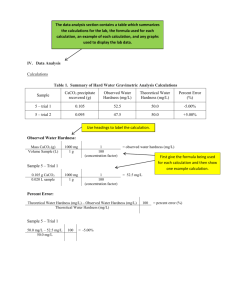
CHEM111 Section 103 A QUANITATIVE ANALYSIS OF DISSOLVED SOLID CONTENT IN VARIOUS WATER SOURCES Grant Kovich Partners: William Boring, Josh Boyer, and Victoria Bosman Date Performed: October 19, 2010 TA: Eric Miller Introduction Water is an essential compound that is necessary for many types of life to exist. It is responsible for many cellular processes, hydration, and overall well being. Water is truly everywhere, and it makes up a larger portion of the Earth’s surface than all of the landmasses. However, water quality is not universal. Many bottled water companies compete to have the purest water, and other companies have created home water purifiers so that water from a tap is just as pure from a mountain spring. Although these measures are in place, there are many environmental elements that can affect water quality. Hardness occurs in natural waters and can be based on the concentration of divalent cations present in the water, with Mg2+ and Ca2+ being the most common. The more ions dissolved in the water, the harder the water will be.1 Hardness also affects the suitability of water for use in the textile and paper industries and certain other industries that depend on steam boilers and water heaters.1 Dissolved calcium can have a large impact on the biological life processes of fish. It is essential for bone formation, blood clotting, metabolic reactions, and can also help to reduce the loss of other salts via osmosis such as sodium and potassium.2 There are no known human health effects as a result of drinking hard water, but it can lead to many industrial problems. When hard water is used in industrial boilers and evaporators, rocklike deposits form on the surface of pipes, boiler walls, and evaporator surfaces.1 As a result of the deposition, water has a smaller area to travel through, due to the narrowing of the pipes and tubes. Scale formation is extremely expensive to remove, and the harder the water is, the larger 1 Keiser, Joseph T. PSU Version of Chemtrek. Prentice-Hall, Englewood Cliffs, NJ, 2011, pages 6—10-8, 10-15—10-20. 2 10-2—10-3, 10- Wurts, William A. "Understanding Water Hardness." World Aquaculture 24.1: 18. Web. 28 Oct. 2010. <http://www.ca.uky.edu/wkrec/Hardness.htm>. 2 the problem will be in the long run. Large water circulating components may experience energy insufficiencies as a result of deposition due to the heating of hard water.1 There are ways, however, to eliminate hard water, not only for industrial use, but for household use as well. Water hardness can be measured in a variety of ways, but technological advancements have replaced earlier, more primitive methods. Soap titrations were used, but ended up being very time consuming and returned inaccurate results. Newer methods have been developed to measure water hardness including total dissolved solids analysis, EDTA titration, cation exchange resin, and atomic absorption spectrophotometry (AA). Total dissolved solids analysis (TDS) is one of the simplest methods of determining what solutes are present in a sample of water, and in what quantity they are present as well. The water sample is evaporated, and the residue of remaining solids is examined in order to determine the solids present in the sample.1 EDTA titration is a type of complexation titration in which a chelating agent, ethylenediaminetetracetic acid (EDTA), is used. If there is Mg2+ present in the sample, the ions will react with an indicator, eriochrome black T (EBT), forming a wine red chelate. When EDTA is added, it will first react with the Ca2+, forming a colorless chelate. After a sufficient amount of EDTA is added to chelate the entirety of Ca2+, it will then begin to react with the previous wine red chelate of magnesium, producing a colorless MgEDTA chelate. As the magnesium ions are removed from the indicator, it will begin to return to a sky blue color. Thus, the endpoint of the titration is reached when there is a distinct change from wine red to sky blue. When the titration is complete, the concentration of dissolved ions in the water sample can be calculated.1 3 Atomic absorption spectrophotometry (AA) can also be used to determine the amount of dissolved metals in a water sample. The atomic absorption spectrophotometer is a machine that analyzes the amount of metal atoms (divalent cations in this case) in a sample of water (Fig. 1).3 The sample is read when an aspirator is placed in the sample, which allows the sample to be aspirated into the machine. At this point, monochromatic light with energy that corresponds to the divalent cation ΔΕ is placed into the sample. The divalent cations in the sample having the same ΔE will absorb the light.1 The amount of absorption is proportional to the concentration of divalent cations in the sample. By using the Beer-Lambert law, the unknown metal concentration of the sample can be calculated. Beer’s law states that the amount of light absorbed is an exponential function of the concentration of absorbing substance. Therefore, if the absorbance of a compound is plotted on a graph, the concentration of that substance in the solution can be calculated because absorbance and concentration are proportional.4 Figure 1. Diagram of standard AA machine. 3 “AAS Basics.” NMSU – Department of Chemistry and Biochemistry. Web. 29 Oct. 2010. <http://www.chemistry.nmsu.edu/Instrumentation/AAS1.html 3 4 "Beer's Law Tutorial." UCLA Chemistry and Biochemistry. Web. 28 Oct. 2010. <http://www.chem.ucla.edu/~gchemlab/colorimetric_web.htm>. 4 Water samples can be softened, either by using a commercial softening agent such as Arm and Hammer or by using cation exchange resins. Water softening is the removal of Mg2+ and Ca2+ ions that cause hard water.5 When using a commercial softening agent, it usually contains sodium carbonate, which adds Na+ ions to the water.6 EDTA titration or AA can be used to determine the hardness of the sample after the softening agent is added. Ion exchange is another way of softening a water sample, which is done by adding cation exchange resin to the water sample. Upon aggravation of the sample, ion exchange will occur between the sample and the cation exchange resin in which monovalent cations such as Na+ or H+ are exchanged with the divalent cations present in the water sample (Fig 2). After the exchange is complete, the concentration of dissolved solids in the sample can be determined by EDTA titration or AA in order to determine the effectiveness of water softening.1 - + SO3 Ca - + 2+ - SO3 Na + Ca - 2+ - SO3 Na + SO3 + 2Na - 2+ SO3 Ca + SO3 Na + Figure 2. Diagram of ion exchange. Monovalent cations (Na ) are exchanged with divalent 2+ 1 cations (Ca ). In this experiment, there were four different water samples taken: an apartment at 642 Oakwood Ave., State College; Beta Theta Pi Fraternity, 220 N. Burrowes St., State College; 5 Brown, Theodore L.; LeMay Jr., H. Eugene; Bursten, Bruce E.; Murphy, Catherine J. Chemistry: The Central Science. Pearson Custom Publishing, 11th Edition: Upper Saddle River, NJ, 2009, page 788. 6 Kovich, Grant J. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, MI, 2009, page 34. 5 Delta Theta Sigma Fraternity, 101 N. Patterson St., State College; and the fountain at the Penn State Arboretum. I believe that the samples taken from the three residential areas to be of similar hardness, due to the fact that the water should be coming from the same treatment plant in State College. The hardness of tap water may differ by location, which will determine the effectiveness of water purification. The residential samples may also differ if each residence has additional methods of water purification such as a water softener. Not all municipalities require water softening, but the sample taken from each tap will show if the borough of State College uses water softening, and also if each residence has additional methods of water softening. The hardness of the sample taken from the Arboretum fountain will depend on when it is retrieved. We expect the water flowing through the fountain to be from a tap, just like the residential areas, but rainwater can also accumulate in the fountain. Although rainwater contains dissolved gases and man-made pollutants from the atmosphere, it still should be relatively soft.7 However, the water in the fountain may not be treated in the same manner as residential water is treated, due to the fact that it is not to be consumed. Ultimately, the hardness of the fountain water will depend on the amount of fountain water compared to the amount of rainwater present. If there is a larger amount of rainwater, the sample should be relatively soft, but if there is little rainwater accumulation in the fountain when the sample is collected, I predict that the water will be just as hard as residential water. Procedure Experimentation was done in a controlled environment in which safety regulations were followed at all times. A teacher’s assistant, who helped with problems or concerns, conducted 7 Leeden, Frits van der; Troise, Fred L.; Todd, David Keith. The Water Encyclopedia. Lewis Publishers, 2nd Edition: Chelsea, MI, 1990, pages 418,452. 6 supervision. In order to determine a baseline value for comparison, the determination of each sample’s hardness was done by atomic absorption spectroscopy. This method is considered to be the most accurate, due to the fact that there is little room for experimental error except for calibration discrepancies. After running the samples through the AA, an absorbance value was given, which was used to calculate the amount of calcium and magnesium ions in the sample, thus giving a hardness value for the water sample. The remaining methods were compared to the AA results to see whether or not they are effective indicators of water hardness.1 A small drop of each water sample was placed on a small piece of aluminum foil (2 cm x 2 cm) along with a drop of dH2O and 1.0 x 10-3 M Ca2+. The distilled water and calcium ion solution were used to compare the TDS reading with the water sample. The foil was placed on a hotplate where it was heated until total evaporation was complete. The amount of nonvolatile salts in each water sample could then be determined by TDS.1 The next part of the procedure was used as an indicator for the titrations done throughout the rest of the experiment. The titrations were carried out in a serial manner, good wash, rinse, and transfer techniques were used during the process. Samples of Ca2+ and Mg2+ with known concentrations were tested to ensure that the titration was being carried out in the correct way before analysis of each water sample was performed. These titrations followed the same steps as the initial EDTA titration to determine the hardness of each water sample.1 The hardness of each water sample was then determined by EDTA titration, in which one drop of the water sample was added to each well of a 1x12 well strip. One drop of EBT indicator and one drop of NH3/NH4Cl/MgEDTA buffer was also added before serial titration using EDTA solution was executed. Once the EDTA titration was familiar and the hardness of 7 each water sample could be determined, a comparison was made between AA results and EDTA results.1 The final focus of the experiment was to determine the effectiveness of different water softening techniques, and use titration techniques previously learned to determine the hardness of each water sample after it had been softened. First, each sample was softened using a commercial water-softening agent, such as Arm and Hammer. The samples were then titrated using the EDTA method to determine the hardness and whether or not the softening technique worked. Second, an ion exchange resin was used to exchange divalent cations within the water sample with monotonic ions in the exchange resin, thus changing the chemical composition of the water and ultimately making it softer, which was done by adding a small amount of cation exchange resin to a vial containing the water sample. The vial was then shaken, and the supernatant liquid was analyzed using EDTA titration.1 8 Results Sample Tested AA Values Calcium Molarity Values (mol/L) Magnesium EDTA Titration 3.2 x 10-3 NaCO3 Ion Exchange William 0.3986 0.2938 2.2 x 10-3 4.0 x 10-4 Boring8 Victoria 0.4385 0.3465 1.0 x 10-3 1.2 x 10-3 4.0 x 10-4 Bosman9 Josh Boyer10 0.4240 0.2982 2.4 x 10-3 2.4 x 10-3 <2.4 x 10-3 Grant 0.3871 0.3001 3.6 x 10-3 3.6 x 10-3 2.0 x 10-4 6 Kovich Figure 3. Table displaying determined absorbance values and molarity of dissolved ions in each water sample. Sample Tested EDTA Titration* mol/LA -3 ppmB NaCO3* gr/galC mol/LA Ion Exchange* ppmB gr/galC -3 mol/LA -4 ppmB 18.71 2.2x10 220 12.87 4.0x10 40 William 3.2x10 320 8 Boring 5.85 1.2x10-3 120 7.02 4.0x10-4 40 Victoria 1.0x10-3 100 9 Bosman 2.4x10-3 240 14.04 2.4x10-3 240 14.04 < < 240 Josh -3 10 2.4x10 Boyer 3.6x10-3 360 21.05 3.6x10-3 360 21.05 2.0x10-4 20 Grant Kovich6 Figure 4. Hardness values of each water sample presented in various units of hardness: (mol/L), parts per million (ppm), and grains per gallon (gr/gal). gr/galC 2.34 2.34 < 14.04 1.17 molarity William Boring – Delta Theta Sigma Fraternity 101 N. Patterson St., State College, PA Victoria Bosman – Apartment 642 Oakwood Ave., State College, PA Josh Boyer – Fountain at Penn State Arboretum Grant Kovich – Beta Theta Pi Fraternity 220 N. Burrowes St., State College, PA *Superscripts represent sample calculations listed on following page. 8 Boring, William M. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, 2009, page 33. 9 Bosman, Victoria R. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, 2009, page 34. 10 Boyer, Joshua G. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, 2009, page 31. 9 MI, MI, MI, Sample Calculations A. M of EDTA (V of EDTA) = M of H2O (V of H2O) 2.0 x 10-4 (6 drops) = 1.0 (M of H2O) M of H2O = 1.2 x 10-3 M (dilution factor of 3) = 3.6 x 10-3 M B. 3.6 x 10-3 mol CaCO3 x 100.0 g CaCO3 x 1000 mg CaCO3 = 360 mg CaCO3 = 360 ppm 1 mol CaCO3 1 g CaCO3 1 L solution C. 360 ppm CaCO3 x 1 gr/gal = 21.06 gr/gal 17.1 ppm Calcium Ion Concentration (ppm) Calcium Ion Absorbance and Concentration 50 45 40 35 30 25 20 15 10 5 0 -5 0 y = 80.983x - 1.3939 R² = 0.9932 0.05 0.1 0.15 0.2 0.25 0.3 0.35 0.4 Absorbance at 422.7 nm 0.45 0.5 0.55 0.6 0.65 Magnesium Ion Conentration (ppm) Figure 5. Graph of absorbance values from AA to determine Ca2+ concentration. Magnesium Ion Absorbance and Concentration 30 25 y = 50.282x - 0.6649 R² = 0.9983 20 15 10 5 0 0 0.05 0.1 0.15 0.2 0.25 0.3 0.35 0.4 Absorbance at 202.5 nm 0.45 0.5 0.55 0.6 Figure 6. Graph of absorbance values from AA to determine Mg2+ concentration. 10 0.65 Discussion As previously stated, a prediction was made that for the most part, the water coming from residential sources should have similar hardness values, with the only factor making them different from one another being that there was additional water softening within the residence. There was also a prediction made that water from the fountain would not be as hard as the water from the residential areas, due to the fact that rainwater is considerably soft compared to Pennsylvania groundwater, and that the mixture of softer rainwater and hard groundwater would give way to a water sample that was not as hard as the tap water. Pennsylvania groundwater is considerably hard compared to other states, containing about 145 ppm in Centre County. Even though Centre County has hard water, Pennsylvania as a whole has harder water than most of the states along the east coast and in the Midwest.7 As shown in the prior data, it can be seen that the water sample from Beta Theta Pi Fraternity and William’s sample from Delta Theta Sigma Fraternity returned similar hardness values (Fig. 4). The location of these residences may give light to why the hardness values are similar. Beta Theta Pi is located at 220 N. Burrowes Street in State College, but it is technically considered a part of Penn State University property, located directly across from the Deike Building. Delta Theta Sigma Fraternity is located a mere five blocks away from Beta, but not on Penn State University property, rather it is considered to be located in downtown State College. Regardless of whether or not the residence is located on University property or not, we believe that most, if not all of the water throughout downtown State College and University Park comes from the same treatment plant, and runs through the same pipes, thus making it have the same hardness level. Delta Theta Sigma’s water had a smaller concentration of dissolved metal ions (~40 ppm difference) than Beta. This could be due 11 to additional water treatment techniques carried out in that residence, such as having a water softener installed in the residence. The samples from the two fraternities were fairly similar, but they were not close in dissolved metal ion concentration of the sample collected from an apartment at 642 Oakwood Ave., State College. One reason for this could be the geographic distance between the sampling locations. Beta Theta Pi Fraternity is located 3.2 miles away from the apartment, and is located “on-campus” while the apartment is located in a housing development. Perhaps the development is located closer to the water treatment plant, which allows for softer water to be delivered first to the development. If that is the case, it may be true that as water moves through pipes, it picks up crystallized salts on the interior of pipes that are dissolved in water during transport. This could explain a higher level of water hardness. Another reason why the apartment water sample could be softer than the Fraternity’s water is the fact that the apartment complex is located very close to Toftrees Golf Course, which has many natural bodies of water. The water from the hazards on the golf course may seep into the ground, which could potentially be softer because it is not passing through limestone or other types of shale, which add salts to water. The sample collected from the Arboretum fountain was very interesting to examine. It was previously thought that the water from the fountain would either be all from a tap, or a mixture of tap water and rainwater, depending on when the sample was taken. It was determined that the fountain water was actually softer than the Fraternity samples. Since both fraternity houses are relatively close to the fountain, it is safe to say that their water most likely comes from the same source. It was previously mentioned that rainwater is relatively soft, and when the sample of water was taken from the fountain, there must have been a significant amount of 12 rainwater present for the tap water to soften. The rainwater almost acted as a softening agent that lowered the hardness value of the fountain water (Fig. 4). When water softening was performed on the fountain water, interesting results were found. The hardness values did not change between the EDTA and commercial water-softening agent, and the value using ion exchange resin was undeterminable (< 2.4 x 10-3 mol/L). This may be because rainwater is very soft and does not contain many dissolved solids, so it acted as a natural water-softening agent. This can explain the similarity in hardness between EDTA and the commercial softening agent, but when the titration was performed for ion exchange, the value was undeterminable. This could mean that the solution was too diluted to contain a reportable hardness value (too few ions dissolved in solution) or that the exchange resin worked extremely well when presented with fountain water, which was found to contain both rainwater and tap water (Fig. 4). The rainwater may have lowered the hardness of the entire sample, making it easier for the ion exchange resin to work and soften the water sample. Although the samples from Beta Theta Pi Fraternity and Delta Theta Sigma returned similar AA values, the hardness values using EDTA titration were not similar (Fig. 3). This could have been because of various sources of error. The same principle holds true for the apartment and fountain sample; the AA values were very similar, but the hardness values reported were significantly different (Fig. 3). One factor that returned different values between EDTA and AA is the fact that EDTA accounts for total divalent cations dissolved in solution, not just calcium and magnesium. The AA machine only found the absorbance of calcium and magnesium cations, with which those values were used to determine hardness. Depending on how hard the water sample was, dilutions may have been done in order to analyze the sample better in the AA, and those dilutions may not have been carried over into calculations when 13 performing EDTA titration. Also, the experimental error when performing EDTA titration may have skewed results. By ignoring proper pipetting technique, and not paying attention to the amount of each compound was added to each well, the titration could have easily been thrown off one or two wells. Similar errors in water softening using a commercial water-softening agent may be to blame for misleading data (Fig. 4). The amount of softening agent may not have been measured correctly, or it may not have been added to the correct volume of solvent, which in this case was the water sample. Even if the mass of the softening agent was off by 0.001, it could have made a difference of how effective the softening agent was in eliminating dissolved divalent cations, due to the fact that there was such a large concentration, even in a single drop. When the ion exchange method was used to soften each water sample, there wasn’t necessarily an exact amount of resin to be used; rather we were supposed to simply cover the bottom of the vial with resin. Also, the amount of water added was not exact either, because we were supposed to add a few cm of water. These factors could have played a role in how much of the ions were exchanged. Also, the time allocated for mixture of the ion exchange resin and the water sample may have skewed results. If the mixing process was not carried out for a long enough time, the resin may have lost its effectiveness. On the other hand, if the mixing process was carried out for too long, too many ions may have been exchanged, giving a false hardness value. We feel that the AA method is an accurate method to determining water hardness, due to the fact that a check standard was performed. After calibration, a check standard was placed into the machine of a known concentration to determine the accuracy of results found. These results were not as close to the known value as expected, but the machine was still more accurate than 14 performing a titration where volumes were solely based on drops, which could have fluctuated based on improper pipetting technique. We also believe that the machine is very precise, because the water sample could have been placed in the machine multiple times, and the same reading would have been delivered, due to the extensive calibration needed. There are only two reasons, in our opinion, that the AA would deliver incorrect readings. If the sample was too hard, in that it contained a very large concentration of dissolved solids, the AA may not have been able to give an accurate reading. This was the case for many samples that were too hard and had to be diluted before being analyzed using the AA. The second reason why the AA may deliver incorrect results would be due to incorrect calibration. Just like any other instrument, it can function correctly even if the calibration is not set correctly, so the calibration of the instrument is extremely important to its accuracy. Conclusion In conclusion, the difference in water hardness values among residential areas can be related to geographic location of where the water sample is being taken from. We believe that the farther water has to travel, the more potential dissolved solids the water can have dissolved in it, due to deposition of solute crystals on the interior of pipes. The closer a water sample is to a water treatment plant, the softer it will be, whereas the farther a sample is taken from the treatment plant, the harder the sample will be. Our hypothesis of the three residential water samples being of similar can be partially accepted, due to the fact that the samples taken from both fraternity houses were of similar hardness, but the sample taken from the apartment was softer, perhaps because it was in a residential area closer to bodies of water. It is true, however, that rainwater did play a role in softening the water in the fountain, because if the fountain contained solely water from an underground source, it would have been just as hard as the water 15 taken from the taps at the fraternity houses. Rainwater also played a role in allowing for almost complete softening of the sample using ion exchange resin, which leads one to believe that rainwater can be used as a natural softening agent, or even a factor that aids in the softening process. Whatever the case, water hardness was interesting to examine due to the geographical differences of where the samples were collected, and also the chemistry of the water that made each sample unique from the other. Not all water is the same; it may be treated at the same treatment plant, but it is along the journey water takes from plant to source that gives it an identity, which in this case, is the amount of dissolved solids present in the sample. References 1. Keiser, Joseph T. PSU Version of Chemtrek. Prentice-Hall, Englewood Cliffs, NJ, 2011, pages 10-2—10-3, 10-6—10-8, 10-15—10-20. 2. Wurts, William A. "Understanding Water Hardness." World Aquaculture 24.1: 18. Web. 28 Oct. 2010. <http://www.ca.uky.edu/wkrec/Hardness.htm>. 3. "AAS Basics." NMSU - Department of Chemistry and Biochemistry. Web. 29 Oct. 2010. <http://www.chemistry.nmsu.edu/Instrumentation/AAS1.html>. 4. "Beer's Law Tutorial." UCLA Chemistry and Biochemistry. Web. 28 Oct. 2010. <http://www.chem.ucla.edu/~gchemlab/colorimetric_web.htm>. 5. Brown, Theodore L.; LeMay Jr., H. Eugene; Bursten, Bruce E.; Murphy, Catherine J. Chemistry: The Central Science. Pearson Custom Publishing, 11th Edition: Upper Saddle River, NJ, 2009, page 788. 6. Kovich, Grant J. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, MI, 2009, page 34. 16 7. Leeden, Frits van der; Troise, Fred L.; Todd, David Keith. The Water Encyclopedia. Lewis Publishers, 2nd Edition: Chelsea, MI, 1990, page 452. 8. Boring, William M. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, MI, 2009, page 33. 9. Bosman, Victoria R. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, MI, 2009, page 34. 10. Boyer, Joshua G. Penn State University Chemistry 111/113 Lab Notebook. Hayden McNeil, Plymouth, MI, 2009, page 31. 17