Chapter 4 Theoretical Prediction of A New Class of Xenon
advertisement

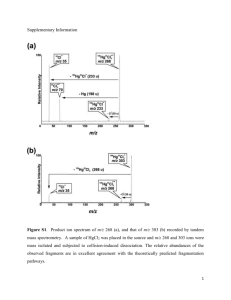
Chapter 4 Theoretical Prediction of A New Class of Xenon Containing Molecules and Anions NXeOnFm Abstract We have predicted a new series of xenon containing noble-gas molecules. These molecules consist of xenon, nitrogen, oxygen and fluorine atoms with the general formula of NXeOnFm and NXeOnFm, for example: NXeOF3 , NXeF3 , NXeF5 , NXeOF2, NXeO2F2 , NXeOF4 and NXeF4. The best estimates of the atomization energies of the most stable species NXeF5 and NXeOF4 were 104 and 140 kcal/mol, respectively. These are higher than those of XeNO2 and XeNO3 in our previous study by ~50 kcal/mol. These molecules were all predicted to have very short XeN bond lengths (~1.8 Å), suggesting XeN triple bonds. The lowest unimolecular dissociation barriers of the most stable species NXeOF4 were estimated to be 33 kcal/mol. Many of these molecules and anions were predicted to be kinetically stable at low temperature. 107 Introduction Traditionally, noble gases were found to be extremely stable and were difficult to form molecules with other elements. However, since the first Xe compound XePtF6 had been synthesized in 1862 by Bartlett et al,1 a large variety of noble gases containing compounds, mostly containing Xe, F, and O atoms. have been found in various laboratories,.2,3 Recently, Räsänen and coworkers4 have found many xenon containing molecules of the type HXeY (where Y is usually an electronegative group such as Cl, Br, I, OH, CN, NC, CCH, SH, NCO, etc.) using matrix isolation/photolysis techniques. Xenon is also known to play the role of ligands to transition-metal ions and to form actinide complexes with the CUO molecule.5 The first compound that contains the XeN bond, FXeN(SO2F)2, was synthesized in 1974 by LeBlond and DesMarteau.6 About twenty other compounds containing XeN bond had been synthesized in the experiment during the next 25 years.7 The study on the XeN compounds was notbly followed by Schrobilgen and coworkers in recent years. 8,9 In our previous theoretical studied,10 the XeNO2 and XeNO3 anions had been predicted to be stable with 50 and 101 kcal/mol atomization energies and very short XeN bond lengths (~1.8 Å). We have been wondering if there are any other types of stable molecules or anions also containing the XeN triple bond motif. From past experience, stable Xe compounds are chemically bonded to fluorine or oxygen atoms.11 Therefore, in this research, we tried to replace one or more oxygen atoms in XeNO2 and XeNO3 with fluorine atoms to generate a new series of neutral and mono-negative charged species such as NXeO2F and NXeOF4. The molecules and anions we tested can be expressed as NXeOnFm or NXeOnFm. For Xe atom, at most 8 valance electrons can be shared to surrounding electronegative atoms. For example, 108 in XeNO3, 3 valence electrons of Xe are shared with the N atom and 6 valence electrons were shared with three O atoms, and the negative charge is delocalized within the entire molecule. Thus the combinations we tested were n=1-2 and m=1-4 for NXeOnFm or NXeOnFm. The negative charge keeps the molecules with even electrons. Species with some of the combinations were found be unstable. The stable (with atomization of at least 30 kcal/mol) neutral and mono-negative charged species we founded were NXeOF3 , NXeF3 , NXeF5 , NXeO2F2 , NXeOF4 , NXeOF2 and NXeF4. Some isoelectronic neutral molecules without N atom, such as XeO3F2, XeO2F4, XeO2F2 and XeOF4 were also compared in this study. 109 Method The molecular geometry was calculated using the MP212 and CCSD(T)13 theory and the hybrid density functional theory B3LYP14 and MPW1PW9115 with the aug-cc-pVTZ basis sets for N, O and F atoms.16 For Xe atoms, the aug-cc-pVTZ-pp basis sets was used. The “pp” means that a pseudo-potential was used to replace the core electrons. Our previous study10 showed that the B3LYP method with aug-cc-pVTZ(-pp) basis sets predicted structures and energies in reasonable agreement with the higher level coupled cluster method. Single-point energy calculation was also performed at CCSD(T)/aug-cc-pVTZ(-pp) level using the B3LYP geometry. For brevity, the basis sets will just be described as aug-cc-pVTZ for the rest of this article, and they are abbreviated as apnz in all the Tables. All calculations were performed using the Gaussian 03 program.17 110 Result and Discussion (a) Geometry In our previous study, the bonding between xenon and nitrogen atoms of XeNO2 and XeNO3 could be assigned as a triple bond. Three valence electrons of xenon are shared with the nitrogen atom. Bases on the Lewis structure, there are five other valence electrons of xenon can be shared with other oxygen and fluorine atoms. Thus, the possible NXeOnFm molecules with even numbers of electrons are NXeF, NXeOF, NXeO2F, NXeF3, NXeOF3, NXeF5, NXeO, NXeF2, NXeOF2, NXeO2F2 and NXeF4. However, the NXeF, NXeOF, NXeO2F and NXeO were found to have very small (less than 30 kcal/mol) atomization energies at B3LYP/aug-cc-pVTZ level. Therefore, these molecules will not be further discussed in this study. Figure 1 shows the calculated structures of NXeF3, NXeOF3, NXeF5, NXeF2, NXeOF2, NXeO2F2 and NXeF4. The structure of XeNO3 was also showed in Figure 1 for comparison. The calculated vibrational frequencies are listed in the Table 1. The calculated frequencies of XeN stretching mode are predicted to be 866~965 cm-1 at B3LYP/aug-cc-pVTZ level. Those were slightly higher than the XeN stretching frequency of the XeNO3 of 848 cm-1 calculated at the same level of theory. Table 2 shows the calculated XeN bond lengths at various theoretical levels. The NXeF4 was predicted with the shortest XeN bond of 1.757 Å at B3LYP/aug-cc-pVTZ level. At CCSD(T)/aug-cc-pVTZ level, the XeN bond length of NXeF4 is 1.754 Å which is significantly shorter than the predicted length of 1.800 Å in XeNO3 of.ref. The XeN bond lengths of NXeF5 and NXeF4 in Table 2, also 111 show that the B3LYP/aug-cc-pVTZ method predicted XeN bond lengths in better agreement with the higher level CCSD(T)/aug-cc-pVTZ method than the MPW1PW91 and MP2 methods. The calculated XeN bond length at B3LYP/aug-cc-pVTZ level in Table 2 were all in the range of 1.757~1.792 Å, and, as discussed in our previous study, bonding could be assigned as triple bonds. The XeO and XeF bond length of these molecules were also shown in Figure 1. The XeF bond length predicted by B3LYP/aug-cc-pVTZ were 1.902~2.095 Å, which can be compared with the experimental XeF bond lengths of 1.974 Å .18 The two types of XeF bond lengths of NXeF5 were predicted to be 1.906Å and 1.868Å at the CCSD(T)/aug-cc-pVTZ level. For comparison, the bond length of XeF6 (Oh) was 1.942 Å at the same level, which was in good agreement with experimental value of 1.941 Å18. The B3LYP/aug-cc-pVTZ calculation predicted that the XeN bond is shorter than XeO bond by approximately 0.02~0.03 Å for anions. This is consistent with our previous study on XeNO2 and XeNO3. However, the XeN bond is slightly longer than XeO bond in the neutral molecule NXeOF3 by 0.003 Å. (b) Thermodynamic stability Table 3 shows the relative energies to the most stable atomic species of the molecules in this study at various theoretical levels. These energies can also be described as the total atomization energies (TAE) of the molecules. The NXeF5 , NXeOF4 , and NXeF4were found to have the highest TAE of ~104, 140, and 134 kcal/mol at CCSD(T)/aug-cc-pVTZ level, respectively. Table 3 also shows the TAEs obtained by the CCSD(T)/aug-cc-pVTZ//B3LYP/aug-cc-pVTZ calculations (CCSD(T)/aug-cc-pVTZ single point calculations using the B3LYP/aug-cc-pVTZ 112 structures) were very close to the CCSD(T)/aug-cc-pVTZ values for the NXeF5 and NXeF4. The TAEs listed in Table 3 are 57.5~139.7 kcal/mol at CCSD(T)/aug-cc-pVTZ//B3LYP/aug-cc-pVTZ level. In comparison, the atomization energy of XeNO2 and XeNO3 were 40.9 and 87.0 kcal/mol at the CCSD(T)/aug-cc-pVTZ level. The calculated triplet state energies relative to the ground singlet state (using the singlet structure) at various theoretical levels are listed in Table 4. At the singlet structures, the triplet states were 28.7~103.2 kcal/mol higher in energy at CCSD(T)/aug-cc-pVTZ//B3LYP/aug-cc-pVTZ level, and thus the calculated singlet state is the ground electronic state. The spin-orbital coupling could certainly have some effects on the ST gaps, relative energies, and molecular geometry. However, according to our pervious study, the typical spin-orbital energies of these species are on the order of 1 kcal/mol or less. Thus, to a good approximation, the spin-orbital coupling would not affect the calculated results significantly. (c) kinetic stability The possible unimolecular dissociation products on the singlet-state surface and their relative energies for the unimolecular dissociation channels at various theoretical levels are listed in Table 5. For the very endoergic unimolecular dissociation channels, the barriers were less important. For the exoergic unimolecular dissociation channels on the singlet-state surface, at least one transition state has been found for these molecules. The calculated barrier heights for these channels were also listed in Table 5. The transition state structures calculated by B3LYP/aug-cc-pVTZ method of these molecules were showed in Figure 3. The lowest unimolecular dissociation barriers of three neutral molecules NXeOF3, NXeF3 and NXeF5 were found be 15~21 kcal/mol 113 at the same level which were significantly lower than those of the XeNO2 or XeNO3( ~40 kcal/mol at CCSD(T)/aug-cc-pVTZ//MP2/aug-cc-pVDZ level). However, the dissociation barriers of the three anions NXeOF2, NXeO2F2 and NXeOF4 were significantly higher(26.0~36.2 kcal/mol). The TS of NXeOF4of the dissociation channel to XeF4 + NO(S) was not be found. However, the barrier of another similar dissociation channel NXeOF2 XeF2 + NO(S) was calculated to be 26.0 kcal/mol at CCSD(T)/aug-cc-pVTZ//B3LYP/aug-cc-pVTZ level. Thus, we believe that the dissociation barriers of NXeOF4 XeF4 + NO(S) would be quite similar if the TS exists. The barrier for the NXeF4dissociation to XeF3 + NF was predicted to be 27.3 kcal/mol, and this is an endoergic reaction by 19.7 kcal/mol. The only two exoergic unimolecular dissociation channels of NXeF4are reactions to XeF2 + NF2 and XeF + NF3. These two channels are unlikely to proceed by a concerted step (these would require breaking at least three bonds at the same time), and should involve very high barriers. On the other hand, the barrier of NXeF4 XeF3 + NF is 27.3 kcal/mol. This channel was involved with breaking only one XeF and one XeN bond. That is, we believe the dissociation barriers of NXeF4 into XeF2 + NF2 and XeF + NF3 will be significantly higher. The dissociation of NXeF4 to NXeF3 + F is endoergic by 76 kcal/mol, which indicates a very strong interaction between the neutral NXeF3 molecule and the fluoride ion. In our previous study,19 we have established a relationship between the stability 114 and the dissociation barrier heights for noble gas molecules. In order to have a half-life of ~102 seconds for spectroscopic study in the gas phase at 100, 200 and 300 K, the noble gas molecule with neighboring heavy atoms must at least have barriers of 9, 17, 25 kcal/mol respectively. Thus, these mono-negative charged species were predicted to be kinetically stable below 300 K, and the neutral molecules will be stable below 100K. (d) isoelectronic molecules Several isoelectronic molecules of NXeO2F2 , NXeOF4 , NXeOF2 and NXeF4 were also investigated in the current study. By replacing the N with an O atom, these molecules were XeO2F2, XeO3F2, XeO2F4 and XeOF4. The calculated structures of XeO2F2, XeO3F2, XeO2F4 and XeOF4, are depicted in Figure 3. At B3LYP/aug-cc-pVTZ level, the XeF and XO bonds in these molecules are all slightly shorter than their isoelectronic anions. For example, the XeF and XO bond lengths of XeO2F4 are 1.903 Å and 1.764 Å, respectively, which are 2.020 Å and 1.797 Å in NXeOF4. The total atomization energies (TAE) of these molecules were listed in Table 6, which were predicted to be ~20 kcal/mol higher than their isoelectronic anions. For example, the TAE of XeO2F4 and NXeOF4 are 158.9 and 139.7 kcal/mol, respectively at CCSD(T)/aptz//B3LYP/aptz level. This result was actually expected since the XeO bond could be slightly stronger than the XeN bond, as we discovered in the difference of TAE for XeNO2 and XeO3 in our previous study10. 115 Conclusions In the current study, we have predicted a new series of xenon containing noble-gas molecules, in particular, NXeOF3 , NXeF3 , NXeF5 , NXeOF2, NXeO2F2 , NXeOF4 and NXeF4. These molecules or anions showed strong bonding between xenon and nitrogen atoms with very short bond distances. Based on the calculated structures and the Lewis structures, these XeN bonding can be tentatively assigned as triple bonds. The total atomization energies (TAE) of the molecules are all higher than 50 kcal/mol, and the dissociation barriers are higher than 15 kcal/mol. These molecules were predicted to be stable at cryogenic condition, and could be targets of future experimental identification. This study showed that species obtained by replacing the oxygen atoms with fluorine atoms from NXeO2 or NXeO3 can also be stable. Thus it is also possible that other types of noble gas molecules or anions obtained by replacing the fluorine by other monovalent functional groups, such as CH3, C2H5, OH, Cl, NO2, etc., might also be stable at cryogenic conditions. In addition, the N and O atoms on NXeO2 or NXeO3 might also be able to bond to other atoms and form ring-type Xe molecules or even Xe-containing polymers. 116 Reference (1) Bartlett, N. Proc. Chem. Soc. 1962, 218. (2) Greenwood, N. N.; Earnshaw, A. In Chemistry of the Elements; Butterworth-Heinemann: Oxford, 2001; p 888. (3) Grochala, W. Chem. Soc. Rev. 2007, 36, 1632. (4) (a) Pettersson, M.; Lundell, J.; Räsänen, M. J. Chem. Phys. 1995,102, 6423. (b) Pettersson, M.; Lundell, J.; Räsänen, M. Eur. J. Inorg. Chem.1999, 729. (c) Khriachtchev, L.; Tanskanen, H.; Lundell, J.; Pettersson,M.; Kiljunen, H.; Räsänen, M. J. Am. Chem. Soc. 2003, 125, 4696. (d)Pettersson, M.; Khriachtchev, L.; Lundell, J.; Räsänen, M. J. Am. Chem.Soc. 1999, 121, 11904. (e) Khriachtchev, L.; Pettersson, M.; Lundell, J.;Tanskanen, H.; Kiviniemi, T.; Runeberg, N.; Räsänen, M. J. Am. Chem.Soc. 2003, 125, 1454. (f) Khriachtchev, L.; Isokoski, K.; Cohen, A.;Räsänen, M.; Gerber, R. B. J. Am. Chem. Soc. 2008, 130, 6114. (g)Pettersson, M.; Lundell, J.; Khriachtchev, L.; Räsänen, M. J. Chem. Phys.1998, 109, 618. (h) Pettersson, M.; Lundell, J.; Khriachtchev, L.; Isoniemi,E.; Räsänen, M. J. Am. Chem. Soc. 1998, 120, 7979. (i) Pettersson, M.;Khriachtchev, L.; Lundell, J.; Jolkkonen, S.; Räsänen, M. J. Phys. Chem.A 2000, 104, 3579. (j) Khriachtchev, L.; Räsänen, M.; Gerber, R. B. Acc.Chem. Res. 2009, 42, 183. (5) (a) Li, J.; Bursten, B. E.; Liang, B.; Andrews, L. Science 2002,295, 2242. (b) Andrews, L.; Liang, B.; Li, J.; Bursten, B. E. J. Am. Chem.Soc. 2003, 125, 3126. (c) Liang, B.; Andrews, L.; Li, J.; Bursten, B. E.Inorg. Chem. 2004, 43, 882. (6) (a) LeBlond, R. D.; DesMarteau, D. D. J. Chem. Soc., Chem.Commun. 1974, 555. (b) DesMarteau, D. D.; LeBlond, R. D.; Hossain, S. F.;Nothe, D. J. Am. Chem. Soc. 1981, 103, 7734. 117 (7) Grochala, W. Chem. Soc. Rev. 2007, 36, 1632. (8) (a) Sawyer, J. F.; Schrobilgen, G. J.; Sutherland, S. J. Inorg. Chem.1982, 21, 4064. (b) Schumacher, G. A.; Schrobilgen, G. J. Inorg. Chem.1983, 22, 2178. (c) Faggiani, R.; Kennepohl, D. K.; Lock, C. J. L.;Schrobilgen, G. J. Inorg. Chem. 1986, 25, 563. (d) Emara, A. A. A.;Schrobilgen, G. J. J. Chem. Soc., Chem. Commun. 1987, 1644. (9) (a) Fir, B. A.; Whalen, J. M.; Mercier, H. P. A.; Dixon, D. A.;Schrobilgen, G. J. Inorg. Chem. 2006, 45, 1978. (b) Smith, G. L.; Mercier,H. P. A.; Schrobilgen, G. J. Inorg. Chem. 2007, 46, 1369. (c) Smith, G. L.;Mercier, H. P. A.; Schrobilgen, G. J. Inorg. Chem. 2008, 47, 4173. (d)Smith, G. L.; Schrobilgen, G. J. Inorg. Chem. 2009, 48, 7714. (e) Hughes,M. J.; Mercier, H. P. A.; Schrobilgen, G. J. Inorg. Chem. 2009, 48, 4478. (10) Sun, Y.-L.; Hong, J.-T.; Hu, W.-P. J. Phys. Chem. A 2010, 114, 9359. (11) Krouse, I. H.; Hao, C.; Check, C. E.; Lobring, K. C.; Sunderlin,L. S.; Wenthold, P. G. J. Am. Chem. Soc. 2007, 129, 845. (12) Møller, C.; Plesset, M. S. Phys. ReV. 1934, 46, 618. (13) Raghavachari, K.; Trucks, G. W.; Pople, J. A.; Head-Gordon, M. Chem. Phys. Lett. 1989, 157, 479. (14) (a) Becke, A. D. J. Chem. Phys. 1993, 98, 1372. (b) Becke, A. D.J. Chem. Phys. 1993, 98, 5648. (15) Adamo, C.; Barone, V. J. Chem. Phys. 1998, 108, 664. (16) (a) Dunning, T. H., Jr. J. Chem. Phys. 1989, 90, 1007. (b) Kendall,R. A.; Dunning, T. H., Jr.; Harrison, R. J. J. Chem. Phys. 1992, 96, 6796.(c) Woon, D. E.; Dunning, T. H., Jr. J. Chem. Phys. 1993, 98, 1358. (d)Peterson, K. A. J. Chem. Phys. 2003, 119, 11099. (e) Peterson, K. A.;Figgen, D.; Goll, E.; Stoll, 118 H.; Dolg, M. J. Chem. Phys. 2003, 119, 11113. (17) Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb,M. A.; Cheeseman, J. R.; Montgomery, J. A., Jr.; Vreven, T.; Kudin, K. N.;Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.; Barone, V.;Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.;Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.;Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li,X.; Knox, J. E.; Hratchian, H. P.; Cross, J. B.; Bakken, V.; Adamo, C.;Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.;Cammi, R.; Pomelli, C.; Ochterski, J. W.; Ayala, P. Y.; Morokuma, K.;Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich,S.; Daniels, A. D.; Strain, M. C.; Farkas, O.; Malick, D. K.; Rabuck, A. D.;Raghavachari, K.; Foresman, J. B.; Ortiz, J. V.; Cui, Q.; Baboul, A. G.;Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz,P.; Komaromi, I.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.;Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson,B.; Chen, W.; Wong, M. W.; Gonzalez, C.; Pople, J. A. Gaussian03, revision D02; Gaussian, Inc.: Wallingford, CT, 2004. (18) Dixon, D. A.; de Jong, W. A.; Peterson, K. A.; Christe, K. O. J. Am. Chem.Soc. 2005, 127, 8627. (19) Li, T.-H.; Liu, Y-L.; Lin, R-J.; Yeh, T.-Y.; Hu, W.-P. Chem. Phys. Lett. 2007, 434, 38. 119 Table 1. The calculated frequencies at B3LYP/aug-cc-pVTZ level of several molecules in this study (a) NXeOF3 mode Frequencies (cm-1) IR Intensity NXeF scissoring OXeF and FXeF scissoring NXeO and FXeF scissoring FXeN wagging N wagging 132.6 159.9 234.2 283.8 294.1 0.6 0.0 8.9 2.7 1.8 OXeF and FXeF scissoring O wagging 294.1 333.8 493.1 534.1 560.4 796.8 883.2 24.0 6.1 3.5 101.8 218.0 26.5 8.2 FXe asymmetric stretching FXe symmetric stretching FXe asymmetric stretching OXe stretching NXe stretching 120 (b) NXeF3 mode Frequencies (cm-1) IR Intensity NXeF and FXeF scissoring FXeF wagging F wagging N wagging N wagging FXe asymmetric stretching FXe symmetric stretching FXe asymmetric stretching 111.2 147.6 182.0 207.9 272.7 452.9 496.3 532.3 3.1 5.2 0.2 23.5 1.9 37.1 83.5 264.6 NXe stretching 934.6 3.3 irreducible representation (mode) Frequencies (cm-1) IR Intensity E (FXeF wagging) B2 (FXeF twisting) B1 (FXeF scissoring) E (N wagging) 167.1 222.5 229.6 271.4 0.0 0.0 0.0 1.8 A1 (FXeF wagging) E (F wagging) 323.8 364.5 492.6 519.5 563.1 578.6 929.9 30.1 1.9 0.0 18.0 212.7 50.5 12.8 (c) NXeF5 B2 (FXe asymmetric stretching) A1 (FXe asymmetric stretching) E (FXe asymmetric stretching) A1 (FXe stretching) A1 (NXe stretching) 121 (d) NXeOF2 mode Frequencies (cm-1) IR Intensity FXeF scissoring FXeF wagging NXeO and FXeF twisting 117.5 138.1 17.4 18.4 193.0 0.6 NXeO wagging NXeO scissoring FXe symmetric stretching FXe asymmetric stretching 222.7 286.5 354.4 390.4 0.1 28.9 35.3 318.7 OXe stretching NXe stretching 733.1 901.9 91.0 55.6 irreducible representation (mode) Frequencies (cm-1) IR Intensity A' (OXeO rocking) A' (OXeF twisting) A' (NXeF wagging) A" (N wagging) 166.6 171.4 269.2 291.6 0.0 0.2 24.0 3.1 A' (OXeO scissoring) A" (OXeO twisting) A" (OXeO wagging) A' (FXe symmetric stretching) A" (FXe asymmetric stretching) A' (OXe symmetric stretching) A' (OXe asymmetric stretching) A' (NXe stretching) 310.8 311.3 334.8 398.4 460.3 731.6 771.5 866.5 32.0 0.0 6.1 8.0 320.6 37.4 107.9 55.6 (e) NXeO2F2 122 (f) NXeOF4 irreducible representation (mode) Frequencies (cm-1) IR Intensity E (FXeF wagging) B1 (FXeF scissoring) B2 (FXeF twisting) E (N wagging) B2 (FXe asymmetric stretching) A1 (FXe symmetric stretching) 123.0 173.0 216.0 307.5 308.8 356.8 390.5 442.6 0.1 0.0 0.0 3.1 27.3 2.0 0.0 13.7 E (FXe asymmetric stretching) A1 (OXe asymmetric stretching) A1 (NXe stretching) 484.2 737.9 866.4 309.2 27.5 30.1 irreducible representation (mode) Frequencies (cm-1) IR Intensity B2 (FXeF twisting) E (FXeF wagging) 115.5 126.2 161.4 0.0 0.3 0.0 194.1 257.0 351.5 432.4 447.2 965.4 36.1 0.5 0.0 40.5 327.9 20.0 A1 (FXeF wagging) E (O wagging) (g) NXeF4 B1 (FXeF scissoring) A1 (FXeF wagging) E (N wagging) B2 (FXe asymmetric stretching) A1 (FXe symmetric stretching) E (FXe asymmetric stretching) A1 (NXe stretching) 123 Table 2. Calculated Xe-N Bond Lengths (Å) of molecules in this study B3LYP/apdz B3LYP/aptz MPW1PW91/apTZ MP2/apTZ NXeOF3 1.814 1.783 1.763 1.755 NXeF3 1.806 1.773 1.754 1.791 NXeF5 1.791 1.761 1.744 1.732 NXeOF2 1.823 1.792 1.773 1.772 NXeO2F2 1.818 1.79 1.771 1.76 NXeOF4 1.811 1.78 1.761 1.747 NXeF4 1.785 1.757 1.74 1.768 124 CCSD(T)/aptz 1.758 1.754 Table 3. Calculated Total Atomization Energies (kcal/mol) of the molecules in this study B3LYP/apTZ MPW1PW91/apTZ MP2/apTZ NXeOF3 33.5 60.5 53.7 102.8 63.8 NXeF3 43.8 60.8 51.6 96.5 57.5 NXeF5 69.4 99.2 92.7 149.3 NXeOF2 51.8 71.1 65.8 96.9 70.6 NXeO2F2 61.0 89.8 87.7 128.6 96.2 NXeOF4 108.1 136.4 132.0 179.3 139.7 NXeF4 121.9 139.7 132.0 174.0 125 CCSD(T)/aptz CCSD(T)/aptz // B3LYP/aptz B3LYP/apDZ 104.8 134.3 103.7 133.8 Table 4. Calculated S-T gap (kcal/mol) of molecules in this study S-T gap B3LYP/apDZ B3LYP/apTZ MPW1PW91/apTZ MP2/apTZ CCSD(T)/aptz // B3LYP/aptz NXeOF3 12.0 20.7 26.6 68.6 33.4 NXeF3 33.3 43.3 41.8 84.7 50.5 NXeF5 4.5 14.9 20.6 46.7 28.7 NXeOF2 47.1 55.4 60.8 86.7 64.3 NXeO2F2 30.5 38.4 45.1 65.0 50.4 NXeOF4 15.7 26.0 32.4 55.2 38.3 NXeF4 72.5 66.8 70.8 155.7 103.2 126 Table 5. The calculated relative energies (kcal/mol) and the transition states (TS) for the unimolecular dissociation channels. CCSD(T)/aptz // B3LYP/aptz MP2/aptz B3LYP/aptz NXeOF3 0.00 0.00 0.00 XeOF2 + NF(S) 44.67 12.16 33.10 TS 21.27 46.75 26.13 XeF3(D) + NO(D) 166.56 119.68 143.12 TS 9.73 37.33 14.74 NXeOF + F2(S) 20.33 36.84 23.31 NXeF3 0.00 0.00 0.00 XeF2 + NF(S) 41.80 7.28 35.53 TS 19.00 54.11 18.71 NXeF + F2 46.84 88.42 51.13 NXeF5 0.00 0.00 0.00 XeF4 + NF(S) 63.93 29.26 45.90 TS 16.74 53.19 20.96 NXeF3 + F2(S) 1.30 10.90 9.97 127 CCSD(T)/aptz // B3LYP/apTZ MP2/apTZ B3LYP/aptz NXeOF2 0.00 0.00 0.00 XeF2 + NO(S) 45.59 20.13 33.89 TS 48.30 49.98 26.04 XeOF + NF(S) 1.29 26.11 4.57 TS 22.82 52.20 24.40 NXeO + F2 101.14 120.02 99.01 NXeO2F2 0.00 0.00 0.00 XeOF2 + NO(S) 29.43 0.36 12.20 TS 31.90 57.02 36.21 XeO2F + NF(S) 3.38 28.05 7.43 TS 28.05 52.32 33.32 NXeF2 + O2(S) 24.06 7.43 0.54 NXeO2 + F2 62.92 74.48 64.78 NXeOF4 0.00 0.00 0.00 XeOF3 + NF(S) 31.36 1.06 19.18 TS 25.43 N/A 32.46 XeF4 + NO(S) 40.75 12.45 21.37 NXeOF2 + F2 28.27 40.56 32.87 NXeOF3 + F 75.9 76.5 75.9 128 CCSD(T)/aptz // B3LYP/apTZ MP2/apTZ B3LYP/aptz NXeF4 0.00 0.00 0.00 XeF3 + NF(S) 13.75 48.95 19.67 TS 25.59 N/A 27.34 XeF2 + NF2 21.54 12.05 13.24 XeF + NF3 73.16 45.43 69.63 NXeF2 + F2 73.43 94.16 73.98 XeF4 + N-(T) 91.21 123.87 99.11 NXeF3 + F 78.9 77.5 76.2 129 Table 6. Calculated Atomization Energies (kcal/mol) of XeO2F2, XeO3F2, XeO2F4 and XeOF4 B3LYP/apDZ B3LYP/apTZ MPW1PW91/apTZ MP2/apTZ CCSD(T)/aptz // B3LYP/aptz XeO2F2 70.9 96.0 91.4 129.5 99.3 XeO3F2 69.0 103.6 101.1 152.4 114.6 XeO2F4 111.5 147.6 145.2 203.7 158.9 XeOF4 123.3 147.7 141.8 186.0 130 (a) NXeOF3 (CS symmetry) (b) NXeF3 (CS symmetry) (c) NXeF5 (C4v symmetry) (the CCSD(T)/aptz structure in parentheses) 131 132 (d) NXeOF2 (CS symmetry) (e) NXeO2F2 (CS symmetry) 133 (f) NXeOF4 (C4v symmetry) (g) NXeF4 (C4v symmetry) (the CCSD(T)/aptz structure in parentheses) 134 (h) XeNO3 (C3v symmetry) (CCSD(T)/aptz structure) Figure 1. The calculated structures of NXeOF3, NXeF3, NXeF5, NXeF2, NXeOF2, NXeO2F2, NXeF4 and XeNO3 at B3LYP/aug-cc-pVTZ level. 135 (a) XeO2F2 (b) XeO3F2 136 (c) XeO2F4 (d) XeOF4 Figure 2. Calculated structures of XeO3F2, XeO2F4, XeO2F2 and XeOF4 by B3LYP/aug-cc-pVTZ method 137 (a) NXeOF3 XeF3(D) + NO(D) (b) NXeOF3 XeOF2 + NF(S) 138 (c) NXeF3 XeF2 + NF(S) (d) NXeF5 XeF4 + NF(S) 139 (e) NXeOF2 XeF2 + NO(S) (f) NXeOF2 XeOF + NF(S) 140 (g) NXeO2F2 XeOF2 + NO(S) (h) NXeO2F2 XeO2F + NF(S) 141 (i) NXeOF4 XeOF3 + NF(S) (j) NXeF4 XeF3 +NF(S) Figure 3 Transition state structures calculated by B3LYP/aug-cc-pVTZ. 142