jgrd52446-sup-0001-supinfo
advertisement

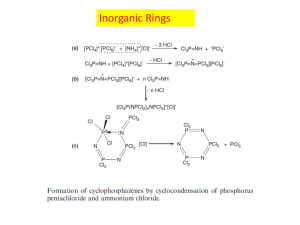
Journal of Geophysical Research-Atmospheres Supporting Information for Chemical imaging of ambient aerosol particles: observational constraints on mixing state parameterization Rachel E. O’Brien,1,2# Bingbing Wang,3 Alexander Laskin,3 Nicole Riemer,4 Matthew West,5 Qi Zhang,6 Yele Sun,6 Xiao-Ying Yu,7 Peter Alpert,8% Daniel Knopf,8 Mary K. Gilles,2 Ryan Moffet,1* [1] Department of Chemistry, University of the Pacific, Stockton, CA 95211, USA [2] Lawrence Berkeley National Laboratory, Berkeley, California, 94720-8198, USA # Now at, Department of Civil and Environmental Engineering, Massachusetts Institute of Technology, Cambridge, MA, 02139, USA [3] William R. Wiley Environmental and Molecular Sciences Laboratory, Pacific Northwest National Laboratory, Richland, WA, 99352, USA [4] Department of Atmospheric Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA [5] Department of Mechanical Science and Engineering, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA [6] Department of Environmental Toxicology, University of California Davis, Davis, CA 95616, USA [7] Fundamental and Computational Sciences Directorate, Pacific Northwest National Laboratory, Richland, WA, 99354, USA [8] Institute for Terrestrial and Planetary Atmospheres, School of Marine and Atmospheric Sciences, Stony Brook University, Stony Brook, New York, 11794-5000, USA % Now at, Institut de Recherches sur la Catalyse et l’Environnement de Lyon, Centre National de la Recherche Scientifique, Université Claude Bernard Lyon 1, 69626 Villeurbanne, France Contents of this file Text S1 Figure S1 Tables S1-S2 Additional Supporting Information (Files uploaded separately) Captions for Tables S3 to S8 Introduction 1 Text includes additional information on the calculation of the composition for the mixed inorganic and the combination of the two techniques. Figure S1 shows bulk mass fractions for T0 and T1 with the concentration of organic and elemental carbon (soot or BC) measured during the same hour overlaid on top. Tables S1-S8 provides data supporting the figures in the main text. Text S1. Mixed inorganic composition calculation The assumption that ammonium sulfate is the inorganic component leads to the largest uncertainty in the mass calculations. To test the effect of different assumed inorganic compositions, an analysis was carried out using a mixture of typical inorganic components: (NH4)2SO4, Na2SO4, NH4NO3, and NaNO3. Aerosol Mass Spectrometry (AMS) [Setyan et al., 2012] and Particle into Liquid Samplers (PILS) [Zaveri et al., 2012] were co-located at both field sites and provided quantitative measurement of ions and anions in aerosols. The AMS has a size cut-off of ~1 m vacuum aerodynamic diameter and the PILS has a pre-impactor with a d50 of 1 m [Sorooshian et al., 2006]. AMS and PILS data at T0 and T1 were used to estimate the mole ratios of the ions (SO42- : NO3- and NH4+: Na+). The measurements with the AMS at T0 stopped a few days before the samples discussed here so the AMS concentrations for T0 are taken from the average values a few days prior (6/22/2010-6/25/2010) [Zaveri et al., 2012]. From AMS measurements during the same time stamps (T1) or a few days prior (T0), the ratio of SO42- to NO3- averaged 2-5. The charge balance of NH4+ to SO42- and NO3- was nearly even at T1 over the 27th and 28th ( 1.1 on average) consistent with AMS results that the particles were fully neutralized at T1 [Setyan et al., 2012]. At T0 for the 22nd- 25th, the charge balance was slightly lower (0.93 on average). To compare Na+ to NH4+, PILS data was used and the NH4+ concentration was estimated by taking the SO42- and NO3- concentrations from the PILS data and calculating the concentration of NH4+ needed to balance the charges. Using this estimate, there was approximately 1-3 times as much NH4+ as Na+ from the PILS data at T0 and T1. The ratios for NH4+ to Na+ of 3:1 and for SO42- to NO3- of 2:1 provided the largest difference in the mass absorption coefficient compared to (NH4)2SO4 as the inorganic component. Thus, these ratios were used giving estimated ratios for the mixed inorganics of 6:2:3:1 for (NH4)2SO4:Na2SO4:NH4NO3:NaNO3. These ratios lead to weighted absorption coefficients of in*,320 = 9,470 cm2/g and in*,278 = 12,300 cm2/g [Henke et al., 1993] and a density of 1.95 g/cm3. These values were used for new mass and mixing state parameter calculations. The difference between the values determined using the mixed inorganic and ammonium sulfate are shown as error bars on Figure 2 in the main text. STXM/NEXAFS and CCSEM/EDX and future work The analysis presented in this manuscript was carried out on data sets collected for previous publications [Knopf et al., 2014, Moffet et al., 2013]. For those projects, the data from STXM/NEXAFS and CCSEM/EDX were collected on samples from the same two days but were not collected on the same samples. Thus, the analysis presented in this manuscript provides insights into the information that can be gained from each of the techniques separately. Future work in this area, however, would greatly benefit from the application of both techniques to the same samples and, in the best case, to the same particles. The application of both techniques to the same samples would provide C, N, and O 2 quantification from STXM/NEXAFS and quantification of the other elements from CCSEM/EDX. This data would reduce the uncertainty in the identity of the inorganic used for the STXM calculations. The application of both techniques to the same particles would be a more time intensive process but would enable a per-particle mass based mixing state analysis for the full elemental composition. Additionally, the elemental composition could be apportioned to expected inorganic components. This analysis could provide the ability to estimate the O/C of the organic component which could provide additional information on aerosol sources and aging. References Henke, B. L., E. M. Gullikson and J. C. Davis (1993), X-Ray Interactions Photoabsorption, scattering, transmission and reflection at E=50-30,000 eV, Z=1-92 (VOL 54, PG 181, 1993). Atom. Data Nucl. Data Tables 55(2): 349-349. Knopf, D. A., P. A. Alpert, B. Wang, et al. (2014), Microscpectriscopic imaging and characterization of individually identified ice nucleating particles from a case field study. J. Geophys. Res.-Atmos. 119 (17), doi: 10.1002/2014JD021866. Moffet, R. C., T. C. Roedel, S. T. Kelly, et al. (2013), Spectro-microscopic measurements of carbonaceous aerosol aging in Central California. Atmos. Chem. Phys. 13(20): 10445-10459. Setyan, A., C. Song, M. Merkel, et al. (2014), Chemistry of new particle growth in mixed urban and biogenic emissions - insights from CARES. Atmos. Chem. Phys. 14(13): 6477-6494. Sorooshian, A., F. J. Brechtel, Y. L. Ma, et al. (2006), Modeling and characterization of a particle-into-liquid sampler (PILS). Aerosol Sci. Technol. 40(6): 396-409. Zaveri, R. A., W. J. Shaw, D. J. Cziczo, et al. (2012), Overview of the 2010 Carbonaceous Aerosols and Radiative Effects Study (CARES). Atmos. Chem. Phys. 12(16): 7647-7687. 3 Figure S1. Bulk mass fractions for T0 (a, b) and T1 (c, d) for organic carbon (OC, green), inorganic (IN, blue),and black carbon (BC, red). The concentration of OC and BC (also called elemental carbon or EC) measured during the same hour with a collocated Sunset ECOC are shown with white markers on the right axis. For the sample collected at T1 6/28/2010 at 11:37 am there was no corresponding ECOC measurement. 4 size range (um) 0.001-0.09 0.1-0.19 0.2-0.29 0.3-0.39 0.4-0.49 0.5-0.59 0.6-0.69 0.7-0.79 0.8-0.89 0.9-0.99 1.0-1.09 1.1-1.19 1.2-1.29 1.3-1.39 1.4-1.49 1.5-1.59 1.6-1.69 1.7-1.79 1.8-1.89 1.9-1.99 2.0-2.09 2.1-2.19 2.2-2.29 2.3-2.39 number of particles 24 944 1357 1263 1214 927 766 560 386 307 232 182 115 97 70 60 47 34 27 29 26 19 21 16 Average organic mass (fg) 0.11 ± 0.035 0.46 ± 0.26 1.3 ± 0.62 2.8 ± 1.3 5.5 ± 2.6 9.6 ± 5.1 15 ± 6.6 20 ± 9.8 30 ± 15 38 ± 19 47 ± 21 61 ± 32 72 ± 31 88 ± 44 130 ± 72 140 ± 83 160 ± 87 140 ± 50 160 ± 32 230 ± 105 220 ± 104 250 ± 67 240 ± 62 380 ± 178 Average inorganic mass (fg) 0.40 ± 0.070 0.73 ± 0.55 2.2 ± 1.6 4.6 ± 3.3 8.2 ± 5.2 14 ± 8.8 21 ± 13 29 ± 18 44 ± 31 59 ± 36 83 ± 49 100 ± 53 140 ± 83 180 ± 110 230 ± 120 240 ± 120 330 ± 190 360 ± 160 420 ± 150 570 ± 260 650 ± 300 730 ± 230 730 ± 370 1100 ± 400 Average black carbon mass (fg) 0.00 ± 0.004 0.030 ± 0.15 0.13 ± 0.51 0.31 ± 1.1 0.47 ± 1.8 0.95 ± 3.4 1.3 ± 4.6 1.8 ± 6.7 2.2 ± 8.4 3.9 ± 15 3.8 ± 16 5.9 ± 24 1.8 ± 4.5 4.7 ± 21 3.5 ± 12 4.1 ± 18 0.72 ± 2.0 24 ± 100 22 ± 76 0.87 ± 3.1 13 ± 31 8.2 ± 24 2.6 ± 7.4 12 ± 37 Average particlespecific diversity, Di 1.7 ± 0.12 1.9 ± 0.24 1.9 ± 0.27 1.9 ± 0.29 1.9 ± 0.27 1.9 ± 0.26 2.0 ± 0.27 1.9 ± 0.24 1.9 ± 0.24 1.9 ± 0.25 1.9 ± 0.24 1.9 ± 0.24 1.9 ± 0.19 1.9 ± 0.19 1.9 ± 0.18 1.9 ± 0.20 1.8 ± 0.15 1.9 ± 0.23 1.8 ± 0.21 1.8 ± 0.13 1.8 ± 0.20 1.8 ± 0.21 1.8 ± 0.14 1.7 ± 0.11 Table S1. Size separated data for T0, STXM/NEXAFS analysis. The average masses of organic, inorganic, and black carbon are given in femtograms. The standard deviation is given for each size bin. Size bins with less than 10 particles were not included in the figure. 5 size range (um) 0.001-0.09 0.1-0.19 0.2-0.29 0.3-0.39 0.4-0.49 0.5-0.59 0.6-0.69 0.7-0.79 0.8-0.89 0.9-0.99 1.0-1.09 1.1-1.19 1.2-1.29 1.3-1.39 1.4-1.49 1.5-1.59 1.6-1.69 1.7-1.79 1.8-1.89 Average number of organic particles mass (fg) 0.12 ± 9 0.040 632 1002 1123 986 846 671 560 408 301 212 160 118 66 44 30 27 15 11 0.48 ± 0.27 1.3 ± 0.67 3.0 ± 1.4 5.5 ± 2.7 9.5 ± 4.5 15 ± 7.3 23 ± 11 33 ± 17 45 ± 22 56 ± 28 74 ± 34 93 ± 49 130 ± 57 140 ± 65 180 ± 75 220 ± 120 230 ± 130 230 ± 110 Average inorganic mass (fg) Average black carbon mass (fg) 0.28 ± 0.17 0.00 ± 0.00 0.012 ± 0.092 0.079 ± 0.33 0.21 ± 0.85 0.34 ± 1.3 0.50 ± 1.9 0.69 ± 2.8 0.99 ± 3.6 1.3 ± 5.7 2.0 ± 8.1 2.9 ± 13 3.6 ± 11 3.2 ± 11 1.3 ± 3.0 1.9 ± 4.8 0.72 ± 1.9 4.9 ± 15 3.9 ± 7.3 1.7 ± 3.0 0.39 ± 0.31 1.0 ± 0.71 2.2 ± 1.4 4.4 ± 2.8 7.3 ± 4.1 11 ± 6.0 17 ± 11 24 ± 13 31 ± 15 43 ± 29 53 ± 33 69 ± 48 93 ± 71 85 ± 38 110 ± 54 140 ± 74 150 ± 69 190 ± 100 Average particlespecific diversity, Di 1.7 ± 0.17 1.8 ± 0.20 1.9 ± 0.26 1.9 ± 0.25 1.9 ± 0.26 2.0 ± 0.26 2.0 ± 0.24 2.0 ± 0.25 2.0 ± 0.21 2.0 ± 0.24 2.0 ± 0.21 2.0 ± 0.26 2.0 ± 0.25 1.9 ± 0.13 2.0 ± 0.18 1.9 ± 0.09 2.0 ± 0.24 1.9 ± 0.19 1.9 ± 0.20 Table S2. Size separated data for T1, STXM/NEXAFS analysis. The the average masses of organic, inorganic, and black carbon are given in femtograms. The standard deviation is given for each size bin. Size bins with less than 10 particles were not included in the figure. 6 Table S3. Size separated data for T0, CCESM/EDX analysis. The average mass of the elements (in femtograms) are given with ± the standard deviation. Table S4. Size separated data for T1, CCSEM/EDX analysis. The average mass of the elements (in femtograms) are given with ± the standard deviation. Table S5. STXM/NEXAFS data from Cares T0 6/27/2010 and 6/28/2010. The sample day and time are given. Total mass (in femtograms) of organic (OC), inorganic (IN) and black carbon/soot (BC) per particle. Labels correspond to: 1 = OC, 2 = INBCOC, 3 = BCOC, 4 = INOC; the size of the particle is give as the area equivalent diameter (AED) in microns Table S6. STXM/NEXAFS data from Cares T1 6/27/2010 and 6/28/2010. The sample day and time are given. Total mass (in femtograms) of organic (OC), inorganic (IN) and black carbon/soot (BC) per particle. Labels correspond to: 1 = OC, 2 = INBCOC, 3 = BCOC, 4 = INOC; the size of the particle is give as the area equivalent diameter (AED) in microns Table S7. CCSEM/EDX data from Cares T0 6/27/2010 and 6/28/2010. The sample day and time are given. Total mass (in femtograms) of each element for each particle are given. Labels correspond to: 1 = sea salt, 2 = sea salt/sulfate, 3 = CNOS (carbonaceous/sulfate), 4 = CNO (carbonaceous), 5 = other; the size of the particle is give as the average diameter in microns Table S8. CCSEM/EDX data from Cares T1 6/27/2010 and 6/28/2010. The sample day and time are given. Total mass (in femtograms) of each element for each particle are given. Labels correspond to: 1 = sea salt, 2 = sea salt/sulfate, 3 = CNOS (carbonaceous/sulfate), 4 = CNO (carbonaceous), 5 = other; the size of the particle is give as the average diameter in microns 7