Heat Transfer: Conduction, Convection, Radiation Explained
advertisement

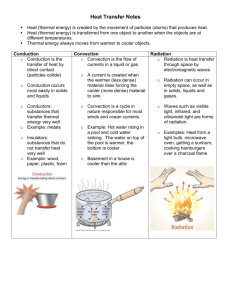
The Transfer of Heat energy – the ability to make things move or change thermal energy – how heat moves from one object to another conductor – a material through which heat can move easily *examples – stainless steel, iron, silver, copper, aluminum insulator – a material that heat does not move through easily *examples – rubber, brick, wood How Heat Transfers Heat always moves from a warmer place to a cooler place. Hot objects in a cooler room will cool to room temperature. Cold objects in a warmer room will heat up to room temperature. Heat transfers in three ways o conduction o convection o radiation Conduction the transfer of heat between things that are touching objects are in direct contact with each other occurs when a substance is heated – o particles gain energy and vibrate more. o these particles bump into nearby particles and transfer some of their energy to them metal (iron, silver, copper, stainless steel) = good conductor Example of Conduction o heating a pot of water on an electric stove – heat moves from the hot surface of the stove to the metal of the pot that is touching the stove Convection the transfer of heat by the movement of the heated parts of a liquid or gas the transfer of heat by the actual movement of the warmed matter occurs when warmer areas of liquid or gas rise to cooler areas in the liquid or gas o Examples of Convection water boiling in a pan heat is passed by movements called convection currents molecules in liquids and gases move apart when they are heated. convection current - movement of heated molecules to cooler areas Radiation the transfer of energy by electromagnetic waves electromagnetic waves can transfer energy through matter and also through empty space The sun is the earth’s main source of electromagnetic waves Examples of radiation o heat from the sun warming your face o heat from an open fire o heat from a light bulb kinetic energy – energy that is in motion potential energy – possible or stored energy