Exploring Density Worksheet: Simulation & Calculations
advertisement

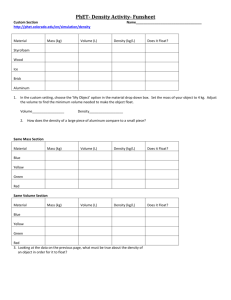
Name: Class: Date: Exploring Density Simulation Exploration: Login to the PHET Website. A basic property of any homogeneous substance is its density: the ratio of its mass to its volume. The density of a liquid or solid is an intensive property, which means that the density does not vary with the size of the sample but only depends upon its composition. The determination of density is a nondestructive physical process for distinguishing one substance from another. The mathematical relationship is: 𝐷𝑒𝑛𝑠𝑖𝑡𝑦 = 𝑚𝑎𝑠𝑠 𝑣𝑜𝑙𝑢𝑚𝑒 . Part A: 1. Find the blocks tab in the right hand corner. Click on the “Same Mass” tab. 2. Insert the blocks one at a time in the pool of water. Observe the change in the water level. a. Note: Only 1 block should be in the water at any given time. 3. Completely submerge the blocks that float. Observe the change in the water level. Calculate the density of each block: Block Yellow Mass Volume Density Float or Sink Blue Green Red Part B: 1. Find the blocks tab in the right hand corner. Click on the “Same Volume” tab. 2. Insert the blocks one at a time in the pool of water. Observe the change in the water level. a. Note: Only 1 block should be in the water at any given time. 3. Completely submerge the blocks that float. Observe the change in the water level. Block Yellow Mass Volume Blue 1 Density Float or Sink Name: Class: Date: Green Red Part C: 1. Use the scale and the pool to determine the density of each of the blocks. Block Mass Volume Density A Float or Sink B C D E 2. Click on “Show Table”. Use the density values in the table to determine the unknown substance in each block. Write it next to the letter in the table above. 3. Your friend has three blocks (A, B, and C) of the same size, but they each float differently in water. A C B a. What do you think is making them float differently? ____________________________________________________________________________________ ____________________________________________________________________________________ 2 Name: Class: Date: POST LAB QUESTIONS and ANALYSIS 1. You have 5 blocks that are the same size, but different masses. The lightest one is 1kg, the heaviest one is 5kg. The picture shows how the 2kg and 5kg blocks float and sink in water. On the picture, draw where the other blocks would end up if you put them in the water. Explain why you think it would look that way? 2 kg _____________________________________________ _____________________________________________ _____________________________________________ _____________________________________________ 5 kg (Adapted from Univ. of Washington assessment question) 2. This picture shows a large block that sinks in the water, and a much smaller block of the exact same material sitting outside of the water. On the picture, draw what you think would happen if you put the smaller block into the water. Would it float or sink, or does it depend? __________________________________________________________ The density of the smaller block is _________________ the density of the larger block. (smaller than, same as, larger than) 3 Name: Class: 3. You have 5 blocks of different shapes, different sizes, and different materials. You put them in water and see that some float and some sink (see picture). Date: A D B Can you tell which one has the smallest density? _____ If so, which one: ______ C Explain how you can tell: ______________________________________________ ______________________________________________ E Can you tell which one has the largest density? _____ If so, which one: ______ Explain: ____________________________________________________________________________________ In the empty picture, redraw the blocks in the water in order from the smallest density one to the largest density one. If you don’t have enough information, explain what information you would need. Smallest Density 4 Largest Density