Identification and expression of Acetylcholinesterase in Octopus
advertisement

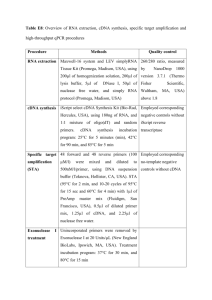
1 SUPPLEMENTAL INFORMATION 2 3 4 5 Identification and expression of Acetylcholinesterase in Octopus vulgaris arm development and regeneration: a conserved role? 6 7 8 Sara Maria Fossati1, Simona Candiani2, Marie-Therese Nödl1, Luca Maragliano1, Maria Pennuto1,3, Pedro Domingues4, Fabio Benfenati1,5, Mario Pestarino2, Letizia Zullo1 9 1 Istituto Italiano di Tecnologia, Department of Neuroscience and Brain Technologies, Genova, Italy 10 2 University of Genova, DISTAV, Genova, Italy 11 3 Dulbecco Telethon Institute, Lab of Neurodegenerative Diseases, CIBIO, University of Trento, Italy 12 4 Instituto Español de Oceanografía, Centro Oceanográfico de Vigo, Vigo, Spain 13 5 Department of Experimental Medicine, University of Genova, Genova, Itally 14 15 16 Corresponding author: Sara Maria Fossati, Via Morego 30, 16163, Genoa, Italy, Telephone number: +39 3475752235, Fax number: +39 010 71781230, e-mail: sara.fossati@iit.it 17 18 19 20 Contents: Supplemental Experimental Procedures (Adult animal treatment, O. vulgaris ACHE gene cloning and identification, Synthesis of DIG-labeled ACHE RNA probe for in situ hybridization, In situ hybridization, RT-PCR, Molecular Dynamics Simulations), Supplemental References, Supplemental Figure Captures. 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 1 1 SUPPLEMENTAL EXPERIMENTAL PROCEDURES 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 Adult animal treatment Following capture, adult animals were each placed in a 120x50x45 cm marine aquarium. The tanks were filled with artificial sea water (ASW) and kept at a temperature of 18°C on 12 h light/dark cycle. Water cleaning and oxygenation was ensured by a pump-filter and aeration system which continuously circulated the water through biological filters and all relevant chemo/physical water parameters were constantly checked. Animals were left to adapt to captivity for at least 10 days before experimentation. The experimental animals were selected on the basis of the following criteria: healthy shape (all the arms and body parts intact), the animal showed normal reflexes and voluntary movements (e.g., arm extension, walking, etc.), regular eating and motivation to attack prey. Following surgery, each animal was replaced in the experimental tank where it slowly recovered from anesthesia (about 2-5 minutes). All the animals employed in this study did not display behavioral modifications after the operation, and their amputated arms regenerated as in the natural environment [1]. At the end of the experiments, the possibility of returning the animals to their natural environment was evaluated on the basis of the animal’s health. Animals were then either released into the wild at suitable habitats far from the collecting sites or received a terminal overdose of anesthetic. Our research conformed to the ethical principles of replacement, reduction, refinement and minimization of suffering [2]. Particular attention was given to the method of capture from the wild, the housing, the animal care and health monitoring program as well as to identifying signs of pain or distress in the animals. Animal experimentation was conducted following the guidelines reported in the European Directive 86/609/EEC. 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 In situ hybridization Both sample sections and embryos were incubated with a DIG-labeled ACHE probe at 60°C overnight. Endogenous peroxydase inactivation and SSC washes were not performed. O. vulgaris ACHE gene cloning and identification Given the absence of an O. vulgaris Genebank database, the cloning of the ACHE full length cDNA from O. vulgaris arm was obtained from degenerate primers whose design was based on the deposited sequences from Loligo vulgaris (GenBank Accession No. AF065384; [3]) and Electrophorus electricus (GenBank Accession No. AF030422; [4]). The degenerate primers were designed on the basis of three highly conserved regions (EDCLYLN, FGESAG and WPEWVG) (Fig. S3). The sequencing of the full length ACHE cDNA was continued by performing 5’ and 3’ RACE-PCR. The 5' RACE reaction was carried out according to the instruction manual of the 5’ RACE System (Invitrogen) using AAP and GSP2 primers (FIGURE S2). The 3' RACE reaction was carried out according to the instruction manual of the 3’ RACE System (Invitrogen). The 3' end of cDNA was amplified using UAP and GSP primers (Fig. S2). Synthesis of DIG-labeled ACHE RNA probe for in situ hybridization The cDNA obtained by cloning the PCR product of about 1400 bp was used as a template for in vitro transcription by a Boehringer Mannheim DIG RNA labeling Kit, according to the supplier's instructions. The restriction site used for the construction of the probe for in situ hybridization is shown in Fig. S3. The insert was linearized with BamHI and the probe was synthesized using SP6 RNA polymerase. RT-PCR Each sample was replicated three times in each real-time PCR experiment to capture intra-assay variability. PCR amplifications were performed with Power SYBR Green Master Mix (Qiagen) in a 7900HT Fast Real-Time PCR System (Applied Biosystems). Each assay included a no-template control for each primer pair and a dissociation curve analysis was performed to detect the presence of non-specific products. All data were analyzed using the 2-DDCT method. Molecular Dynamics Simulations The system to be modeled comprised the ACHE protein model, discussed above, immersed in a box of TIP3P model water molecules [5] and counter ions to neutralize the total charge, making a total of 85,618 atoms. Residues’ protonation states were determined using HHpred [6]. Periodic boundary conditions were used to replicate the system and remove box surface effects [7]. Short-range non-bonded interactions were cut off at 12 Å, while long-range electrostatic interactions were computed using the Particle Mesh Ewald (PME) method [8]. Chemical bonds connecting hydrogen atoms to heavy atoms were kept fixed using SHAKE [9]. The integration time step was 1 fs to ensure stability of the dynamics algorithm. The proper size for the simulation box corresponding to a pressure of 1 atm was obtained by simulating the system in the constant pressure and temperature (NPT) ensemble using the Nosé-Hoover Langevin piston 2 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 method for 0.5 ns [10]. The simulation ensemble was then switched to constant volume and temperature (NVT) for the rest of the simulation by keeping the temperature stationary around 300K using Langevin dynamics. The full simulation run lasted 60 ns. 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 Fig. S1 Percent identity and divergence were calculated by using the ClustalW algorithm provided in the MEGALIGN module. Percent identity compares sequences directly without accounting for phylogenetic relationships. Percent divergence is calculated by comparing sequence pairs in relation to the phylogeny reconstructed by MegAlign (Neighbor Joining method). Sequences used in the phylogenetic analysis with the corresponding accession numbers are also reported. Fig. S2 Sequences (FW: forward; REV: reverse) of primers used for cloning and RT-qPCR experiments. According to GenBank (http://www.ncbi.nlm.nih.gov/genbank/) for each gene is listed the accession number and the product size. Fig. S3 Nucleotide and deduced amino acid sequences of ACHE cDNA from O. vulgaris. Red asterisk indicates the stop codon. The regions where primers were designed are underlined. Red underlined: primers corresponding to the EDCLYLN, FGESAG and WPEWVG regions; orange underlined, Gene Specific Primer used for 5’RACE-PCR; green underlined, Gene Specific Primer used for 3’RACE-PCR; dashed blue underlined, BamH1 restriction site used for the construction of the probe for in situ hybridization. Fig. S4 Volume representation of the catalytic gorge and protein stability. A) Volume representation of the catalytic gorge (gray). The PAS (orange, top left) and CAT (red, bottom right) residues are shown as sticks. B) Backbone RMSD of O. vulgaris ACHE from the starting conformation along the simulated trajectory for the full protein (black), the CAT (red) and the PAS (orange) residues. Fig. S5 Confocal stacks through the midline of developing arm buds stained for Phallacidin (a-d) and DAPI (e-h). Arm buds are shown in a top view, except for stage 29, which is a side view (proximal to the left). a, e: stage 21; b, f: stage 23; c, g: stage 25; d, h: stage 29. Scale bars: 100 µm. lm: longitudinal muscle; tm: transversal muscle; m: future muscle layer; e: epithelium; anc: axial nerve cord. Movie S1 Movie showing O. vulgaris ACHE protein conformation and the localization of the active residues. SUPPLEMENTAL REFERENCES 1. Crook RJ, Lewis T, Hanlon RT, Walters ET (2011) Peripheral injury induces long-term sensitization of defensive responses to visual and tactile stimuli in the squid Loligo pealeii, Lesueur 1821. The Journal of experimental biology 214 (Pt 19):3173-3185. doi:10.1242/jeb.058131 2. Fiorito G, Affuso A, Anderson DB, Basil J, Bonnaud L, Botta G, Cole A, D'Angelo L, De Girolamo P, Dennison N, Dickel L, Di Cosmo A, Di Cristo C, Gestal C, Fonseca R, Grasso F, Kristiansen T, Kuba M, Maffucci F, Manciocco A, Mark FC, Melillo D, Osorio D, Palumbo A, Perkins K, Ponte G, Raspa M, Shashar N, Smith J, Smith D, Sykes A, Villanueva R, Tublitz N, Zullo L, Andrews P (2014) Cephalopods in neuroscience: regulations, research and the 3Rs. Invertebrate neuroscience : IN. doi:10.1007/s10158-013-0165-x 3. Talesa V, Grauso M, Arpagaus M, Giovannini E, Romani R, Rosi G (1999) Molecular cloning and expression of a full-length cDNA encoding acetylcholinesterase in optic lobes of the squid Loligo opalescens: a new member of the cholinesterase family resistant to diisopropyl fluorophosphate. Journal of neurochemistry 72 (3):1250-1258 4. Simon S, Massoulie J (1997) Cloning and expression of acetylcholinesterase from Electrophorus. Splicing pattern of the 3' exons in vivo and in transfected mammalian cells. The Journal of biological chemistry 272 (52):33045-33055 5. Jorgensen W, Chandrasekhar J, Madura J, Impey R, Klein M (1983) Comparison of simple potential functions for simulating liquid water J Chem Phys 79:926-935 6. Soding J, Biegert A, Lupas AN (2005) The HHpred interactive server for protein homology detection and structure prediction. Nucleic acids research 33 (Web Server issue):W244-248. doi:10.1093/nar/gki408 7. Frenkel D, Smit B (2001) Understanding Molecular Simulation: From Algorithms to Applications. Academic Press:664 8. Darden TA, Pedersen LG (1993) Molecular modeling: an experimental tool. Environmental health perspectives 101 (5):410-412 9. Ryckaert J, Ciccotti G, Berendsen H (1977) Numerical Integration of the Cartesian Equations of Motion of a System with Constraints: Molecular Dynamics of n-Alkanes Journal of Computational Physics 23 (3):327–341 10. Martyna G, Tobias D, Klein M (1994) Constant pressure molecular dynamics algorithms J Chem Phys 101 4177– 4189 SUPPLEMENTAL FIGURE CAPTURES 55 3