Citric Acid in Popular Drinks
advertisement

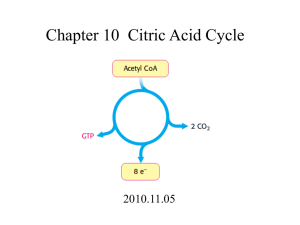
Part 1 - Get a Lab Appointment and Install Software: Find the email from your instructor with the URL (link) to sign up at the scheduler. Set up your scheduling system account and schedule your lab appointment. NOTE: You cannot make an appointment until two weeks prior to the start date of this lab assignment. You can get your username and password from your email to schedule within this time frame. Install the Citrix software: – go to http://receiver.citrix.com and click download > accept > run > install (FIRST TIME USING NANSLO). You only have to do this ONCE. Do NOT open it after installing. It will work automatically when you go to your lab. (more info at http://www.wiche.edu/info/nanslo/creative_science/Installing_Citrix_Receiver_Program.pdf) Scheduling Additional Lab Appointments: Get your scheduler account username and password from your email. Go to the URL (link) given to you by your instructor and set up your appointment. (more info at http://www.wiche.edu/nanslo/creative-science-solutions/students-scheduling-labs) Changing Your Scheduled Lab Appointment: Get your scheduler account username and password from your email. Go to http://scheduler.nanslo.org and select the “I am a student” button. Log in to go to the student dashboard and modify your appointment time. (more info at http://www.wiche.edu/nanslo/creative-science-solutions/students-scheduling-labs) Part 2 – Before Lab Day: Read your lab experiment background and procedure below, pages 1-8. Submit your completed Pre-Lab Questions (page 4-5) per your faculty’s instructions. Watch Titration Apparatus Control Panel Video Tutorial http://www.wiche.edu/nanslo/lab-tutorials#titration Part 3 – Lab Day Log in to your lab session – 2 options: 1)Retrieve your email from the scheduler with your appointment info or 2) Log in to the student dashboard and join your session by going to http://scheduler.nanslo.org NOTE: You cannot log in to your session before the date and start time of your appointment. Use Internet Explorer or Firefox. Click on the yellow button on the bottom of the screen and follow the instructions to talk to your lab partners and the lab tech. Remote Lab Activity SUBJECT SEMESTER: ____________ TITLE OF LAB: Citric Acid in Popular Drinks – Titration Lab format: This lab is a remote lab activity. Relationship to theory (if appropriate): In this lab you will apply your knowledge of acid/base titrations to determine the amount of citric acid in various drinks. This activity is meant to be done after a student has a basic understanding of titrations. Instructions for Instructors: This protocol is written under an open source CC BY license. You may use the procedure as is or modify as necessary for your class. Be sure to let your students know if they should complete optional exercises in this lab procedure as lab technicians will not know if you want your students to complete optional exercises. Instructions for Students: Read the complete laboratory procedure before coming to lab. Under the experimental sections, complete all pre-lab materials before logging on to the remote lab. Complete data collection sections during your online period, and answer questions in analysis sections after your online period. Your instructor will let you know if you are required to complete any optional exercises in this lab. Remote Resources: Primary – Titration Apparatus; Secondary – Acetic Acid Titration CONTENTS FOR THIS NANSLO LAB ACTIVITY: Learning Objectives........................................................................................................ 2 Background Information ............................................................................................... 2-3 Estimating Error ............................................................................................................ 3-4 Pre-lab Questions .......................................................................................................... 4-5 Equipment ..................................................................................................................... 5 Preparing for this NANSLO Lab Activity ........................................................................ 6 Experimental Procedure ............................................................................................... 6 Exercise 1: Titration of two drinks ................................................................................ 7 Post Lab Analysis ........................................................................................................... 7 Creative Commons Licensing ........................................................................................ 8 U.S. Department of Labor Information ......................................................................... 8 1|Page Last Updated May 26, 2015 LEARNING OBJECTIVES: After completing this laboratory experiment, you should be able to do the following things: 1. Understand the role of citric acid in food products. 2. Determine the concentration of an unknown concentration of citric acid using titration. o Report the uncertainty (experimental error) in this result. 3. Describe the purpose and mechanism of a titration. 4. Describe the interactions of multiple acidic protons in a polyprotic weak acid. 5. Explain the conditions at the equivalence point for the titration of citric acid with sodium hydroxide. BACKGROUND INFORMATION: Citric acid is a naturally-occurring weak acid that is not only critical to the energy product cycle of most cells (known as the Krebs cycle) but is also used in thousands of food, drinks, and household products. Like any acid, citric acid has a sour taste and is the reason that citrus fruits (for which the acid is named) taste sour. Citric acid is commonly confused with so-called “Vitamin C”, which is a different weak acid, ascorbic acid (Error! Reference source not found.). Although citric and ascorbic acids are both found in most citrus fruits, they are very different from one another. In most citrus fruits, citric acid is present in concentrations of 20 to 100 times that of ascorbic acid (Nour). Although it looks like citric acid might be tetra-protic, only three of the protons are acidic, enough to be removed during a normal titration with a strong base. Notice that each of the three acidic protons in citric acid look very similar to one another in terms of the chemical bonds around them. This means that they will have similar acidic characteristics. Since the “middle” acidic proton (at the top of Error! Reference source not found.) is closer to the non-acidic hydroxide group (at the middle bottom of Error! Reference source not found.), it is the easiest to remove from the molecule, because the oxygen in the hydroxide group is pulling electron density away from it more than from the other two acidic protons on either side of the molecule. The acid equilibrium constants for the three acidic protons in citric acid are 2|Page Last Updated May 26, 2015 (Weast): Ka1 Ka2 Ka3 8.4 x 10-4 1.8 x 10-5 4.0 x 10-6 Table 1: Acid Equilibrium Constants for Citric Acid Citric acid is used in a very large variety of food products for various reasons. Citric acid can be used to provide sourness in many drinks and foods, as a way to help curdle cheese, as an anticoagulant, to help preserve dye colors, and also can be used in various industrial processes such as electroplating and waste-stream conditioning (NCBI). Citric acid content in juices and drinks is important to know, because it can be an aid for people who suffer from kidney stones and other diseases of the renal system (Penniston). There are many methods of determining the citric acid content of foods and drinks, but we are going to focus on one of the oldest: titration with a strong chemical base. Like any weak acid, citric acid can be titrated with a strong base of known concentration. Unlike monoprotic weak acids, however, there are three protons that must be removed during the titration process. Citric acid is kind of unique among polyprotic acids in that its three protons are so similar to each other. This gives some unique features to its titration curve, which you will discover as you do this activity. ESTIMATING ERROR: You can always estimate the error in any measurement by taking several measurements of it and calculating the standard deviation of the set of results. In other words, if you measure something several times, you are likely to end up with a set of results that are not all exactly the same number. The standard error of that set of numbers is an estimate of how much error there is in your results, and it is equal to the standard deviation divided by the square root of the number of samples in the data set. Here’s an example. Let’s say we measured something five times, and the result was: 3|Page Last Updated May 26, 2015 Trial #1 1 Measurement 0.324 2 0.332 3 0.325 4 0.328 5 0.322 We would report that the value was: average +/- standard error, which in this case is: 0.326 +/- 0.002. This means that the uncertainty in your result (0.326) starts in the third decimal place, because that’s the first digit in your standard error. Since the uncertainty starts in the thousandths place, we have to limit the average value to the thousandths place as well. When reporting uncertainty in this way, you always report only one digit in the standard error. The standard deviation can be calculated manually (you’ve probably done this in a math course), or you can use a spreadsheet program like Excel. If you have fewer than 100 samples to work with, use the “stdev.s” function, which is more accurate for small samples. Sources: Nour, V., Trandafir, I., Ionica, M. E. HPLC Organic Acid Analysis in Different Citrus Juices under Reversed Phase Conditions. Not. Bot. Hort. Agrobot. Cluj 2010, 38, 1, pp. 44-48 Weast, R. C., Ed. Handbook of Chemistry and Physics, The Chemical Rubber Company, Ohio, U.S. 1968 NCIB - National Center for Biotechnology Information. Compound Summary for Citric Acid, http://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=311#x94 (accessed Oct 2, 2014) Penniston, K. L., Nakada, S. Y., Holmes, R. P., Assimos, D. G. Quantitative Assessment of Citric Acid in Lemon Juice, Lime Juice, and Commercially-Available Fruit Juice Products. J Endourol. Mar 2008; 22, 3, pp. 567–570. 4|Page Last Updated May 26, 2015 PRE-LAB QUESTIONS: 1. When you titrate citric acid with a strong base, do you expect the titration curve to look more like a or b below? 2. Explain the reasoning for your choice in question 1. 3. Why do the second and third protons on the citric acid molecule have different values of Ka, even though they are basically equivalent? EQUIPMENT: 1. 2. 3. 4. Paper Pencil/pen Computer with Internet access (for the remote laboratory and for data analysis) A spreadsheet program such as Excel 5|Page Last Updated May 26, 2015 PREPARING FOR THIS NANSLO LAB ACTIVITY: Read and understand the information below before you proceed with the lab! Scheduling an Appointment Using the NANSLO Scheduling System Your instructor has reserved a block of time through the NANSLO Scheduling System for you to complete this activity. For more information on how to set up a time to access this NANSLO lab activity, see www.wiche.edu/nanslo/scheduling-software. Students Accessing a NANSLO Lab Activity for the First Time For those accessing a NANSLO laboratory for the first time, you may need to install software on your computer to access the NANSLO lab activity. Use this link for detailed instructions on steps to complete prior to accessing your assigned NANSLO lab activity – www.wiche.edu/nanslo/lab-tutorials. Video Tutorial for RWSL: A short video demonstrating how to use the Remote Web-based Science Lab (RWSL) control panel for the titration apparatus can be viewed at http://www.wiche.edu/nanslo/lab-tutorials#titration. NOTE: Disregard the conference number in this video tutorial. AS SOON AS YOU CONNECT TO THE RWSL CONTROL PANEL: Click on the yellow button at the bottom of the screen (you may need to scroll down to see it). Follow the directions on the pop up window to join the voice conference and talk to your group and the Lab Technician. EXPERIMENTAL PROCEDURE: Read and understand these instructions BEFORE starting the actual lab procedure and collecting data. Feel free to “play around” a little bit and explore the capabilities of the equipment before you start the actual procedure. Once you have logged onto the remote lab, you will perform the following laboratory procedures: 6|Page Last Updated May 26, 2015 EXERCISE 1: Titration of Two Drinks Before you begin, ask the Lab Technicians to tell you the name and standardized concentration of the strong base that you will be using to titrate with. You will have two samples of orange juice and two samples of a sports drink that you can titrate with the strong base. Hint: For each addition of titrant, add small quantities of the strong base titrant (less than 0.5 mL) so you don’t over-shoot the equivalence point. 1. Titrate the orange juice samples and download the graph image and data and paste them into a document for later analysis. 2. Titrate the sports drink samples and download the graph image and data and paste them into a document for later analysis. 3. Ensure that each person in the group gets a chance to do at least one full titration. 4. Ensure that each person in the group downloads and saves all the graph images and data sets. POST-LAB ANALYSIS (Can be done offline if you run out of time) 1. Each drink sample you titrated was composed of 5.0 mL of the original drink (orange juice or sports drink) and some water to provide enough volume to do the titration. Use the data you collected to determine the moles of citric acid in each sample you titrated. 2. Use the data you calculated in the first step to determine the average concentration of citric acid in the original drinks. 3. Report the experimental error in each average value. 4. Does the titration curve match the expectation you had in the pre-lab assignment? Do some research (cite your sources) and try to explain why the titration curve of citric acid has the shape that it does. 7|Page Last Updated May 26, 2015 For more information about NANSLO, visit www.wiche.edu/nanslo. All material produced subject to: Creative Commons Attribution 3.0 United States License 3 This product was funded by a grant awarded by the U.S. Department of Labor’s Employment and Training Administration. The product was created by the grantee and does not necessarily reflect the official position of the U.S. Department of Labor. The Department of Labor makes no guarantees, warranties, or assurances of any kind, express or implied, with respect to such information, including any information on linked sites and including, but not limited to, accuracy of the information or its completeness, timeliness, usefulness, adequacy, continued availability, or ownership. 8|Page Last Updated May 26, 2015