File ch 10 states of matter guided reading notes
advertisement

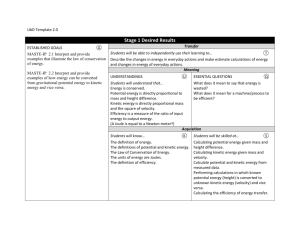
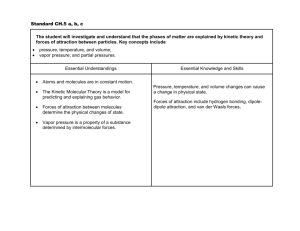
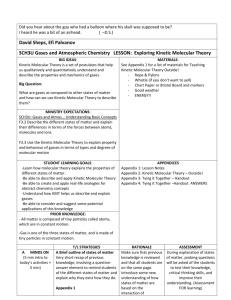
CH 10 States of Matter 10.1 The Kinetic Molecular Theory Objectives State the kinetic molecular theory of matter and describe how it explains certain properties of matter List the 5 assumptions of the kinetic molecular theory of gasses. Define the terms ideal gas and real gas Describe each of the following properties of gasses: expansion, density, fluidity, compressibility, diffusion and effusion Describe the conditions under which a real gas deviates from ideal behavior What is the Kinetic Molecular Theory? Know the 5 assumptions of the kinetic molecular theory The kinetic molecular theory applies only to ____________________________________________ Under what circumstances do real gasses behave like ideal gases? You should be able to explain all the properties of a gas using the kinetic molecular theory. Each explanation should begin with “Because the particles…………….” Use the arrangement and motion of the gas particles to explain each property. Why don’t real gasses behave like ideal gases? Under what conditions do real gasses most differ from ideal gasses? 10.1 assignment address the objectives p 314 1 to 7 p 336 1 to 6 ws 1,2 10.2 Liquids Objectives Describe the motion of particles in liquids according to the kinetic molecular theory Discuss the process by which liquids can change into a gas. Define vaporization Discuss the process by which liquids can change into a solid. Define freezing What is the least common state of matter in the universe? Explain Why do liquids have a definite volume and shape? Why are liquids hundreds of times more dense than gasses? Why can’t a liquid be compressed? Why can it diffuse? What causes surface tension? Why does water have a high surface tension? How are surface tension and capillary action related? Why do liquids form nearly spherical drops? Does every single molecule in a liquid have exactly the same kinetic energy at a given temperature? What is the difference between boiling and evaporation? At what temperatures do they occur? 10.2 assignment address the objectives p 318, 1 to 6 p336, 7 to 8 ws3,4 10.3 Solids Objectives Describe the motion of particles in solids and the properties of solids according to the kinetic molecular theory Distinguish between the 2 types of solids Describe the 2 types of crystal symmetry. Define crystal structure and unit cell Why are amorphous solids referred to as supercooled liquids? Name an amorphous solid Know the forces that hold the different types of crystal together. Be able to give an example of each kind of crystal. Which type of crystal has the strongest forces holding it together? Which kind of crystal has the highest melting point? Which type of crystal has the weakest forces holding it together? Which kind of crystal has the lowest melting point? What does amorphous mean 10.3 assignment address the objectives p323, 1 to 6 p336, 10 to 12 ws5,6 10.4 Changes of State Objectives Explain the relationship between equilibrium and changes of state Interpret phase diagrams Explain what is meant by equilibrium vapor pressure Describe the process of boiling, freezing, melting and sublimation Remember, you cannot see a gas. What you can see is a vapor. What is a vapor? In fig 4.2 p 325, could equilibrium be reached if the flask was not stoppered? Remember, temperature in the measure of the average kinetic energy of the particles Why does increasing the temperature increase the equilibrium vapor pressure? Do volatile liquids have a high or low vapor pressure? Explain Where, in the liquid, does boiling occur? What happens to the boiling point if the vapor pressure is raised? While boiling, energy in the form of heat, is still being supplied. What is this energy being used to do if not raise the temp? Enthalpy can be thought of as heat energy for now. What does molar enthalpy of vaporization measure? Why is water’s so high? What happens to the enthalpy of vaporization if you increase the amount of liquid? What are at equilibrium at the freezing point? In what phase or phases is water at 100C? At 0C? Are the melting point and freezing point the same? When a substance freezes, is it energy in or out? How about when it melts? p 328, 4th paragraph. What does it mean when it states that adding energy will cause the equilibrium to shift to the right? What will be present in greater quantity, liquid or water? Equilibrium does NOT mean that equal amounts of solid and liquid are present. It means that the melting and freezing processes are occurring at the same rate. What do both molar enthalpy of fusion and molar enthalpy of vaporization depend upon? Why does a thin layer of snow disappear even though the temp is below 0C? Use the Phase Diagram on p 329 to answer the following questions: At what temp and pressure can water exist in all 3 phases? At what temp can water no longer exist as a liquid What is the phase of water at 100 atm and 100C What is the phase of water at 100 atm and 200C? What happens to the melting point of ice as the pressure decreases? P 330 fig 4.6 label the green arrows as endothermic or exothermic. Do the same for the blue arrows 10.4 assignment address the objectives p 330 1 to 7, 336, 13 to 18, adv also do 19 to 22 ws 7,8 10.5 Water Objectives Describe the structure of the water molecule Discuss the physical properties of water and explain how they are determined by the structure of water Calculate the amount of energy absorbed or released when a quantity of water changes state. ADV only Why is water a liquid and not a gas at room temp? What accounts for the relatively low density of ice? Is hydrogen bonding an inter or intra molecular force? At what temperature is water most dense? What prevents large body of water from freezing solid? Compare the molar enthalpies of fusion and vaporization of water to other similar molecules Explain why water’s high molar enthalpy of vaporization makes water a good choice for heating systems. Do practice probs 1,2 on p 333 ADV only 10.5 assignment address the objectives p333, 1 to 7 Ch 10 review assignment p334, 1,2 ADV only p 337 DV 27-35, CP 33-35 p337, 23 to 26 ws 9,10 p339 ADV 1 to 11 CP 1 to 8 WS11,12