File
advertisement

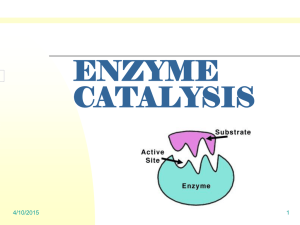
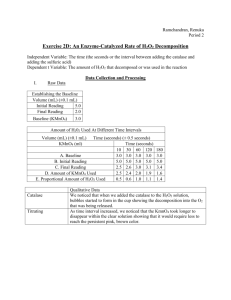
Laboratory extension notes. Post lab discussion Question 1c - What could be done to more accurately determine the unknown concentration. Several things could be done. You could use a light meter or colorimeter to accurately assess the frequency of the light waves of the unknown sample and then obtain a concentration from a colour intensity vs concentration plot made from known standards. Alternatively you could do smaller serial dilutions. For example ½ dilutions rather than 1/10 dilutions to more accurately estimate the concentrations. Lastly you could titrate the solution with a known concentration of reactant and calculate concentration from the number of moles of reactants. For example: Chemical equation for titration of KMno4 and H2O2 2MnO4–(aq) + 3H2O2(aq) → 2MnO2(s) + 2H2O(l) + 3O2(g) + 2OH–(aq) In this reaction we know the ratio of H2O2 with KmnO4. This is 2 KMnO4 : 3 H2O2 If we know the concentration and volume of hydrogen peroxide which reacts with the KMnO4 then we can calculate exactly the number of moles of KMnO4 in the reaction flask From this we can calculate a concentration given a specific volume. Example: 25.2ml of a 1M H2O2 solution is reacted with 250ml of KMnO4. The reaction is done to completion with no excess reagent. What is the concentration of the KMnO4 solution? Step 1 (Calculate n H2O2) n(H2O2) = CV Step 2 (Calculate n (KMnO 4) n (KMnO 4) = (2/3)x n(H2O2) = 1 x 0.0252 = 2/3 x 0.0252 = 0.0252 M = 0.0168 Step 3 (Calculate [KMnO4]) C (KMnO4) = n / v = 0.0168 / 0.25 = 0.0672M