Name: IB Chemistry Summer Assignment This assignment is for all
advertisement

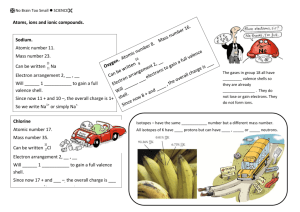
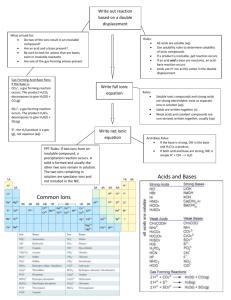
Name: ________________________________ IB Chemistry Summer Assignment This assignment is for all students enrolled in IB Chemistry HL for the 2014 – 2015 academic year. The assignment is due on the first day of class and will be counted. Show all work or credit will not be given. The assignment is outlined below: Read and study Chapters 1, 2, and 3 in the grey text, Chemistry, The Central Science, Brown, LeMay, and Burstein, 11th edition. Be prepared to take a test on the material shortly after you arrive in August. 1. Complete exercises in Chapter 1 on pages 30 – 35. a. Visualizing Concepts: # 1, 3, 6, 8, 9 b. Classification of Properties and Matter: # 11, 13, 15, 20 c. Units of Measurement: 23, 25, 27, 29 d. Uncertainty in Measurement: 33, 35, 37 e. Dimensional Analysis: 41, 43, 49 2. Complete exercises in Chapter 2 on pages 69 – 77 a. Visualizing Concepts: # 2, 4, 5, 7 b. Atomic Theory and the Discovery of Atomic Structure: # 10, 11, 16 c. Modern View of Atomic Structure: # 20,21,22,24,26.31,33 d. The Periodic Table; Molecules and Ions: #40,41,44,48,49,55, 58 e. Naming Organic Compounds; Organic Molecules: #61,63,66,68,69,73 3. Complete exercises in Chapter 3 on pages 108 – 114 a. Visualizing Concepts: # 3, 5,7 b. Balancing Chemical Equations:#9,11,13 c. Patterns of Chemical Reactivity: # 15,19 d. Formula Weights: #22,23,26 e. Avogadro’s number and the mole: #27,29,33,38 f. Empirical formulas: # 43,45,47,51 g. Calculations based on Chemical Equations: # 56, 57, 59 h. Limiting Reactants and Theoretical Yield: #67,69,71,73, 77, 79 4. Use the list of cations and anions below to make names for each cation with each anion(60 compounds). Remember to use rules for naming ionic compounds(Stock System) Cations Anions Aluminum Bromide Ammonium Carbonate Calcium Ethanoate(acetate) Chromium(II, III) Hydroxide Copper (I, II) Titanium (II, IV) Vanadium (V) Nitrate phosphate 5. The following acids and ions will be used throughout the course. You will be expected to know these acids and ions. Acids Polyatomic Ions Hydrochloric acid Ammonium Sulfuric acid Acetate Phosphoric acid Hydrogen carbonate Acetic acid Hydroxide Nitric acid Nitrate Carbonic acid Nitrite Nitrous acid Permanganate Sulfurous acid Carbonate Sulfate Sulfite phosphate 6. Give the name or formula for each of the following on the line. a. K2SO4 ________________________________________________________________________ b. P2O5 _______________________________________________ c. N3F7 _______________________________________________ d. Mg3N2 ______________________________________________ e. Mg(OH)2 ____________________________________________ f. Aluminum nitride _____________________________________ g. Phosphorus pentabromide ______________________________ h. Dichlorine heptoxide ___________________________________ i. Silver (I) chloride ______________________________________ j. Tin (IV) hydroxide _____________________________________ 7. For each of the following reactions, identify the type and predict the products. Assume all reactions will occur. a. Ag2O ------> __________ + ____________ ___________________________________________________ b. C4H8 + O2 ----> ________ + _________ ___________________________________________________ c. Al + N2 ----> ___________ ___________________________________________________ d. Cu + ZnSO4 ---> ____________ + ___________ ___________________________________________________ e. Al2(SO4)3 + Ba(OH)2 ----> ___________ + __________ ___________________________________________________