SUPPLEMENTARY DATA OUTLINE - Springer Static Content Server
advertisement

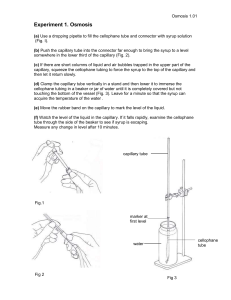
SUPPLEMENTARY INFORMATION For JASMS manuscript: Bipolar Mass Spectrometry of Labile Co-ordination Complexes, Redox Active Inorganic Compounds and Proteins Using a Glass Nebulizer for Sonic-Spray Ionization M. M. Antonakis,† A. Tsirigotaki,† K. Kanaki,† C. J. Milios‡ and Spiros A. Pergantis*,† † Environmental Chemical Processing Laboratory and ‡Division of Inorganic Chemistry, Department of Chemistry, University of Crete, Voutes Campus, Heraklion 71003, Greece Supplementary Information Content: Figure S1. Modified Meinhard® pneumatic glass nebulizer used for V-EASI-MS: (1) fused silica capillary located at glass nebulizer tip; (2) nut and PEEK sleeve used to secure fused silica capillary in place; (3) fused silica capillary going to sample; (4) 40-60 psi N2 gas line. Figure S2. (a) Meinhard® V-EASI source coupled to mass spectrometer (LCQ, Thermo). Inset shows top view of fused silica capillary coming out of the back of nebulizer and going into sample tube; and (b) close-up of V-EASI nebulizer next to mass spectrometer entrance. Figure S3. ES mass spectrum acquired from the analysis of 100 µg mL-1 cytochrome c in 80% DMSO, at 300 oC heated inlet capillary and source induced dissociation potential of 15 V. Figure S4. Negative ion mass spectrometric analysis of the [Cu6Dy] complex in acetonitrile solution, using V-EASI-MS with a heated ion transfer capillary at 205 oC and sid voltage at 15 V (a), and using ES-MS with a heated ion transfer capillary at 205 oC and sid voltage at 10 V (b). Figure S5. Time course stability of Se(IV) species of selenous acid in solution as screened by negative ion V-EASI-MS analysis. Expected peak for the oxidized analyte [Μ+Ο–Η]- is at m/z 145. Figure S6. Negative ion mass spectra for arsenite using V-EASI-MS (a), and ES-MS at 4 kV spray voltage (b). Figure S7. Negative ion mass spectrum obtained using a dual V-EASI nebulizer configuration for the separate aspiration of [Cu6Tb] in acetonitrile and STFA in acetonitrile/water. Numbers in green correspond to STFA ions, whereas numbers in blue correspond to [Cu6Tb] complex ions. Masses in boxes represent bracketing masses for potential accurate mass measurements. Figure S2. Modified Meinhard® pneumatic glass nebulizer used for V-EASI-MS: (1) fused silica capillary located at glass nebulizer tip; (2) nut and PEEK sleeve used to secure fused silica capillary in place; (3) fused silica capillary going to sample; (4) 40-60 psi N2 gas line. a b Figure S2. (a) Meinhard® V-EASI source coupled to mass spectrometer (LCQ, Thermo). Inset shows top view of fused silica capillary coming out of the back of nebulizer and going into sample tube; and (b) close-up of V-EASI nebulizer next to mass spectrometer entrance. CYT_100ppm _80% DMSO #2-25 RT: 0.07-0.97 AV: 24 NL: 5.56E4 T: + p ESI s id=20.00 Full m s [ 100.00-2000.00] 1767.1 100 95 90 85 80 75 70 1546.5 Relative Abundance 65 60 55 50 45 40 1125.1 35 1375.0 30 1237.5 25 1031.5 20 1551.3 15 1772.7 10 5 0 200 400 600 800 1000 1200 1400 1600 1800 2000 m /z Figure S3. ESI (a) and SSI (b) mass spectra acquired from the analysis of 100 µg mL-1 cytochrome c in 80% DMSO, at 300 oC heated inlet capillary and source induced dissociation potential of 15-20 V, [Μ+NO3]100 a 1630.6 2.43E4 Relative Abundance 50 1718.4 1547.7 0 100 1898.9 1553.1 1.83E4 b 1372.5 1613.5 50 1902.7 824.5 1657.1 1840.3 188.9 339.1 408.5 557.6 981.1 692.4 1066.7 1163.9 1511.2 1739.5 1926.9 1280.3 0 200 400 600 800 1000 1200 1400 1600 1800 2000 m/z Figure S4. Negative ion mass spectrometric analysis of the [Cu 6Dy] complex in acetonitrile solution, using V-EASI-MS with a heated ion transfer capillary at 205 oC and sid voltage at 15 V (a), and using ES-MS with a heated ion transfer capillary at 205 oC and sid voltage at 10 V (b). [M-H2O]- t=0days t=3days e c a d n u b A Relative Abundance [M-H]- tiv la e R t=14days m/z Figure S5. Time course stability of Se(IV) species of selenous acid in solution as screened by negative ion V-EASI-MS analysis. Expected peak for the oxidized analyte [Μ+Ο–Η]- is at m/z 145. [M-H]Relative Abundance 100 107.1 4.22e2 a As(III) 60 141,0 20 100 3.61e2 123.1 107.1 60 b [M-H+O]As(V) As(III) 20 80 85 90 95 100 105 110 115 120 125 130 135 140 145 150 155 160 m/z Figure S6. Negative ion mass spectra for arsenite using V-EASI-MS (a), and ESMS at 4 kV spray voltage (b). 792.80 100 [M+NO3]1628.33 1200.60 80 1064.60 1608.5 1336.60 928.73 60 1666.5 1472.47 656.87 1744.40 40 289.00 520.87 1880.40 20 248.87 195.07 1948.47 384.87 0 200 400 600 800 1000 1200 1400 1600 1800 2000 m/z Figure S7. Negative ion mass spectrum obtained using a dual V-EASI nebulizer configuration for the separate aspiration of [Cu6Tb] in acetonitrile and STFA in acetonitrile/water. Numbers in green correspond to STFA ions, whereas numbers in blue correspond to [Cu6Tb] complex ions. Masses in boxes represent bracketing masses for potential accurate mass measurements.