Organic Chem Notes
advertisement

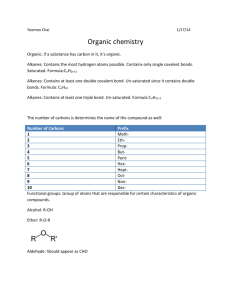
Organic Nomenclature H HCH H methane H HC H ethane propane butane H C H H HC H H HC H H CH H H C H H CH H H C H H CH H Organic chemistry originated as the chemistry of living systems. In this context organic meant “natural”. But as chemists studied the molecular structure of living things, it quickly became obvious that all organic compounds contained carbon as their main constituent. Thus organic chemistry today is defined as the chemistry of carbon compounds. In the compounds carbon atoms are covalently bonded to other atoms in chains. Since carbon has a valence of four, each carbon atoms forms four bonds; because of this, the number of possible organic compounds is limitless, making it the perfect atom for life to be based upon. Today chemists have learned to imitate nature and synthesize organic molecules in the laboratory. Such synthetic organic molecules have become the basis of many industries (such as plastics, textiles, and medicine) and therefore organic chemistry employs more people than all other fields of chemistry combined. In this chapter we will study the vast variety of organic compounds found in nature and those made by man. We will base our study on the system that is called Organic Nomenclature. There are actually two systems of Organic Nomenclature now in use; the older system consists of common names for organic compounds, the newer system, called the IUPAC system is the one which we shall study in this chapter. H HC H H H H H HC H C C C H H H H H H H C C C C H H H H H CH H pentane H CH H hexane The IUPAC names shown on this page with their structural formulas are the basis for all the names of the organic compounds. The molecules shown here are all members of a group of compounds called alkanes. On the next page we will begin listing the categories of organic chemistry in outline form. heptane nonane octane decane Hydrocarbons Saturated Hydrocarbons 1. Alkanes Hydrocarbons are the simplest category of organic compounds. They contain only hydrogen and carbon. Saturated hydrocarbons are those which contain the maximum amount of hydrogen possible. Alkanes are hydrocarbons made up of only single covalent carbon to carbon bonds. The molecular formulas and names of the first ten alkanes are as follows. CH4 C2H6 C3H8 C4H10 C5H12 methane ethane propane butane pentane C6H14 C7H16 C8H18 C9H20 C10H22 hexane heptane octane nonane decane The names of all alkanes end in “ane”. Their molecular formulas have the general formula C nH2n +2. 2. Branched Alkanes Alkanes do not always have their carbons linked together in a straight line. Alkanes may be branched. The names of the branches are based on the names of the straight chained alkanes with ending “ane” changed to “yl”. The following is a list of the names of possible branches. CH3 C2H5 C3H7 C4H9 C5H11 methyl ethyl propyl butyl pentyl C6H13 C7H15 C8H17 C9H19 C10H21 hexyl heptyl octyl nonyl decyl To name a branched alkane by the IUPAC system we name the longest chain of carbons using the standard alkane names. The names of the branches are listed in alphabetical order immediately preceding the name of the longest chain. Each branch is given a number by its position on the main chain. The main chain is always numbered in the direction to give the smallest possible numbers for the branches. The resulting name is one word. Sample Problem 1 H HCH H HCH H HC H H HCH H HCH HCH H H H C C C C CH H H H H H Results in the name H HC H C H HCH H C H C H H C H H CH H If there is more than one of the same kind of branch a prefix is used to indicate how many there are. But even in this case, a number is still required for each of the branches present. The prefixes are as follows: di for two tri for three Sample Problem 2 H HCH HCH HCH H H H HC H C C C H H H Sample Problem 3 Sample Problem 4 H HCH H C HCH H H C CH H H tetra for four Problems For each problem: a) Circle the longest chain of carbons. b) Draw a box around each branch. c) Write the IUPAC name next to the structural formula. For 7-10 Draw the structure for each IUPAC name. 7) 2,2 – dimethyl - 4- propyloctane 8) 2,3,3,5 - tetramethylhexane 9) 3-ethyl-4,4-dipropylheptane 10) 3-ethyl - 2,2 - dimethylpentane 3. Halogenated Alkanes Halogen atoms may be attached to a carbon chain much like methyl group. The rules for numbering and listing halogen atoms are the same as for carbon branches. The following names are used: -F flouro -Br bromo - Cl chloro -I iodo Sample Problem 5 H HC H Br C Br C H HCH H H CH H Unsaturated Hydrocarbons 4. Alkenes Unsaturated hydrocarbons are those which contain less than the maximum number of hydrogens possible. Alkenes contain a double bond between carbon and therefore have 2 less hydrogens than their alkane counterparts. Their molecular formulas have the general formula CnH2n. Alkene names are the same as those for the alkanes except that the “ane” ending is changed to “ene”. The IUPAC system requires that a number be placed immediately in front of the name for the longest chain indicating where the double bond begins. The main chain is numbered from the end to give the double bond the smallest possible number, regardless of where the branches are placed. Sample Problem 6 H HC H HCH C HCH H H H H C C CH H When there is more than one double bond, the standard prefixes (di, tri, or tetra) are placed immediately in front of the “ene” ending. The letter “a” is inserted in front of this prefix to make the name pronounceable. As would be expected, there must be a number for each of the double bonds. Sample Problem 7 H HC H H C H C H C C HCH H H CH H In the sample problem above, numbering from either direction will give the same numbers for the double bonds. Therefore our second consideration is to give the branch the smallest number possible. But as demonstrated in the next sample problem, the double bonds have priority in receiving the smallest number. Sample Problem 8 H HC H H H H H H H C C C C C C CH H HCH H 5. Alkynes Alkynes are unsaturated hydrocarbons containing a triple bond between carbons. The name of all alkynes end in “yne”. Their general formula is CnH2n-2. Sample Problem 9 HC CH Sample Problem 10 HC C C CH Problems: Write the IUPAC name for each of the following.