Lewis Diagram Notes- Student Version

Date: __________________
Lewis Diagrams
Name:__________________
Who invented Lewis diagrams?
A _____________________________________ is a diagram that illustrates chemical bonding by showing only an atom’s ______________________ _____________________ and the chemical symbol.
(Can also be called, Lewis structures or Electron dot diagrams).
WHAT IS THE DIFFERENCE BETWEEN BOHR DIAGRAMS AND LEWIS DIAGRAMS?
Lewis diagrams can be drawn by following these rules:
1) Dots representing electrons are placed around the element symbols at the
______________________________________________________________.
2) Electron dots are placed in singles until the fifth electron is reached, then they are paired.
(Always start EAST).
-***The electrons shown are only the ones in the __________________________________***-
Lewis diagrams of IONS can be drawn by following these rules:
(remember an ion is an atom with an unequal number of electrons).
-_________________________________ are where one electron is removed from the valence shell for each positive charge. The element symbol will remain with the corresponding charge added and encased in square brackets.
-_________________________________ are where one electron is added to the valence shell for each negative charge. The element symbol is surrounded by 8 electron dots (with the exception of H). Square brackets are placed around the diagram with a negative charge in the upper top right corner.
EXAMPLE: NaCl
Lewis diagrams of COMPOUNDS
Lewis diagrams can also show covalent bonds:
EXAMPLE: HF
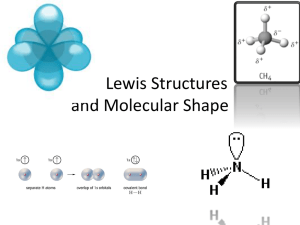
Lewis diagrams of covalent molecules (chemical bonding):
EXAMPLE: H
2
O, NH 3 and CH 4
Lewis diagrams of diatomic molecules:
-A _________________ __________________ is a pair of atoms that are joined by covalent bonds. Diatomic molecules can form this way because the two-atom molecules are more stable than individual atoms. (Mostly non-metals).
EXAMPLES: F 2 , H 2 , N 2 , O 2 , CL 2 , Br 2 and
I
2